Scaling High-Performance Stilbene-Linked Coumarin Dyes for Next-Generation Two-Photon Polymerization
The rapid advancement of micro-fabrication technologies, particularly in the realm of three-dimensional nano-structuring and photonic crystal development, has created an urgent demand for highly efficient photosensitized initiation systems. Patent CN1990604A addresses this critical technological bottleneck by disclosing a novel class of stilbene-linked coumarin dyes that exhibit superior performance in two-photon polymerization processes. Unlike conventional initiators that often struggle to balance high initiation efficiency with a large two-photon absorption cross-section, this innovation provides a robust molecular architecture capable of deep penetration and high spatial resolution. The disclosed technology represents a significant leap forward for industries relying on precision lithography, offering a pathway to manufacture complex micro-elements with unprecedented fidelity and speed.
For research and development directors overseeing the formulation of advanced photoresists, the structural versatility of these dyes offers a compelling value proposition. The patent details a modular synthetic approach where the electronic properties of the dye can be finely tuned by modifying the substituents at the A1 and A2 positions, which can range from simple alkyl groups to complex heterocycles or substituted phenyls. This adaptability ensures that the material can be optimized for specific laser wavelengths, such as the commonly used 800nm Ti-Sapphire femtosecond lasers, without compromising the stability or solubility of the final formulation. Consequently, this technology enables the creation of customized photosensitizers that meet the rigorous demands of next-generation optical data storage and micro-electromechanical systems (MEMS).
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the field of two-photon polymerization has been hindered by the inherent trade-offs present in available photosensitizers. Traditional ultraviolet-visible initiators, while possessing high initiation efficiency, typically suffer from extremely small two-photon absorption cross-sections, necessitating high incident light intensities that can cause thermal damage to the substrate material. Conversely, dyes designed with large cross-sections often lack the necessary quantum efficiency to effectively initiate polymerization, leading to prolonged exposure times and reduced throughput in manufacturing environments. Furthermore, the synthetic routes for many high-performance two-photon dyes are notoriously complex, involving multiple protection and deprotection steps that drive up costs and introduce difficult-to-remove impurities, thereby limiting their commercial viability for large-scale industrial applications.
The Novel Approach
The methodology outlined in patent CN1990604A circumvents these historical challenges through a streamlined, convergent synthesis strategy that prioritizes both yield and purity. By utilizing a Wittig condensation to link a pre-functionalized coumarin intermediate with a specifically designed phosphonium salt, the process constructs the extended conjugated system in a single, high-yielding step. This approach not only simplifies the production workflow but also facilitates the easy separation of the desired trans-isomer through a mild iodine-catalyzed heat treatment. The result is a photosensitizer that successfully unifies a massive two-photon absorption cross-section with high initiation efficiency, allowing for rapid processing at lower laser powers and significantly reducing the risk of material degradation during the fabrication of delicate micro-structures.
Mechanistic Insights into the Modular Wittig Coupling Strategy
The core of this synthetic innovation lies in the strategic assembly of the conjugated backbone, which is essential for maximizing the delocalization of pi-electrons and enhancing nonlinear optical properties. The process begins with the formation of a coumarin aldehyde intermediate via a base-catalyzed condensation between a 3-acetyl-coumarin derivative and terephthalaldehyde. This intermediate serves as the electrophilic partner in the subsequent Wittig reaction, where it couples with a phosphonium ylide generated in situ from a substituted benzyl phosphonium salt. The use of mild alkaline catalysts, such as sodium hydroxide or piperidine, in biphasic solvent systems ensures that the reaction proceeds smoothly under reflux conditions, minimizing side reactions and preserving the integrity of the sensitive coumarin lactone ring.
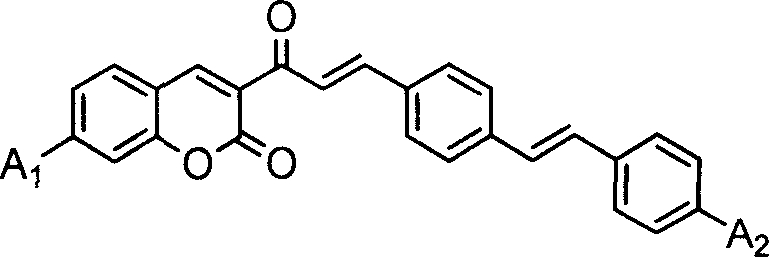
Following the initial coupling, the crude product undergoes a critical isomerization step where a catalytic amount of iodine is employed to convert any cis-isomers into the thermodynamically more stable trans-configuration. This stereochemical control is paramount, as the planar trans-structure allows for maximum conjugation length across the molecule, directly correlating to enhanced two-photon absorption capabilities. The final purification via column chromatography ensures that the resulting dye meets the stringent purity specifications required for electronic grade applications, free from residual phosphine oxides or unreacted aldehydes that could act as quenchers in the final polymerization system. This meticulous attention to mechanistic detail underscores the robustness of the process for producing high-quality optoelectronic materials.
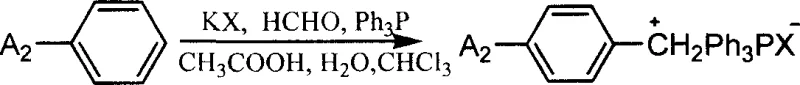
How to Synthesize Stilbene-Linked Coumarin Dyes Efficiently
The synthesis protocol described in the patent provides a clear roadmap for producing these advanced functional dyes, leveraging standard organic transformations that are well-suited for scale-up. The procedure involves three distinct stages: the preparation of the coumarin-aldehyde intermediate, the synthesis of the phosphonium salt precursor, and the final Wittig coupling followed by isomerization. Each step utilizes readily available commodity chemicals and avoids the need for exotic catalysts or extreme reaction conditions, making the entire sequence highly accessible for manufacturing facilities equipped with standard glass-lined reactors and purification units. For a detailed breakdown of the specific molar ratios, solvent choices, and temperature profiles required to replicate this high-yield process, please refer to the standardized synthesis guide below.
