Advanced Synthesis of Triphenylamine-Based Visible Light Photosensitizers for Industrial Applications
Advanced Synthesis of Triphenylamine-Based Visible Light Photosensitizers for Industrial Applications
The rapid evolution of photopolymerization technologies has created an urgent demand for high-performance photosensitizers capable of operating efficiently within the visible light spectrum. Patent CN101602822A introduces a groundbreaking class of visible light photosensitizers featuring a triphenylamine core modified with aldehyde groups, specifically designed to overcome the spectral limitations of traditional UV-only initiators. This innovation represents a significant leap forward for industries relying on photo-curing materials, including advanced coatings, printing inks, and electronic component manufacturing. By chemically modifying the triphenylamine structure, the patent describes a method to red-shift the UV-visible absorption peak to above 370nm, thereby unlocking the potential for visible light polymerization of ethylenic monomers. This capability is critical for developing next-generation optical functional materials that require deep curing and high sensitivity without the hazards associated with high-energy UV radiation.
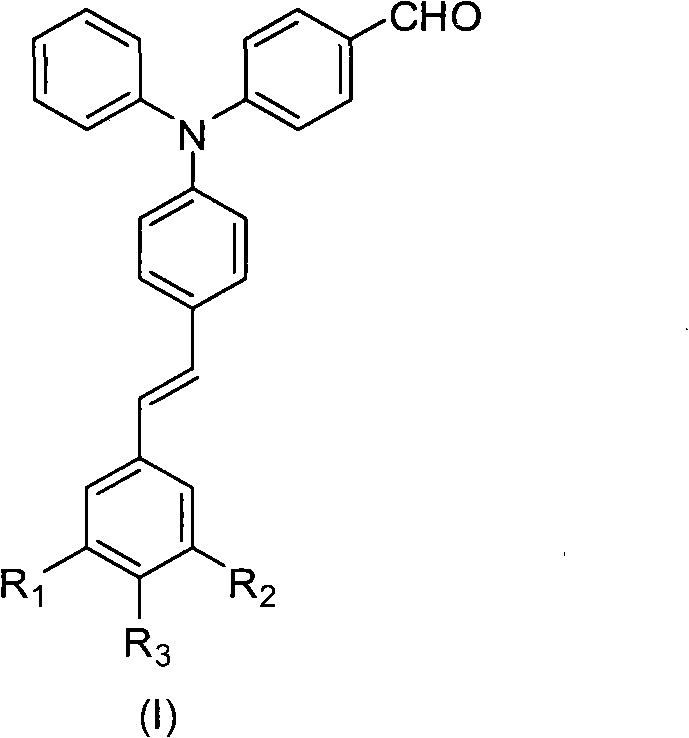
For R&D directors and procurement specialists seeking a reliable photosensitizer supplier, understanding the structural versatility of these compounds is paramount. The general formula presented in the patent allows for extensive customization through various substituents (R1, R2, R3), enabling fine-tuning of solubility, absorption characteristics, and reactivity to match specific application requirements. Whether the goal is to enhance the performance of photo-resists in semiconductor processing or to improve the durability of light-curable inks, this molecular scaffold offers a robust foundation. The ability to achieve a half-peak width of approximately 100nm ensures broad spectral coverage, making these sensitizers highly effective when paired with co-initiators in complex photosensitive systems. As we delve deeper into the synthesis and application of these materials, it becomes clear that they offer a strategic advantage for companies aiming to reduce costs in electronic chemical manufacturing while simultaneously improving product quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the field of visible light photopolymerization has been constrained by the inadequate performance of available sensitizers. Traditional systems often relied on squaraine dyes or combinations of iodine salts, which, while functional, suffered from significant drawbacks such as poor solubility in common organic solvents and limited initiation efficiency. Furthermore, conventional photoinitiators like benzophenone and benzoin ethers are restricted to the UV range (200-300nm), necessitating the use of expensive and potentially hazardous UV light sources that pose safety risks and limit penetration depth in thick coatings. The reliance on these older technologies often results in incomplete curing, surface tackiness, and inferior mechanical properties in the final polymer products. Additionally, the synthesis of some alternative visible light sensitizers involves complex multi-step routes with low overall yields, driving up production costs and creating supply chain bottlenecks for manufacturers of high-purity OLED materials and other specialty chemicals.
