Advanced Solvent-Free Synthesis of N-Phenyl Morpholine Compounds for Commercial Scale
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to synthesize complex heterocyclic structures that serve as critical building blocks for next-generation therapeutics. Patent CN106928162B introduces a groundbreaking synthesis method for substituted N-phenyl morpholine compounds, addressing long-standing inefficiencies in the production of these vital pharmaceutical intermediates. This innovation represents a significant leap forward in process chemistry, moving away from the reliance on precious metal catalysts and complex purification protocols that have historically plagued this sector. By leveraging a direct heating and ring-closing strategy under alkaline conditions, this technology offers a pathway to high-purity N-phenylmorpholine derivatives with exceptional economic and operational advantages. For R&D directors and procurement managers alike, understanding the nuances of this patent is essential for optimizing supply chains and reducing the overall cost of goods sold in drug manufacturing. The ability to produce sterically hindered morpholine derivatives efficiently opens new doors for the synthesis of antituberculosis drugs and antibacterial agents like linezolid, ensuring a more reliable supply of these critical medicines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-phenylmorpholine compounds with steric hindrance has been a formidable challenge for process chemists, primarily due to the reliance on the Buchwald-Hartwig reaction. This traditional approach necessitates the use of high-price palladium catalysts, which not only inflate the raw material costs but also introduce significant complications regarding residual metal removal in the final product. Furthermore, conventional methods often require the use of solvents like DMF, which are difficult to recover and pose environmental and safety hazards in a large-scale manufacturing setting. The operational complexity is further exacerbated by the need for column chromatography separation and purification, a process that is notoriously difficult to scale and results in substantial product loss. Most critically, the yields associated with these prior art methods are dismally low, often ranging from merely 2% to 10%, rendering them economically unviable for commercial production. Even alternative approaches using aniline and 2-chloroethyl ether with inorganic bases or microwave assistance have struggled to achieve purification yields above 15-30%, failing to meet the demands of modern industrial efficiency.
The Novel Approach
In stark contrast to these inefficient legacy processes, the method disclosed in patent CN106928162B employs a simple yet highly effective synthesis route that eliminates the need for expensive catalysts and problematic solvents. This novel approach utilizes low-price aniline and an excess of 2-chloroethyl ether, which are directly heated and ring-closed under the action of an organic alkali to obtain the desired product. The reaction is carried out in the absence of a solvent, which drastically simplifies the work-up procedure and removes the burden of solvent recovery and disposal. This solvent-free condition not only reduces the environmental footprint but also significantly lowers the operational costs associated with energy consumption and waste management. By optimizing the reaction conditions, this method successfully produces N-phenylmorpholine with steric hindrance from sterically hindered aniline, achieving yields that are substantially higher than any previously reported method. The use of organic alkalis such as triethylamine or diisopropylethylamine further enhances the reaction efficiency, providing a clean and scalable solution for the manufacturing of these complex intermediates.
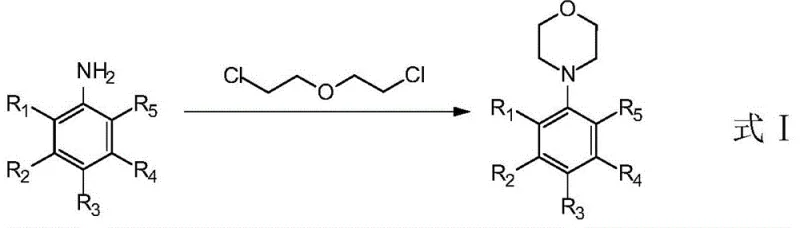
Mechanistic Insights into Solvent-Free Alkaline Cyclization
The core of this technological breakthrough lies in the mechanistic efficiency of the solvent-free alkaline cyclization process. The reaction proceeds through a nucleophilic substitution mechanism where the nitrogen atom of the substituted aniline attacks the electrophilic carbon of the 2-chloroethyl ether. Under the influence of the organic base, the proton on the amine is abstracted, increasing its nucleophilicity and facilitating the initial alkylation step. Subsequently, the intramolecular cyclization occurs, closing the morpholine ring and releasing a chloride ion. The absence of solvent plays a crucial role in this mechanism by increasing the effective concentration of the reactants, thereby driving the equilibrium towards the product side and accelerating the reaction rate. High temperatures, typically ranging from 130°C to 180°C, provide the necessary activation energy to overcome the steric barriers presented by ortho-substituted anilines, ensuring that even bulky substrates can react efficiently. This mechanistic pathway avoids the formation of side products often associated with transition metal catalysis, resulting in a cleaner reaction profile and simplified downstream processing.
Impurity control is another critical aspect where this method excels, particularly for R&D directors focused on purity and impurity profiles. The use of excess 2-chloroethyl ether serves a dual purpose: it acts as both a reactant and a pseudo-solvent that can be easily recovered by distillation after the reaction is complete. This recoverability ensures that unreacted starting materials do not contaminate the final product, and the excess reagent can be recycled, contributing to the overall atom economy of the process. Furthermore, the selection of specific organic bases like triethylamine or pyridine helps to neutralize the hydrochloric acid byproduct formed during the reaction, preventing acid-catalyzed degradation of the sensitive morpholine ring. The purification process involves simple extraction with ethyl acetate and decolorization with activated carbon, which effectively removes colored impurities and residual bases without the need for complex chromatographic techniques. This streamlined purification strategy ensures that the final high-purity N-phenylmorpholine compounds meet stringent quality specifications required for pharmaceutical applications.
