Advanced Synthesis of Cyclic Acetal Benzophenone Photoinitiators for Commercial Scale
The landscape of UV curing technology is undergoing a significant transformation driven by the urgent demand for safer, more efficient photoinitiating systems. As detailed in the groundbreaking patent CN101812141B, a novel class of diphenyl ketone photoinitiators containing cyclic acetals has emerged as a superior alternative to traditional formulations. This technology addresses critical limitations in the light-curing industry, particularly the reliance on toxic amine co-initiators that have long plagued manufacturers with issues of yellowing and regulatory compliance. By integrating a cyclic acetal base directly into the benzophenone structure, this innovation enables the compound to function independently as a photoinitiator. This structural modification not only enhances the photochemical efficiency but also drastically simplifies the formulation process for end-users. For R&D directors and procurement specialists seeking reliable specialty chemical suppliers, understanding the mechanistic advantages of this patent is crucial for next-generation product development. The ability to achieve high double bond conversion rates without auxiliary amines represents a paradigm shift in how we approach polymer network formation in industrial coatings and dental materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the photocuring industry has relied heavily on Type II photoinitiators, specifically benzophenone derivatives, which necessitate the presence of a hydrogen donor, typically an amine co-initiator, to generate active free radicals. While effective, this binary system introduces substantial complications regarding toxicity, odor, and long-term stability. Amines are well-documented for their potential carcinogenicity and tendency to cause significant yellowing in cured films upon exposure to heat or light over time. This yellowing effect severely restricts the application of such materials in aesthetic-critical fields like clear wood coatings, white pigmented paints, and especially food contact materials. Furthermore, the volatility of low molecular weight amines can lead to migration issues, compromising the integrity of the final polymer matrix. From a supply chain perspective, managing two distinct reactive components increases inventory complexity and the risk of formulation errors. The environmental burden of disposing of amine-containing waste streams also adds to the operational costs, making conventional benzophenone/amine systems increasingly unsustainable in the face of tightening global environmental regulations.
The Novel Approach
The innovative methodology disclosed in patent CN101812141B circumvents these drawbacks by chemically modifying the benzophenone core with a cyclic acetal group. This structural integration creates a single-component photoinitiator that possesses intrinsic hydrogen abstraction capabilities or modified electronic states sufficient for initiation without external amines. The synthesis involves a straightforward condensation reaction between 4-hydroxybenzophenone derivatives and formaldehyde in an acidic aqueous environment. 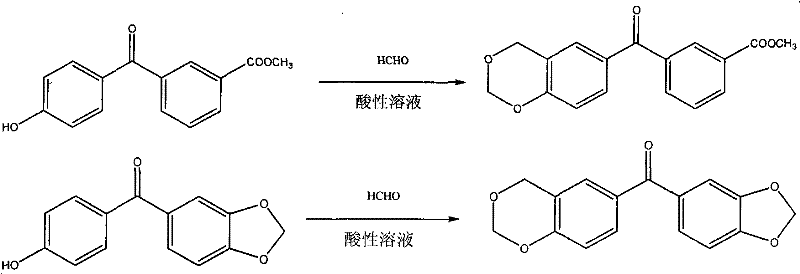 . As illustrated in the reaction scheme, the process transforms readily available starting materials into high-value functional molecules through a robust and scalable pathway. This approach eliminates the need for toxic co-initiators, thereby inherently solving the yellowing and toxicity problems at the molecular level. For procurement managers, this translates to a simplified supply chain where a single, stable ingredient replaces a complex multi-component system, significantly reducing the risk of quality variance and enhancing the overall reliability of the manufacturing process.
. As illustrated in the reaction scheme, the process transforms readily available starting materials into high-value functional molecules through a robust and scalable pathway. This approach eliminates the need for toxic co-initiators, thereby inherently solving the yellowing and toxicity problems at the molecular level. For procurement managers, this translates to a simplified supply chain where a single, stable ingredient replaces a complex multi-component system, significantly reducing the risk of quality variance and enhancing the overall reliability of the manufacturing process.
