Advanced Synthesis of Benzotriazole Dyes for High-Efficiency Visible Light Photoinitiation Systems
Advanced Synthesis of Benzotriazole Dyes for High-Efficiency Visible Light Photoinitiation Systems
The rapid evolution of laser imaging and photocuring technologies has created an urgent demand for photofunctional materials capable of responding to long-wavelength visible light rather than limited ultraviolet spectra. Patent CN101885924B introduces a groundbreaking class of benzotriazole dye compounds designed specifically to address the spectral limitations of traditional photoinitiators. By chemically modifying the benzotriazole core structure, this innovation successfully extends the absorption wavelength to over 400nm, enabling efficient visible light polymerization of vinyl monomers. This technical breakthrough positions these compounds as superior candidates for next-generation photosensitive systems, offering a reliable photosensitizer supplier solution for industries ranging from photoresists to advanced coating formulations. The structural versatility allows for R groups such as OH or OCH3, providing tunable physicochemical properties for diverse application requirements.
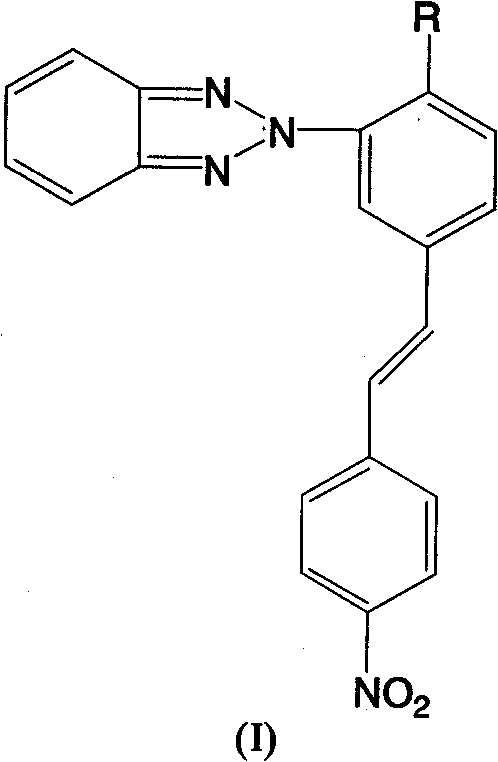
As a leading entity in fine chemical manufacturing, we recognize that the transition from UV to visible light curing represents a significant paradigm shift in material science. The compounds described in this patent exhibit not only extended spectral absorption but also demonstrate robust fluorescence emission, making them dual-functional materials suitable for both initiation and sensing applications. The synthesis pathway outlined in the patent avoids the complexity often associated with heterocyclic dye synthesis, utilizing accessible starting materials and standard reaction conditions. This accessibility is crucial for supply chain stability, ensuring that high-purity benzotriazole derivatives can be produced consistently without reliance on scarce or highly regulated precursors. For procurement managers seeking cost reduction in photocurable material manufacturing, this route offers a compelling alternative to expensive squaraine-based systems.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional photoinitiator systems have long been dominated by compounds such as benzophenone, trichloromethyl-s-triazine, and iodonium salts, which are highly efficient but fundamentally restricted to the 200-300nm ultraviolet region. This limitation poses significant challenges for applications requiring deeper penetration or safer operating conditions, as UV radiation can be hazardous and is often absorbed by pigments or fillers in the formulation. Previous attempts to utilize visible light sensitizers, such as squaraine dyes, have encountered substantial hurdles regarding solubility and initiation efficiency. As noted in prior art, squaraine dyes often exhibit poor solubility in common organic solvents, complicating the formulation process and leading to heterogeneous mixtures that compromise the mechanical integrity of the cured polymer. Furthermore, the initiation efficiency of these legacy visible light systems frequently falls short of industrial requirements, necessitating higher loading levels that can adversely affect the optical clarity and thermal stability of the final product.
