Advanced Manufacturing of High-Purity Levo-Terbutaline Intermediates via Chiral Sulfinamide Technology
Introduction to Patent CN113264839A: A Breakthrough in Chiral Respiratory Drug Synthesis
The pharmaceutical industry is currently witnessing a paradigm shift towards single-enantiomer drugs, driven by the imperative to maximize therapeutic efficacy while minimizing toxicological risks associated with racemic mixtures. In this context, the recent disclosure of patent CN113264839A represents a significant technological advancement in the manufacturing of levo-terbutaline, a potent beta-2 adrenoceptor agonist widely used for treating bronchial asthma and chronic obstructive pulmonary diseases. Unlike traditional approaches that rely on the resolution of racemates—often discarding half of the synthesized material—this novel methodology employs S-(-)-tert-butyl sulfinamide as a robust chiral prosthetic group to direct the stereochemistry of the synthesis from the outset. The patent outlines a streamlined, four-step synthetic route that not only simplifies operational complexity but also achieves an extraordinary enantiomeric excess (ee) of up to 99.9%. For global procurement and R&D teams, this development signals a new benchmark for reliability and quality in the supply chain of critical respiratory API intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of levo-terbutaline has been plagued by inherent inefficiencies and economic drawbacks associated with legacy technologies. The most prevalent method, optical resolution using tartaric acid or its derivatives, is fundamentally flawed because it is a destructive process that theoretically caps the maximum yield at 50%, necessitating the disposal or recycling of the unwanted enantiomer. Furthermore, achieving high optical purity via resolution often requires multiple, energy-intensive crystallization cycles, which complicates the workflow and inflates production costs. Alternative strategies, such as enzymatic reduction or asymmetric hydrogenation using chiral boron or ruthenium catalysts, introduce their own set of challenges; enzymatic processes often suffer from low catalytic efficiency and require extensive screening, while transition metal-catalyzed routes involve expensive noble metals that pose contamination risks and require stringent purification protocols to meet regulatory limits for residual heavy metals in pharmaceutical products.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the methodology described in patent CN113264839A leverages the power of chiral auxiliary-controlled asymmetric synthesis to overcome these bottlenecks. By utilizing S-(-)-tert-butyl sulfinamide, a commercially abundant and cost-effective chiral source, the process establishes the critical stereocenter early in the synthesis through a highly diastereoselective reduction step. This approach effectively bypasses the 50% yield ceiling of resolution methods and eliminates the dependency on scarce noble metal catalysts. The resulting pathway is characterized by mild reaction conditions, typically ranging from 0°C to 60°C, and the use of standard reducing agents like sodium borohydride, which are far safer and easier to handle on a multi-ton scale than toxic borane reagents. This strategic pivot not only enhances the overall atom economy but also ensures a consistent supply of high-purity intermediates essential for downstream drug formulation.
Mechanistic Insights into Sulfinamide-Controlled Asymmetric Reduction
The cornerstone of this innovative synthesis lies in the precise stereochemical control exerted by the tert-butylsulfinyl group during the reduction of the prochiral ketone intermediate. When the sulfinamide-substituted acetophenone derivative (Compound 5) is subjected to reduction, the bulky tert-butyl group attached to the sulfur atom creates a rigid chiral environment that sterically hinders one face of the carbonyl group. This steric bias forces the incoming hydride nucleophile, delivered by sodium borohydride in the presence of a phase-transfer catalyst like tetrabutylammonium bromide, to attack exclusively from the less hindered face. The result is the formation of a single diastereomer (Compound 6) with exceptional fidelity, where the chirality of the sulfur auxiliary dictates the configuration of the newly formed secondary alcohol center. This mechanism is superior to substrate-controlled reductions because the auxiliary provides a much stronger directing effect, ensuring that minor impurities or diastereomers can be easily removed via simple crystallization before the auxiliary is cleaved.
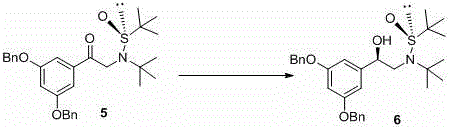
Furthermore, the robustness of the sulfinamide linkage allows for orthogonal deprotection strategies that preserve the integrity of the sensitive phenolic groups protected as benzyl ethers. The subsequent removal of the sulfinyl group under acidic conditions proceeds cleanly without racemization of the adjacent chiral center, a common pitfall in other asymmetric syntheses. This mechanistic reliability translates directly into process stability; because the stereochemistry is locked in by a covalent bond rather than a transient catalyst-substrate interaction, the reaction is less susceptible to fluctuations in temperature or mixing efficiency. For R&D directors, this means the impurity profile of the final API is predictable and manageable, significantly reducing the risk of batch failures during technology transfer and scale-up activities.