Mechanistic Insights into Acid-Catalyzed Cyclic Acetal Formation
The core chemical transformation driving this technology is an acid-catalyzed electrophilic aromatic substitution followed by cyclization, effectively forming a six-membered cyclic acetal ring fused to the aromatic system. In the presence of strong mineral acids or organic sulfonic acids, formaldehyde is activated to form an electrophilic species that attacks the electron-rich positions ortho to the hydroxyl group on the benzophenone ring. This reaction proceeds efficiently in an aqueous medium, which is a notable departure from traditional organic solvent-based syntheses. The cyclic acetal moiety introduced onto the benzophenone skeleton alters the electronic distribution of the molecule, potentially lowering the energy gap for n-π* transitions or stabilizing the radical intermediates formed during photolysis. 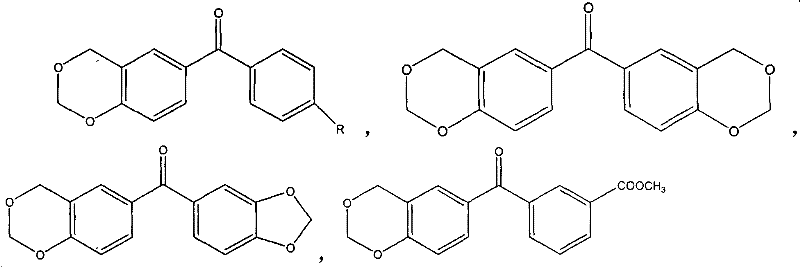 . As shown in the general structure, the versatility of this scaffold allows for various substituents (R = H, alkyl, halogen, ester) to fine-tune solubility and reactivity. This tunability is essential for R&D teams aiming to optimize compatibility with different oligomer resins, such as epoxy acrylates or polyester acrylates. The mechanism ensures that the resulting photoinitiator maintains high absorption in the UV region while possessing the necessary redox potential to initiate polymerization autonomously.
. As shown in the general structure, the versatility of this scaffold allows for various substituents (R = H, alkyl, halogen, ester) to fine-tune solubility and reactivity. This tunability is essential for R&D teams aiming to optimize compatibility with different oligomer resins, such as epoxy acrylates or polyester acrylates. The mechanism ensures that the resulting photoinitiator maintains high absorption in the UV region while possessing the necessary redox potential to initiate polymerization autonomously.
Furthermore, the impurity profile of this synthesis is exceptionally clean due to the use of water as the primary solvent medium. Side reactions are minimized because the acidic conditions selectively promote the formation of the thermodynamically stable cyclic acetal ring. Any unreacted formaldehyde can be easily removed during the workup phase, and the product is typically isolated via simple filtration and recrystallization from ethanol-water mixtures. This high selectivity is critical for achieving the stringent purity specifications required in electronic and medical applications. The absence of heavy metal catalysts or exotic reagents means that the final product is free from trace metal contaminants that could otherwise catalyze unwanted degradation pathways in the cured polymer. For quality assurance teams, this inherent process robustness simplifies analytical testing and ensures batch-to-batch consistency, which is a key metric for qualifying new materials in regulated industries.
How to Synthesize Cyclic Acetal Benzophenone Efficiently
The practical implementation of this synthesis route is designed for ease of operation and scalability, making it highly attractive for commercial production. The process begins with the suspension of the hydroxybenzophenone precursor in a formaldehyde solution, followed by the controlled addition of the acid catalyst. Temperature control is vital, with the reaction typically proceeding between 10-80°C depending on the specific substituent effects. The detailed standardized synthesis steps, including precise stoichiometric ratios and workup procedures, are outlined below to assist technical teams in replicating these results.
- Prepare the reaction mixture by combining 1 molar part of 4-hydroxybenzophenone derivative with 5 molar parts of 37% formaldehyde solution.
- Slowly add an acidic aqueous solution (50-100 times the molar volume) containing hydrochloric acid or p-toluenesulfonic acid while maintaining temperature control.
- Heat the mixture to 10-80°C and stir for 1-10 hours, then filter, recrystallize from ethanol/water, and vacuum dry to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this novel photoinitiator technology offers profound strategic advantages for procurement and supply chain leadership, primarily driven by process simplification and raw material accessibility. The elimination of amine co-initiators removes an entire category of hazardous chemicals from the procurement list, thereby reducing regulatory overhead and storage costs associated with toxic substances. Since the synthesis relies on commodity chemicals like formaldehyde and substituted benzophenones, the supply chain is inherently more resilient against market fluctuations compared to specialized amine suppliers. The aqueous nature of the reaction medium also presents significant opportunities for cost reduction in manufacturing, as it avoids the expense of purchasing, recovering, and disposing of large volumes of organic solvents. This shift towards greener chemistry aligns perfectly with corporate sustainability goals and reduces the environmental footprint of the production facility. Moreover, the high yields reported in the patent examples indicate a highly atom-economical process, maximizing the output per unit of raw material input and directly contributing to improved gross margins.