The Novel Approach
The novel approach detailed in patent CN101885924B circumvents these historical bottlenecks by leveraging the inherent stability and tunability of the benzotriazole scaffold. Through a strategic sequence of diazotization, reductive cyclization, and condensation reactions, the synthesis creates a conjugated system that effectively shifts the absorption maximum into the visible region. Unlike the complex and often unstable squaraine structures, these benzotriazole derivatives maintain excellent solubility profiles, facilitating homogeneous dispersion within resin matrices. The ability to function as a visible light photosensitizer in conjunction with standard UV initiators like iodonium salts creates a synergistic effect, generating free radicals through intermolecular photoinduced electron transfer under visible light irradiation. This method not only broadens the operational window for photocuring processes but also enhances the overall sensitivity of the system, allowing for faster cure times and lower energy consumption during manufacturing.
Mechanistic Insights into Reductive Cyclization and Condensation
The core of this synthetic strategy lies in the construction of the benzotriazole ring via a reductive cyclization mechanism, a critical step that defines the purity and yield of the final photosensitizer. The process begins with the diazotization of o-nitroaniline using hydrochloric acid and nitrous acid at ice-bath temperatures to generate the reactive diazonium salt intermediate. This electrophilic species is then coupled with p-hydroxybenzaldehyde under alkaline conditions to form the azo precursor, 2-(2-nitro)azobenzene-4-formaldehyde-phenol. The subsequent transformation involves a sophisticated reductive cyclization where zinc powder acts as the reducing agent in a sodium hydroxide medium. This step is pivotal as it simultaneously reduces the nitro group and facilitates the intramolecular nucleophilic attack of the resulting amine on the adjacent nitrogen, closing the triazole ring to form 2-(2-hydroxy-5-formyl)-2H-benzotriazole.
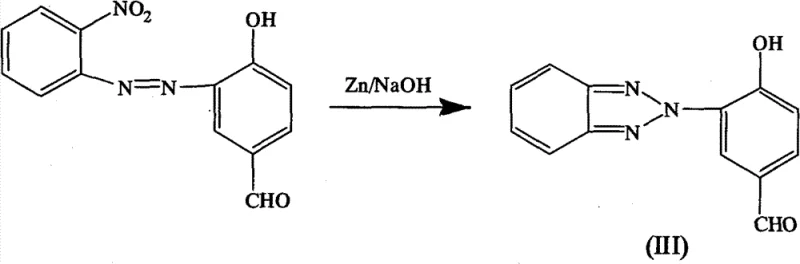
Following the formation of the benzotriazole core, the synthesis proceeds to functionalize the molecule to optimize its electronic properties for visible light absorption. The final stage involves a Knoevenagel condensation reaction between the aldehyde group on the benzotriazole ring and p-nitrophenylacetic acid. Catalyzed by piperidine at elevated temperatures ranging from 100°C to 130°C, this condensation extends the pi-conjugation system across the molecule, which is the primary driver for the red-shift in absorption wavelength. For variants requiring enhanced lipophilicity or specific solubility characteristics, an optional methylation step can be introduced using methyl iodide and potassium carbonate with a phase transfer catalyst. This modular approach allows for precise control over the impurity profile, as each intermediate can be purified via silica gel chromatography, ensuring that the final high-purity OLED material or photocuring additive meets stringent quality specifications required by downstream users.
How to Synthesize Benzotriazole Dye Efficiently
The synthesis of these advanced photosensitizers is designed to be operationally simple yet chemically robust, making it highly suitable for commercial scale-up of complex polymer additives. The process relies on standard laboratory glassware such as three-necked flasks and utilizes common reagents that are readily available in the global chemical market. The initial diazotization requires careful temperature control to prevent decomposition of the diazonium salt, while the reductive cyclization demands precise stoichiometric balance between the zinc powder and the azo substrate to ensure complete conversion without excessive metal waste. Detailed standardized synthetic steps see the guide below for specific operational parameters.
- Perform diazotization of o-nitroaniline followed by coupling with p-hydroxybenzaldehyde to form the azo intermediate.