How to Synthesize Levo-Terbutaline Efficiently
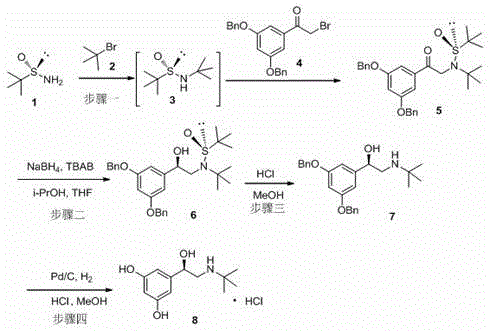
The synthesis of levo-terbutaline via this chiral auxiliary route is designed for operational simplicity and high throughput, making it an ideal candidate for contract development and manufacturing organizations (CDMOs) looking to optimize their production lines. The process begins with the N-alkylation of S-(-)-tert-butyl sulfinamide followed by coupling with a dibenzyloxy-bromoacetophenone to generate the key ketone precursor. This is followed by the critical asymmetric reduction step, which sets the stereochemistry, and concludes with sequential deprotection and hydrogenolysis steps to reveal the final active pharmaceutical ingredient. The detailed standardized operating procedures, including specific solvent ratios, temperature gradients, and workup protocols required to replicate the high yields and purity reported in the patent, are outlined in the technical guide below.
- React S-(-)-tert-butyl sulfinamide with tert-butyl bromide and 3,5-dibenzyloxy bromoacetophenone to form the ketone intermediate (Compound 5).
- Perform asymmetric reduction of Compound 5 using sodium borohydride and a quaternary ammonium salt catalyst to obtain the chiral alcohol (Compound 6).
- Remove the tert-butylsulfinyl protecting group from Compound 6 under acidic conditions to yield the amine intermediate (Compound 7).
- Conduct catalytic hydrogenolysis of Compound 7 using Pd/C and hydrochloric acid to remove benzyl groups and finalize levo-terbutaline hydrochloride.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this sulfinamide-based synthesis route offers compelling economic and logistical advantages over traditional manufacturing methods. The primary driver of value is the drastic reduction in raw material costs achieved by substituting expensive chiral transition metal catalysts with inexpensive, commodity-grade chemicals. Additionally, the elimination of the resolution step effectively doubles the theoretical yield per batch compared to racemic splitting, which significantly lowers the cost of goods sold (COGS) and reduces the environmental footprint associated with waste disposal. The use of non-toxic reagents and mild conditions also simplifies regulatory compliance and safety protocols, further reducing overhead costs related to hazardous waste management and specialized equipment maintenance.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the replacement of precious metal catalysts, such as ruthenium or rhodium complexes, with affordable sodium borohydride and quaternary ammonium salts. This substitution removes the need for costly metal scavenging resins and extensive analytical testing for heavy metal residues, which are mandatory for API release. Furthermore, the high diastereoselectivity of the reduction step minimizes the formation of difficult-to-separate impurities, thereby reducing the consumption of solvents and energy during purification. The overall effect is a leaner, more cost-efficient manufacturing process that delivers substantial savings without compromising on the quality or potency of the final pharmaceutical product.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the reliance on readily available starting materials like S-(-)-tert-butyl sulfinamide and 3,5-dibenzyloxy bromoacetophenone mitigates the risk of raw material shortages that often plague specialized catalyst supply chains. Since the reagents involved are stable and have long shelf lives, manufacturers can maintain strategic stockpiles to buffer against market volatility. The robustness of the reaction conditions also means that the process can be easily transferred between different manufacturing sites or scaled up from pilot plants to commercial production without significant re-optimization, ensuring a continuous and reliable flow of high-purity intermediates to downstream formulation partners.
- Scalability and Environmental Compliance: The environmental profile of this synthesis aligns perfectly with modern green chemistry initiatives, as it avoids the use of toxic borane gases and generates minimal hazardous waste. The reactions are conducted in common organic solvents like acetonitrile, THF, and methanol, which are easily recovered and recycled through standard distillation processes. This ease of solvent recovery, combined with the high atom economy of the asymmetric step, reduces the overall E-factor of the process. For large-scale operations, this translates to lower waste treatment costs and a smaller carbon footprint, making the technology attractive for companies aiming to meet stringent sustainability targets and regulatory environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral auxiliary technology in pharmaceutical manufacturing. These insights are derived directly from the experimental data and beneficial effects reported in patent CN113264839A, providing a clear understanding of the process capabilities and limitations for potential adopters.
Q: What is the enantiomeric excess (ee) achieved in this novel synthesis route?
A: The patented method utilizing S-(-)-tert-butyl sulfinamide as a chiral auxiliary achieves an exceptionally high enantiomeric excess of up to 99.9%, significantly surpassing traditional resolution methods.
Q: How does this method improve cost efficiency compared to noble metal catalysis?
A: By replacing expensive chiral ruthenium or rhodium catalysts with commercially available and inexpensive tert-butyl sulfinamide and sodium borohydride, the process drastically reduces raw material costs and eliminates the need for complex metal removal steps.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the process utilizes mild reaction conditions (0-60°C), common solvents like acetonitrile and methanol, and avoids toxic borane reagents, making it highly scalable and compliant with green chemistry principles for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Levo-Terbutaline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to stay competitive in the global pharmaceutical market. Our team of expert chemists has thoroughly analyzed the sulfinamide-based route for levo-terbutaline and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are fully equipped to implement this high-efficiency process in our state-of-the-art facilities, ensuring that every batch meets stringent purity specifications and rigorous QC labs standards. Our commitment to technical excellence allows us to deliver complex chiral intermediates with the consistency and reliability that top-tier pharmaceutical companies demand for their critical drug pipelines.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to discuss your needs,索取 specific COA data, and review our comprehensive route feasibility assessments to ensure your supply chain is optimized for both performance and profitability.