- Cost Reduction in Manufacturing: The transition to this single-component system fundamentally alters the cost structure of UV curable formulations. By removing the requirement for expensive and often volatile amine synergists, formulators can achieve significant cost savings on a per-kilogram basis. The synthesis itself is economically favorable due to the use of water as a solvent, which eliminates the capital expenditure and operational costs associated with solvent recovery distillation columns. Additionally, the mild reaction conditions (10-80°C) reduce energy consumption compared to high-temperature processes, further lowering the utility costs of production. The simplicity of the workup, involving basic filtration and recrystallization, minimizes labor hours and equipment downtime, allowing for faster turnaround times between batches. These cumulative efficiencies result in a substantially lower cost of goods sold (COGS), providing a competitive pricing advantage in the marketplace without sacrificing performance.
- Enhanced Supply Chain Reliability: Sourcing stability is a critical concern for supply chain heads, and this technology leverages widely available feedstocks to mitigate supply risks. 4-Hydroxybenzophenone and formaldehyde are produced on a massive global scale for diverse industries, ensuring that supply disruptions are unlikely to impact production continuity. The robustness of the synthetic route means that manufacturing can be easily scaled up or shifted between different facilities without requiring specialized equipment or highly trained personnel. This flexibility allows for a more agile supply chain capable of responding rapidly to spikes in demand from the coatings or electronics sectors. Furthermore, the stability of the final cyclic acetal product ensures a long shelf life, reducing the risk of inventory write-offs due to degradation. This reliability fosters stronger partnerships with downstream customers who depend on consistent, uninterrupted material flows for their own just-in-time manufacturing operations.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces unforeseen challenges, but the aqueous acid-catalyzed nature of this reaction is inherently scalable. The exotherm is manageable, and the use of water provides excellent heat transfer properties, reducing the risk of thermal runaway in large reactors. From an environmental compliance standpoint, the process generates minimal hazardous waste; the acidic aqueous filtrate can often be neutralized and treated using standard wastewater protocols, avoiding the complexities of hazardous organic waste disposal. This alignment with green chemistry principles facilitates easier permitting and regulatory approval in jurisdictions with strict environmental laws. The ability to produce high-purity material with a low environmental impact positions the manufacturer as a preferred partner for eco-conscious brands. Consequently, this technology not only meets current regulatory standards but is future-proofed against anticipated tightening of restrictions on volatile organic compounds (VOCs) and toxic additives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of cyclic acetal benzophenone photoinitiators. These insights are derived directly from the experimental data and beneficial effects described in the patent literature, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this technology into existing product lines.
Q: Why replace traditional benzophenone/amine systems with cyclic acetal derivatives?
A: Traditional systems require toxic amine co-initiators which cause yellowing and carcinogenicity concerns. The cyclic acetal derivative functions as a standalone initiator, eliminating amine toxicity and improving color stability in cured films.
Q: What are the typical reaction conditions for this synthesis?
A: The process utilizes mild temperatures between 10-80°C and reaction times of 1-10 hours in an acidic aqueous medium, offering significant energy savings compared to high-temperature organic solvent processes.
Q: Is this photoinitiator suitable for food packaging applications?
A: Yes, by eliminating toxic amine co-initiators and reducing yellowing, this technology addresses key regulatory and safety hurdles associated with food and drug packaging materials.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyclic Acetal Benzophenone Photoinitiator Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the technology described in patent CN101812141B for the UV curing industry. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition to this advanced photoinitiator is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific requirements of acid-catalyzed aqueous reactions, maintaining stringent purity specifications through our rigorous QC labs. We understand that consistency is paramount for high-performance coatings and electronic materials, and our quality management systems are designed to deliver batch-after-batch reliability. By partnering with us, you gain access to a supply chain that prioritizes both technical excellence and operational agility, allowing you to focus on innovation while we manage the complexities of manufacturing.
We invite you to explore how this amine-free photoinitiator technology can enhance your product portfolio and reduce your environmental footprint. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific formulation needs, demonstrating the tangible economic benefits of switching to our cyclic acetal derivatives. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your projects. Together, we can drive the next generation of sustainable, high-performance UV curing solutions, ensuring your market leadership in an increasingly competitive and regulated landscape.
