Advanced Phenazine Derivative Photoinitiators for High-Efficiency UV-LED Curing Applications
The rapid transition of the industrial curing sector from traditional mercury-vapor lamps to energy-efficient Ultraviolet Light Emitting Diodes (UV-LEDs) has exposed critical limitations in legacy photoinitiator chemistries. As detailed in patent CN112707870A, the industry is witnessing a paradigm shift towards specialized phenazine derivatives designed specifically to bridge the spectral gap between modern light sources and photo-active compounds. Traditional Type I photoinitiators, such as the widely used Photoinitiator 907 (2-methyl-1-(4-methylthiophenyl)-2-morpholino-1-propanone), suffer from a fundamental mismatch; their peak absorption lies primarily in the 231nm and 307nm regions, whereas commercial UV-LEDs predominantly emit at 385nm, 395nm, or 405nm. This spectral disconnect results in suboptimal light absorption efficiency, necessitating higher initiator loadings or longer exposure times, which negatively impacts throughput and final material properties. The disclosed technology introduces a novel class of phenazine derivative photoinitiators that integrate an electron-rich benzophenazine group with a 2-methyl-2-morpholinyl-acetone moiety. This structural innovation not only induces a desirable red shift in the absorption spectrum to align perfectly with UV-LED outputs but also leverages intramolecular interactions to drastically reduce migration, addressing a persistent pain point in high-performance coating and ink formulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
For decades, the reliance on Photoinitiator 907 has been a compromise for formulators working with emerging LED curing technologies. The primary deficiency lies in its photo-physical properties; the molecule simply does not absorb the longer wavelengths emitted by solid-state LED arrays effectively. Consequently, manufacturers often face the dilemma of either accepting slow cure speeds, which bottleneck production lines, or increasing the concentration of the photoinitiator, which can lead to issues with yellowing, odor, and extractables in the final product. Furthermore, the relatively small molecular size and lack of steric bulk in conventional initiators allow for significant diffusion within the cured polymer network. This mobility poses severe regulatory and performance risks, particularly in applications like food packaging or electronics where non-migratory behavior is mandatory to prevent contamination or degradation of sensitive components. The inability of traditional initiators to simultaneously offer high reactivity under LED irradiation and low migration has created a pressing demand for next-generation solutions that do not force a trade-off between speed and safety.
The Novel Approach
The patented synthesis route overcomes these historical barriers through a sophisticated molecular architecture that merges the robust photocleavage capability of alpha-amino ketones with the extended conjugation of a phenazine system. By attaching the 2-methyl-2-morpholinyl-acetone group directly to the benzene ring of a phenazine derivative, the inventors have created a system where the electron-rich nature of the phenazine core enhances the overall molar absorptivity in the near-UV region. This structural modification ensures that the photoinitiator captures a maximum amount of photon energy from UV-LED sources, translating directly into faster radical generation and superior curing depth. Moreover, the bulky, planar phenazine structure acts as a physical anchor within the polymer matrix, significantly hindering molecular movement post-cure. This dual functionality—enhanced spectral matching and reduced mobility—represents a substantial technological leap, enabling formulators to achieve complete surface and through-cure with lower additive levels while maintaining strict compliance with migration standards.
