Advancing Anticancer Intermediate Production via Efficient Iron-Catalyzed Selenium Incorporation
Advancing Anticancer Intermediate Production via Efficient Iron-Catalyzed Selenium Incorporation
The development of novel heterocyclic scaffolds containing selenium atoms has garnered significant attention in medicinal chemistry due to their potent biological activities, particularly in oncology. Patent CN112174957B introduces a robust and environmentally friendly methodology for the synthesis of 5-selenoindolo[2,1-a]isoquinoline-6(5H)-one derivatives, a class of compounds exhibiting promising antitumor properties against cell lines such as MGC-803 and HeLa. This technical disclosure represents a pivotal advancement for reliable pharmaceutical intermediate suppliers seeking to diversify their portfolio with high-value selenium-containing scaffolds. By leveraging an iron-catalyzed cascade reaction, the process circumvents the need for expensive noble metals and harsh reaction conditions, thereby aligning with modern green chemistry principles while maintaining high synthetic efficiency.
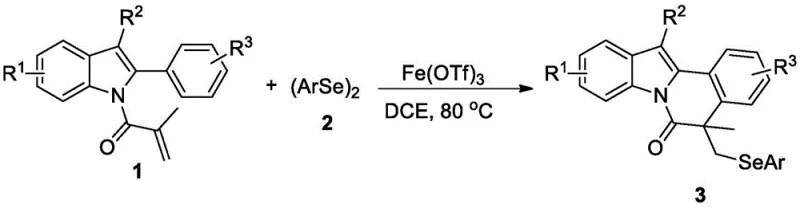
The strategic importance of this technology lies in its ability to construct complex polycyclic frameworks in a single operational step. Traditional approaches to similar fused indole systems often require multi-step sequences involving protecting group manipulations and stoichiometric oxidants, which generate substantial waste and increase production costs. In contrast, the disclosed method utilizes readily available 2-arylindoles and diaryl diselenides as building blocks. The reaction proceeds smoothly in 1,2-dichloroethane (DCE) at a moderate temperature of 80°C under an air atmosphere. This operational simplicity is a critical factor for cost reduction in pharmaceutical intermediates manufacturing, as it eliminates the capital expenditure associated with specialized inert atmosphere reactors and complex downstream processing units required for sensitive organometallic transformations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of selenium-functionalized heterocycles has been plagued by several inherent challenges that hinder commercial scalability. Conventional protocols frequently rely on palladium or copper catalysis, which not only inflates the raw material costs but also introduces the risk of heavy metal contamination in the final active pharmaceutical ingredient (API). Removing trace amounts of these toxic metals to meet regulatory limits (often in the parts per million range) necessitates additional purification steps such as scavenging or recrystallization, which inevitably erode overall yield. Furthermore, many existing selenylation reactions demand strict anaerobic conditions to prevent the oxidation of sensitive intermediates, requiring nitrogen or argon blanketing that complicates reactor design and increases utility consumption. The use of stoichiometric oxidants or harsh acidic conditions in older methods can also lead to poor functional group tolerance, limiting the structural diversity accessible to medicinal chemists exploring structure-activity relationships (SAR).
The Novel Approach
The methodology described in the patent data offers a transformative solution by employing ferric trifluoromethanesulfonate (Fe(OTf)3) as a Lewis acid catalyst to drive the cyclization. This approach is distinctively advantageous because it operates efficiently under aerobic conditions, utilizing molecular oxygen from the air as the terminal oxidant or simply tolerating its presence without degradation. The reaction exhibits excellent atom economy, incorporating the selenium moiety directly into the growing ring system without the need for pre-functionalized leaving groups. Experimental data from the patent indicates that this system is highly robust, delivering isolated yields ranging from 68% to 80% across a variety of substrates. For procurement teams, this translates to a more predictable supply chain where yield fluctuations are minimized, and the reliance on volatile precious metal markets is completely eradicated. The simplicity of the workup procedure, involving basic concentration and column chromatography, further underscores the practical viability of this route for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Fe(OTf)3-Catalyzed Cyclization
The mechanistic pathway likely involves the activation of the diaryl diselenide by the iron catalyst, generating an electrophilic selenium species that attacks the electron-rich indole nucleus. This initial selenylation step is followed by an intramolecular cyclization where the carbonyl oxygen or an adjacent nucleophilic site attacks the activated intermediate, closing the isoquinoline ring. The use of Fe(OTf)3 is particularly effective due to its strong Lewis acidity, which facilitates the cleavage of the Se-Se bond and stabilizes the developing cationic intermediates during the ring-closing event. Unlike radical pathways that might require photochemical initiation or high-energy thermal inputs, this ionic or semi-ionic mechanism proceeds smoothly at 80°C. Understanding this mechanism is vital for R&D directors aiming to optimize the process further, as it suggests that electron-donating groups on the indole ring could potentially accelerate the reaction rate by increasing nucleophilicity, while electron-withdrawing groups might require slight adjustments in catalyst loading or reaction time.