The Novel Approach
The methodology outlined in patent CN101602822A addresses these challenges through a streamlined three-step synthetic route that prioritizes both efficiency and scalability. By employing a Vilsmeier-Haack reaction to introduce formyl groups onto the triphenylamine core, followed by an Arbuzov reaction to generate phosphonate intermediates, the process establishes a versatile platform for constructing the final photosensitizer. The key innovation lies in the final Horner-Wadsworth-Emmons condensation, which links the phosphonate and aldehyde fragments to create an extended conjugated system. This structural modification is what enables the crucial red-shift in absorption, moving the peak into the visible region above 370nm. Unlike previous methods that struggled with solubility, this approach utilizes standard organic solvents like dichloromethane and tetrahydrofuran, facilitating easier purification and handling. The result is a photosensitizer that not only meets the rigorous demands of visible light absorption but also integrates seamlessly into existing manufacturing workflows for commercial scale-up of complex polymer additives.
Mechanistic Insights into Vilsmeier-Haack Formylation and HWE Condensation
The synthesis begins with the electrophilic aromatic substitution known as the Vilsmeier-Haack reaction, where triphenylamine reacts with phosphorus oxychloride and dimethylformamide (DMF) to yield bis(4-formylphenyl)aniline. This step is critical as it installs the reactive aldehyde handles necessary for the subsequent chain extension. The mechanism involves the formation of an iminium salt intermediate, which acts as a potent electrophile attacking the electron-rich para-positions of the triphenylamine rings. Following hydrolysis, the dialdehyde product is isolated, serving as the central hub for the molecule's optical properties. The second phase involves the preparation of substituted phenyl derivatives diethyl phosphonate via the Arbuzov reaction, where a substituted benzyl bromide reacts with triethyl phosphite at elevated temperatures (130°C-160°C). This transformation converts the halide into a phosphonate ester, a stable precursor that retains the ability to act as a nucleophile in the presence of strong bases.
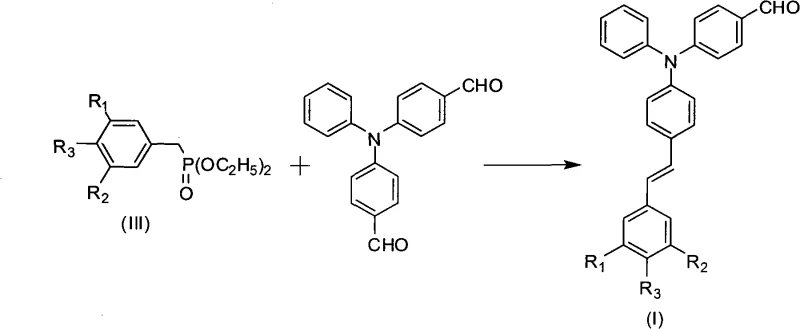
The final and most mechanistically significant step is the Horner-Wadsworth-Emmons (HWE) olefination, depicted in the reaction scheme above, which couples the phosphonate anion with the dialdehyde. Under basic conditions provided by catalysts such as sodium methylate or sodium hydride, the phosphonate is deprotonated to form a carbanion that attacks the carbonyl carbon of the aldehyde. This addition-elimination sequence results in the formation of a carbon-carbon double bond, extending the pi-conjugation system across the molecule. It is this extended conjugation that lowers the energy gap between the HOMO and LUMO orbitals, directly causing the observed bathochromic shift in absorption spectra. From an impurity control perspective, the use of mild bases and room temperature conditions in this final step minimizes side reactions such as polymerization or decomposition, ensuring a cleaner crude product that requires less aggressive purification. This mechanistic elegance translates directly to higher purity specifications, a key metric for any reliable agrochemical intermediate or electronic chemical supplier.
How to Synthesize Triphenylamine Aldehyde Photosensitizer Efficiently
The synthesis of these high-value photosensitizers follows a logical progression that balances reaction kinetics with operational simplicity, making it highly suitable for transfer from laboratory bench to pilot plant. The process leverages widely available starting materials and avoids the need for exotic catalysts or extreme pressure conditions, which simplifies equipment requirements and reduces capital expenditure. Detailed below is the standardized workflow derived from the patent examples, outlining the precise stoichiometry and conditions required to achieve optimal yields. Operators should note that strict control of moisture during the Vilsmeier-Haack step and careful temperature management during the phosphonate synthesis are critical for success.