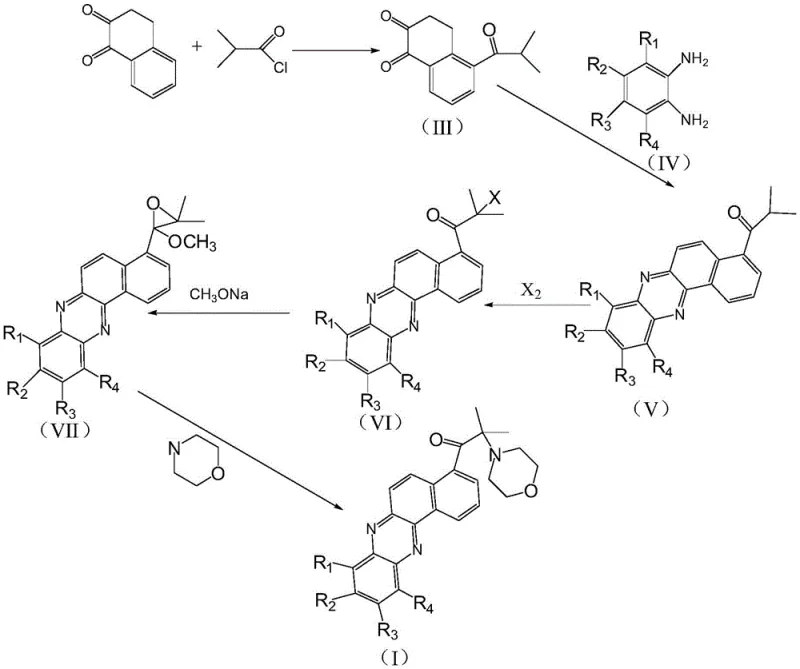
Mechanistic Insights into Friedel-Crafts Acylation and Phenazine Condensation
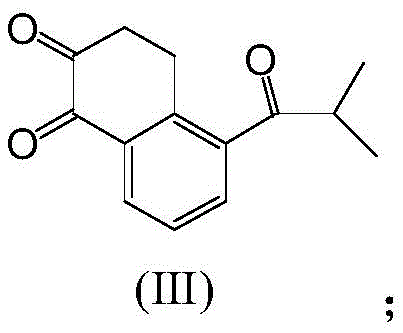
The synthesis of these advanced photoinitiators relies on a robust five-step sequence that begins with the functionalization of 1,2-naphthoquinone. The initial step involves a classic Friedel-Crafts acylation, where 1,2-naphthoquinone reacts with isobutyryl chloride in the presence of a Lewis acid catalyst, typically aluminum trichloride. This reaction is critical as it installs the requisite isobutyryl side chain onto the naphthalene framework, setting the stage for the subsequent construction of the photoinitiator's active center. The reaction conditions are tightly controlled, typically maintained between 5°C and 15°C, to prevent over-acylation or degradation of the quinone system. Following hydrolysis and workup, the resulting ketone intermediate serves as the electrophilic partner for the crucial ring-closing step. The precision required in this stage underscores the importance of high-purity starting materials to minimize side reactions that could complicate downstream purification.
Subsequent steps involve the condensation of this ketone intermediate with various substituted o-phenylenediamines to form the phenazine core, followed by halogenation and epoxidation. A key mechanistic advantage of this route is the final morpholine substitution step, which is facilitated by specific auxiliary agents. Unlike traditional methods that might rely on heterogeneous catalysts requiring difficult filtration, this process utilizes aqueous solutions of strong base-weak acid salts or strong acid-weak base salts. These auxiliaries promote the ring-opening of the epoxide intermediate by morpholine, enhancing reaction kinetics without introducing insoluble particulates that could haze the final product. This homogeneous catalytic approach not only improves yield, with examples demonstrating efficiencies upwards of 88%, but also ensures the optical clarity of the photoinitiator, a critical parameter for transparent coatings and inks. The ability to fine-tune the substituents (R1-R4) on the phenazine ring further allows for the modulation of solubility and compatibility with different resin systems, offering formulators unprecedented flexibility.
How to Synthesize Phenazine Derivative Photoinitiator Efficiently
The preparation of these high-performance photoinitiators follows a logical progression of organic transformations amenable to standard reactor setups. The process begins with the acylation of the naphthoquinone scaffold, proceeds through condensation to build the heterocyclic core, and concludes with functional group manipulation to install the morpholine moiety. Each step has been optimized to balance reaction rate with selectivity, ensuring that the final product meets stringent purity specifications required for electronic and packaging applications. The use of common solvents like dichloroethane, methanol, and acetic acid throughout the sequence simplifies solvent recovery and waste management, contributing to a more sustainable manufacturing profile. For detailed operational parameters, including specific temperature ramps, addition rates, and workup procedures, please refer to the standardized protocol below.
- Perform Friedel-Crafts acylation of 1,2-naphthoquinone with isobutyryl chloride using aluminum trichloride catalyst to form the ketone intermediate.
- Condense the ketone intermediate with substituted o-phenylenediamine in acidic solvent to construct the phenazine core structure.