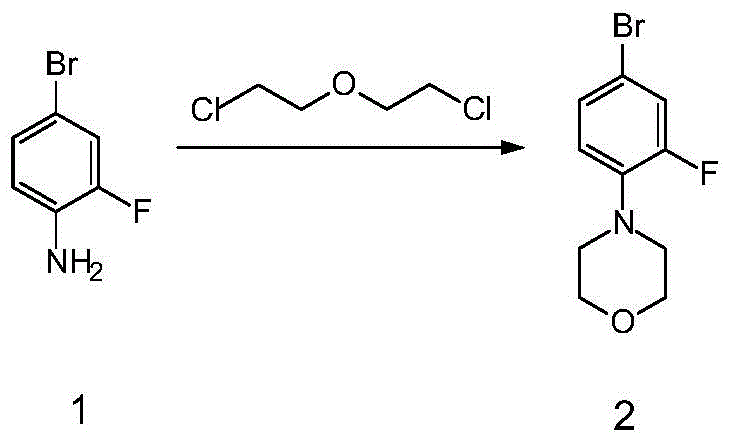
How to Synthesize Substituted N-Phenyl Morpholine Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to reaction parameters to maximize yield and safety. The process begins with the precise mixing of substituted aniline, which may contain various ortho-substituents such as fluorine, chlorine, or alkyl groups, with a significant excess of 2-chloroethyl ether. An organic base is then added to the mixture in a molar ratio optimized to drive the reaction to completion without generating excessive waste. The reaction vessel is heated to a temperature between 150°C and 160°C, and the mixture is maintained at this temperature for a period ranging from 4 to 24 hours, depending on the specific reactivity of the aniline substrate. Monitoring the reaction progress via TLC ensures that the conversion is complete before proceeding to the work-up stage. The detailed standardized synthesis steps for specific derivatives are outlined in the guide below, providing a clear roadmap for technical teams to replicate these high-yield results.
- Mix substituted aniline with excess 2-chloroethyl ether and an organic base such as triethylamine.
- Heat the reaction mixture to 150-160°C for 4 to 24 hours to facilitate ring closure.
- Remove excess solvent by distillation, extract with ethyl acetate, and purify to obtain the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis method translates into tangible strategic advantages that directly impact the bottom line and operational resilience. The elimination of expensive palladium catalysts and the reduction in solvent usage result in a drastic simplification of the raw material procurement process, reducing exposure to volatile metal markets and complex solvent supply chains. The ability to recover and reuse the excess 2-chloroethyl ether further enhances cost efficiency, creating a closed-loop system that minimizes waste disposal costs and raw material consumption. Moreover, the high yields achieved with this method, often exceeding 60% and reaching up to 80% for specific substrates, mean that less starting material is required to produce the same amount of product, effectively lowering the cost per kilogram of the final intermediate. These factors combined create a robust economic model that supports long-term cost reduction in pharmaceutical intermediates manufacturing without compromising on quality or regulatory compliance.
- Cost Reduction in Manufacturing: The removal of precious metal catalysts and the shift to a solvent-free process fundamentally alter the cost structure of production. By avoiding the need for column chromatography and complex purification steps, labor and equipment costs are significantly reduced. The use of low-price aniline and recoverable ether ensures that raw material costs remain stable and predictable. This qualitative shift in process chemistry allows for substantial cost savings that can be passed down the supply chain or reinvested into further R&D initiatives.
- Enhanced Supply Chain Reliability: The simplicity of the reaction conditions and the use of readily available commodity chemicals enhance the reliability of the supply chain. There is no dependency on specialized catalysts that may have long lead times or limited availability. The robustness of the process against variations in substrate structure means that a single production line can be adapted to manufacture a wide range of N-phenylmorpholine derivatives, increasing flexibility and reducing lead time for high-purity pharmaceutical intermediates. This adaptability is crucial for maintaining supply continuity in the face of market fluctuations.
- Scalability and Environmental Compliance: The solvent-free nature of this reaction makes it inherently safer and more environmentally friendly, aligning with modern green chemistry principles. The absence of large volumes of hazardous solvents reduces the risk of fire and explosion, simplifying safety protocols and insurance costs. The ease of scale-up is demonstrated by the simple work-up procedure involving distillation and extraction, which can be easily translated from laboratory to commercial scale. This scalability ensures that the commercial scale-up of complex pharmaceutical intermediates can be achieved rapidly and efficiently, meeting the growing demand for these critical drug components.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical disclosures within the patent, providing a reliable foundation for decision-making. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this process into their existing manufacturing workflows. The clarity provided here aims to eliminate ambiguity and foster confidence in the technology's potential for industrial application.
Q: What are the primary advantages of this solvent-free method over traditional Buchwald-Hartwig coupling?
A: The solvent-free method eliminates the need for expensive palladium catalysts and complex chromatography purification, resulting in significantly lower production costs and higher yields compared to the 2-10% yields typical of prior art methods.
Q: How does this process handle steric hindrance in substituted anilines?
A: The process utilizes high temperatures and excess 2-chloroethyl ether to drive the cyclization reaction even with sterically hindered anilines, achieving yields up to 80.4% where traditional methods often fail.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the method uses simple operation steps, easily recoverable raw materials, and avoids difficult-to-remove solvents, making it highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Phenyl Morpholine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthesis method described in patent CN106928162B for the production of high-value pharmaceutical intermediates. As a leading CDMO expert, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that this innovative chemistry can be seamlessly transitioned from the lab to the plant. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of N-phenylmorpholine compound meets the highest industry standards. We are committed to leveraging our technical expertise to optimize this solvent-free process, delivering consistent quality and reliability to our global partners.
We invite you to collaborate with us to explore how this advanced synthesis route can enhance your supply chain efficiency and reduce your manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions about integrating this technology into your portfolio. Together, we can drive innovation and efficiency in the production of critical pharmaceutical intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