- Perform Vilsmeier-Haack formylation on triphenylamine using phosphorus oxychloride and DMF to generate bis(4-formylphenyl)aniline.
- Synthesize phenyl derivatives diethyl phosphonate via Arbuzov reaction between substituted benzyl bromide and triethyl phosphite at elevated temperatures.
- Conduct Horner-Wadsworth-Emmons condensation between the phosphonate and the dialdehyde intermediate using a basic catalyst like sodium methylate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits that extend beyond mere technical performance. The reliance on commodity chemicals such as triphenylamine, benzyl bromides, and triethyl phosphite ensures a stable and resilient supply chain, mitigating the risks associated with sourcing specialized or scarce reagents. Furthermore, the modular nature of the synthesis allows for the easy substitution of different R-groups, enabling manufacturers to produce a family of related products from a common set of intermediates. This flexibility significantly enhances supply chain reliability, as production lines can be quickly adapted to meet fluctuating market demands for specific variants without requiring complete process revalidation. The elimination of transition metal catalysts in the final coupling step also removes the need for expensive and time-consuming heavy metal removal processes, streamlining the downstream purification workflow.
- Cost Reduction in Manufacturing: The synthetic pathway described eliminates the need for precious metal catalysts often used in cross-coupling reactions, replacing them with inexpensive base catalysts like sodium methylate. This substitution drastically reduces the raw material cost per kilogram of the final active ingredient. Additionally, the high atom economy of the condensation reactions minimizes waste generation, leading to lower disposal costs and improved overall process efficiency. By avoiding complex protection-deprotection strategies, the total number of unit operations is reduced, which directly correlates to lower labor and utility consumption per batch.
- Enhanced Supply Chain Reliability: The precursors utilized in this synthesis, particularly the substituted benzyl bromides and triphenylamine, are produced on a massive global scale for various industries, ensuring consistent availability and competitive pricing. This abundance of raw materials protects against supply disruptions that frequently plague niche chemical sectors. Moreover, the robustness of the reaction conditions means that the process is less susceptible to minor variations in feedstock quality, further stabilizing production schedules and reducing lead time for high-purity photosensitizers.
- Scalability and Environmental Compliance: The reactions operate at atmospheric pressure and moderate temperatures, removing the need for specialized high-pressure reactors and reducing energy consumption. The solvents employed, such as dichloromethane and tetrahydrofuran, are standard industrial solvents with well-established recovery and recycling protocols, facilitating compliance with stringent environmental regulations. The simplified workup procedures, involving standard extraction and chromatography, are easily adaptable to continuous flow chemistry or large-scale batch processing, supporting the commercial scale-up of complex electronic chemicals from kilograms to multi-ton quantities.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of integrating these photosensitizers into their formulations, we have compiled answers to common inquiries based on the patent data. These responses address critical aspects regarding stability, compatibility, and processing parameters. Understanding these details is essential for optimizing the performance of the final photo-cured products.
Q: What is the primary advantage of this triphenylamine-based photosensitizer over traditional initiators?
A: Unlike traditional initiators like benzophenone which absorb primarily in the UV region (200-300nm), this novel triphenylamine derivative exhibits a maximum absorption peak shifted to above 370nm in the visible region, allowing for efficient polymerization under visible light sources.
Q: Can this synthesis method be scaled for commercial production?
A: Yes, the synthesis utilizes standard organic transformations such as Vilsmeier-Haack formylation and Horner-Wadsworth-Emmons condensation, which are well-established in industrial settings. The use of readily available raw materials like triphenylamine and substituted benzyl bromides supports scalable manufacturing.
Q: What are the typical reaction conditions for the final coupling step?
A: The final condensation step is typically conducted at room temperature or under ice-water bath cooling using tetrahydrofuran as a solvent and a basic catalyst such as sodium methylate or sodium hydride, ensuring mild conditions that preserve sensitive functional groups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triphenylamine Photosensitizer Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of visible light photosensitizers in advancing modern material science. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from R&D to full-scale manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom derivatives for specific wavelength applications or bulk quantities for industrial coating lines, our infrastructure is designed to support your growth and innovation goals with unwavering consistency.
We invite you to collaborate with us to explore how this advanced technology can optimize your product portfolio. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can drive value for your organization.