- Execute reductive cyclization using Zinc powder and Sodium Hydroxide at 50-60°C to construct the benzotriazole ring.
- Optional methylation using methyl iodide and potassium carbonate to modify solubility properties.
- Finalize synthesis via Knoevenagel condensation with p-nitrophenylacetic acid using piperidine as a catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this benzotriazole synthesis route offers distinct advantages over legacy photosensitizer technologies, primarily driven by the simplicity of the reaction sequence and the availability of raw materials. The elimination of rare or precious metal catalysts, which are often required in cross-coupling reactions for similar dye classes, significantly reduces the cost of goods sold and mitigates supply risk associated with geopolitical fluctuations in metal prices. Furthermore, the use of zinc and sodium hydroxide, which are commodity chemicals with stable pricing and abundant supply, ensures that production costs remain predictable and manageable even at large volumes. This stability is crucial for procurement managers who need to secure long-term contracts for high-volume manufacturing without exposure to volatile raw material markets.
- Cost Reduction in Manufacturing: The synthetic pathway avoids the use of expensive transition metal catalysts such as palladium or platinum, which typically require costly removal steps to meet residual metal specifications in pharmaceutical or electronic grade products. By utilizing zinc powder for the reductive cyclization, the process simplifies the workup procedure, as zinc salts can be easily removed through aqueous washing and filtration. This reduction in downstream processing complexity translates directly into lower operational expenditures and reduced solvent consumption, contributing to substantial cost savings in photocurable material manufacturing without compromising the quality of the final photosensitizer.
- Enhanced Supply Chain Reliability: The starting materials, including o-nitroaniline and p-hydroxybenzaldehyde, are bulk chemicals produced by numerous suppliers globally, reducing the risk of single-source dependency. This diversity in the supply base ensures continuity of supply even if one vendor faces production issues, a critical factor for supply chain heads managing just-in-time inventory systems. Additionally, the reaction conditions do not require extreme pressures or cryogenic temperatures, allowing the process to be run in standard multipurpose reactors, thereby increasing the flexibility of manufacturing facilities to switch between different products based on market demand.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste compared to alternative dye synthesis routes that might involve heavy metals or chlorinated solvents. The primary byproducts are zinc oxides and salts, which can be treated and disposed of using standard industrial waste management protocols. The scalability of the reaction is supported by the fact that the exothermic nature of the diazotization and condensation steps can be managed effectively with standard cooling and heating systems, facilitating a smooth transition from pilot plant batches to multi-ton annual commercial production while maintaining strict environmental compliance standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this benzotriazole dye technology in industrial settings. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for decision-makers evaluating this technology for adoption. Understanding these nuances is essential for assessing the feasibility of integrating these photosensitizers into existing production lines.
Q: What is the primary advantage of this benzotriazole dye over traditional squaraine dyes?
A: Unlike squaraine dyes which suffer from poor solubility and limited initiation efficiency, this benzotriazole derivative offers extended absorption wavelengths beyond 400nm while maintaining better solubility and fluorescence properties for visible light polymerization.
Q: Can this synthesis process be scaled for industrial production?
A: Yes, the process utilizes common reagents like zinc powder, sodium hydroxide, and standard organic solvents, avoiding expensive transition metal catalysts, which facilitates straightforward scale-up from laboratory to commercial manufacturing.
Q: What is the specific application range of these compounds?
A: These compounds function as visible light photosensitizers that can be combined with UV photoinitiators like iodonium salts or triazines to initiate the polymerization of vinyl monomers such as methyl methacrylate under visible light irradiation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzotriazole Dye Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of benzotriazole dye meets the high performance standards required for visible light photoinitiation systems. We understand that consistency is key in the fine chemical industry, and our commitment to quality assurance means that our customers receive materials with reproducible spectral properties and initiation efficiencies, batch after batch.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific application needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how switching to our benzotriazole derivatives can optimize your formulation costs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions about integrating these high-performance visible light photosensitizers into your next generation of photocurable materials.