- Condense 3-acetyl-coumarin derivatives with terephthalaldehyde using a basic catalyst to form the aldehyde-functionalized intermediate.
- Synthesize the corresponding phosphonium salt by reacting substituted benzene derivatives with formaldehyde, potassium halide, and triphenylphosphine.
- Perform a Wittig reaction between the coumarin intermediate and the phosphonium salt, followed by iodine-catalyzed isomerization to obtain the pure trans-dye.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this synthetic route offers substantial strategic benefits driven by the simplicity and modularity of the chemistry involved. The reliance on bulk commodity chemicals such as triphenylphosphine, formaldehyde, and various substituted benzenes means that raw material sourcing is not constrained by geopolitical bottlenecks or limited supplier bases. Furthermore, the elimination of transition metal catalysts, which are often required in cross-coupling reactions like Suzuki or Heck couplings, removes the need for expensive and time-consuming heavy metal scavenging steps. This simplification of the downstream processing significantly reduces the overall cost of goods sold while simultaneously accelerating the production cycle time, allowing for faster response to market demands for specialized optoelectronic components.
- Cost Reduction in Manufacturing: The economic viability of this process is heavily anchored in its atom economy and the avoidance of precious metal catalysts. By utilizing a Wittig reaction mediated by inexpensive phosphonium salts and basic catalysts, manufacturers can achieve significant cost savings compared to palladium-catalyzed alternatives. Additionally, the high yields reported in the patent examples suggest that raw material waste is minimized, further contributing to a leaner manufacturing cost structure. The ability to recrystallize intermediates and use standard column chromatography for final purification also implies that capital expenditure on specialized filtration equipment is kept to a minimum, enhancing the overall return on investment for production lines dedicated to these high-value dyes.
- Enhanced Supply Chain Reliability: The modular nature of the synthesis, where the A1 and A2 groups can be varied without altering the core reaction conditions, provides exceptional flexibility in managing supply chain risks. If a specific substituent becomes unavailable or cost-prohibitive, the process can be rapidly adapted to utilize alternative electron-donating or accepting groups without requiring a complete re-validation of the manufacturing protocol. This adaptability ensures continuous supply continuity even in volatile raw material markets. Moreover, the stability of the intermediates allows for the potential stocking of key precursors, enabling a make-to-stock strategy that can drastically reduce lead times for customers requiring urgent delivery of custom-formulated photosensitizers for their R&D or production needs.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the use of common organic solvents such as dichloromethane, ethanol, and acetonitrile, which are well-understood in terms of handling and waste management. The reaction conditions, primarily involving reflux temperatures and atmospheric pressure, do not require high-pressure autoclaves or cryogenic cooling, simplifying the engineering requirements for plant scale-up. From an environmental standpoint, the absence of toxic heavy metals simplifies wastewater treatment protocols, aligning the manufacturing process with increasingly stringent global environmental regulations. This compliance reduces the regulatory burden on the supply chain and mitigates the risk of production shutdowns due to environmental non-compliance, ensuring a stable and sustainable supply of these critical electronic chemicals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these stilbene-linked coumarin dyes. The answers are derived directly from the experimental data and technical specifications provided in the underlying patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is crucial for integrating these materials into existing manufacturing workflows and optimizing their performance in two-photon polymerization applications.
Q: What is the primary advantage of stilbene-linked coumarin dyes in two-photon polymerization?
A: These dyes uniquely combine a large two-photon absorption cross-section with high initiation efficiency, overcoming the limitations of traditional UV-visible initiators which often suffer from low sensitivity or small cross-sections in deep-penetration applications.
Q: How is the stereochemistry of the double bonds controlled during synthesis?
A: The synthesis utilizes a Wittig condensation followed by a specific heat treatment with a catalytic amount of iodine. This post-reaction isomerization step ensures the formation of the thermodynamically stable pure trans-isomer, which is critical for optimal optical properties.
Q: Can the optical properties of these dyes be tuned for specific wavelengths?
A: Yes, the molecular structure allows for significant tunability. By varying the electron-donating or electron-accepting substituents at the A1 and A2 positions (such as alkyl, alkoxy, or amino groups), manufacturers can precisely adjust the two-photon absorption cross-section to match specific laser excitation sources.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Stilbene-Linked Coumarin Dye Supplier
As the demand for high-resolution 3D micro-fabrication continues to surge across the semiconductor and biomedical sectors, securing a reliable source of high-performance photosensitizers is more critical than ever. NINGBO INNO PHARMCHEM stands ready to support your development goals with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of stilbene-linked coumarin dye meets the exacting standards required for advanced optoelectronic applications. We understand that consistency is key in photonics, and our robust quality management systems guarantee lot-to-lot reproducibility that you can trust for your most sensitive projects.
We invite you to collaborate with our technical team to explore how this patented technology can be integrated into your specific product lines. By contacting our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements, as well as obtain specific COA data and route feasibility assessments for your target derivatives. Whether you require off-the-shelf samples for initial testing or a fully validated custom synthesis campaign, we are committed to delivering the chemical solutions that will power your next generation of photonic innovations.