From an impurity control perspective, the specificity of the iron catalyst plays a crucial role in minimizing side reactions. The formation of byproducts such as non-cyclized selenylated indoles or polymerized species is suppressed by the rapid kinetics of the intramolecular cyclization step once the initial selenylation occurs. The patent data highlights the successful synthesis of derivatives with diverse substituents, including halogens like fluorine and bromine, as well as alkyl and alkoxy groups, without significant degradation of the core scaffold. This high level of chemoselectivity ensures that the crude reaction mixture is relatively clean, simplifying the purification burden. For quality assurance teams, this means that the impurity profile is consistent and manageable, reducing the risk of unexpected genotoxic impurities that often arise from uncontrolled radical processes. The ability to tolerate sensitive functional groups also allows for late-stage functionalization strategies, enabling the rapid generation of analog libraries for biological screening.
How to Synthesize 5-Selenoindolo[2,1-a]isoquinoline-6(5H)-one Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production. The process begins with the precise weighing of the 2-arylindole substrate and the corresponding diaryl diselenide, ensuring a stoichiometric balance that maximizes conversion. The addition of the iron catalyst in a 1:1 molar ratio relative to the substrates, as demonstrated in the examples, drives the reaction to completion within a short timeframe of approximately 1.5 hours. Monitoring the reaction via Thin Layer Chromatography (TLC) allows operators to determine the exact endpoint, preventing over-reaction or decomposition. Following the reaction, the solvent is removed under reduced pressure, and the residue is subjected to flash column chromatography using a standard gradient of ethyl acetate and petroleum ether. This straightforward isolation technique is compatible with standard industrial equipment, facilitating a seamless transition from benchtop discovery to kilogram-scale manufacturing.
- Charge a sealed tube with 2-arylindole substrate, diaryl diselenide, and Fe(OTf)3 catalyst in 1,2-dichloroethane (DCE) solvent.
- Stir the reaction mixture in air at 80°C for approximately 1.5 hours, monitoring progress via TLC until completion.
- Concentrate the mixture under reduced pressure and purify the crude residue using flash column chromatography with ethyl acetate and petroleum ether.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders responsible for sourcing and logistics, the adoption of this iron-catalyzed technology presents compelling economic and operational benefits. The shift away from precious metal catalysis fundamentally alters the cost structure of the synthesis, removing a major variable expense that is subject to global market fluctuations. Additionally, the use of commodity chemicals like DCE and simple indole derivatives ensures that raw material availability remains stable, mitigating the risk of supply disruptions. The robustness of the reaction under air atmosphere further reduces the complexity of the manufacturing infrastructure, allowing for production in standard glass-lined or stainless steel reactors without the need for specialized inert gas systems. These factors collectively contribute to a more resilient and cost-effective supply chain for high-value selenium intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive palladium or rhodium catalysts results in a direct and significant decrease in raw material costs. Furthermore, the simplified workup procedure reduces solvent consumption and labor hours associated with complex purification steps. By avoiding the need for metal scavengers or extensive recrystallization processes to remove heavy metals, the overall processing cost is substantially lowered, enhancing the profit margin for the final API or intermediate.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials such as 2-arylindoles and diaryl diselenides ensures a stable supply base. Unlike specialized organometallic reagents that may have long lead times or limited suppliers, these precursors are commoditized and can be sourced from multiple vendors globally. This diversification of the supply base reduces the risk of bottlenecks and ensures continuous production capability even during periods of market volatility or logistical constraints.
- Scalability and Environmental Compliance: The reaction conditions are mild and safe, operating at moderate temperatures without high pressure or hazardous reagents. This makes the process inherently safer to scale up to multi-ton quantities. Moreover, the use of iron, a non-toxic and abundant metal, aligns with increasingly stringent environmental regulations regarding heavy metal discharge. The reduced waste generation and lower energy requirements for heating and inert gas maintenance contribute to a smaller carbon footprint, supporting corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for potential partners and licensees. Understanding these details is essential for evaluating the feasibility of integrating this route into existing production workflows.
Q: What are the advantages of using Fe(OTf)3 over traditional noble metal catalysts?
A: Iron catalysts like Fe(OTf)3 are significantly more cost-effective and environmentally benign compared to palladium or rhodium systems. They operate efficiently under aerobic conditions without requiring inert gas protection, simplifying the operational setup for large-scale manufacturing.
Q: What is the substrate scope for this selenylation cyclization reaction?
A: The method demonstrates broad tolerance for various substituents on the indole and phenyl rings, including halogens (F, Br), alkyl groups (methyl, ethyl), and electron-donating groups (methoxy). It accommodates both symmetric and asymmetric diaryl diselenides effectively.
Q: How is the final product purified to meet pharmaceutical standards?
A: The process utilizes standard flash column chromatography with a simple ethyl acetate and petroleum ether eluent system. This ensures high purity removal of unreacted starting materials and side products, yielding compounds suitable for further biological evaluation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Selenoindolo[2,1-a]isoquinoline-6(5H)-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug discovery and development. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries can be translated into reliable commercial supplies. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to handle complex heterocyclic chemistry, including selenium incorporation, positions us as a strategic partner for pharmaceutical companies seeking to secure their supply chains for next-generation anticancer agents.
We invite you to collaborate with us to explore the full potential of this iron-catalyzed technology for your specific project needs. Whether you require custom synthesis services or bulk supply of key intermediates, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can drive efficiency and innovation in your pharmaceutical manufacturing operations.