- Execute halogenation followed by epoxidation with sodium methoxide, and finalize with morpholine substitution to yield the target photoinitiator.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this phenazine-based photoinitiator technology offers compelling advantages that extend beyond mere performance metrics. The synthesis pathway is constructed from readily available commodity chemicals, mitigating the supply chain risks associated with exotic or single-source reagents. This accessibility ensures a stable supply continuity, which is paramount for large-scale coating and ink manufacturers who cannot afford production stoppages due to raw material shortages. Furthermore, the robustness of the chemical steps—operating at moderate temperatures and pressures—reduces the energy intensity of the manufacturing process compared to high-pressure hydrogenations or cryogenic reactions often found in specialty chemical synthesis. This operational efficiency translates directly into a more favorable cost structure, allowing suppliers to offer competitive pricing without compromising on quality or purity.
- Cost Reduction in Manufacturing: The streamlined nature of the five-step synthesis eliminates the need for complex chromatographic purifications or expensive transition metal catalysts that often plague fine chemical production. By utilizing simple acid-base workups and recrystallization techniques, the process minimizes waste generation and reduces the consumption of high-cost consumables. Additionally, the high yields reported in the patent examples indicate a highly atom-economical process, meaning less raw material is lost to byproducts. This efficiency drives down the cost of goods sold (COGS), enabling significant cost reduction in UV-curing material manufacturing for end-users who are sensitive to resin and additive pricing.
- Enhanced Supply Chain Reliability: The reliance on bulk chemicals such as 1,2-naphthoquinone, isobutyryl chloride, and morpholine ensures that the supply chain is resilient against market volatility. These precursors are produced by multiple global vendors, preventing bottlenecks that can occur with proprietary intermediates. The synthetic route's tolerance for various substituted diamines also provides flexibility; if one specific diamine variant faces supply constraints, the chemistry can often be adapted to use alternative analogues without redesigning the entire process. This adaptability is a crucial risk mitigation strategy for procurement managers tasked with securing long-term supply agreements for critical production inputs.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from kilogram-scale laboratory batches to multi-ton commercial production without requiring specialized equipment. The avoidance of heavy metal catalysts simplifies the environmental compliance landscape, as there is no need for rigorous testing and removal of trace metals like palladium or platinum from the final product. This feature is particularly valuable for applications in food contact materials and toys, where regulatory limits on metal residues are extremely strict. The simplified waste stream, primarily consisting of aqueous salt solutions and recoverable organic solvents, facilitates easier treatment and disposal, aligning with increasingly stringent global environmental regulations.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this new photoinitiator class is essential for making informed purchasing decisions. The following questions address common inquiries regarding performance, compatibility, and regulatory status, derived directly from the patent's experimental data and technical disclosures. These insights are intended to clarify how this technology integrates into existing formulation workflows and what benefits it brings to the final cured product.
Q: How does this phenazine derivative improve upon Photoinitiator 907?
A: The novel phenazine derivative exhibits a significant red shift in absorption wavelength, providing superior matching with UV-LED emission spectra (385-405nm) compared to the poor matching of Photoinitiator 907, resulting in higher curing efficiency.
Q: What are the key advantages regarding migration in cured films?
A: The molecular design incorporates a bulky benzophenazine group that interacts with the morpholine moiety, significantly reducing molecular mobility within the polymer matrix and minimizing migration risks in food packaging or electronic applications.
Q: Is the synthesis scalable for industrial production?
A: Yes, the process utilizes standard organic unit operations such as acylation, condensation, and nucleophilic substitution, avoiding exotic reagents, which facilitates straightforward scale-up from laboratory to multi-ton commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phenazine Photoinitiator Supplier
As the global demand for UV-LED curable materials continues to surge, partnering with a manufacturer that possesses deep technical expertise in photochemistry is vital. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of phenazine photoinitiator delivers consistent performance in your formulations. We understand that the transition to new initiators requires validation, and our team is prepared to support you with samples and technical data to accelerate your development cycles.
We invite you to engage with our technical procurement team to discuss how this innovative photoinitiator can optimize your specific application. Whether you require a Customized Cost-Saving Analysis to evaluate the economic impact of switching from traditional initiators or need specific COA data and route feasibility assessments for new derivatives, we are ready to provide the support you need. Contact us today to secure a reliable supply of high-performance photoinitiators that will drive your UV-curing operations into the future.
