Advanced Synthesis of 1,3,4,6-Tetra-Substituted Pyridone Derivatives for Commercial Scale-Up
Advanced Synthesis of 1,3,4,6-Tetra-Substituted Pyridone Derivatives for Commercial Scale-Up
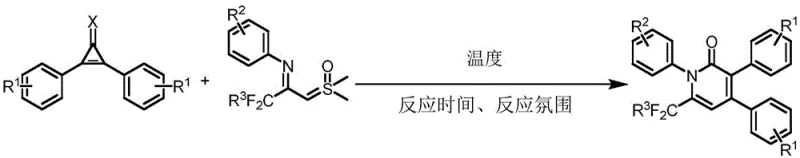
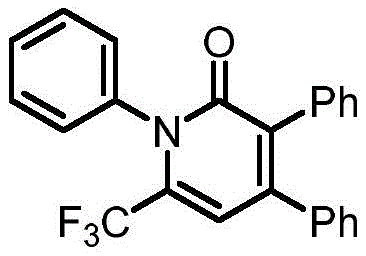
The pharmaceutical industry constantly seeks robust and efficient synthetic routes for complex heterocyclic scaffolds, particularly those serving as critical building blocks for active pharmaceutical ingredients (APIs). A significant breakthrough in this domain is documented in Chinese Patent CN112174880B, which discloses a novel preparation method for 1,3,4,6-tetra-substituted pyridone derivatives. This technology addresses long-standing challenges in heterocyclic chemistry by utilizing a unique condensation strategy between cyclopropenone derivatives and sulfur ylides. The resulting process not only achieves exceptional chemical selectivity but also operates under remarkably mild thermal conditions, typically ranging from 60°C to 100°C. For R&D directors and process chemists, this represents a paradigm shift away from harsh, multi-step sequences towards a more convergent and atom-economical approach. The ability to construct the pyridone core with such precision opens new avenues for generating diverse libraries of bioactive compounds, directly impacting the speed and cost of drug discovery pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of substituted 2-pyridones has relied on methodologies that often suffer from significant drawbacks, limiting their utility in large-scale manufacturing. Classical approaches frequently involve the use of expensive transition metal catalysts, which introduce complications regarding residual metal removal and stringent regulatory compliance for final drug substances. Furthermore, many existing routes require extreme reaction conditions, such as very high temperatures or strong acidic/basic environments, which can lead to the degradation of sensitive functional groups and the formation of complex impurity profiles. These factors collectively result in lower overall yields and increased production costs, creating bottlenecks for procurement teams aiming to secure reliable supplies of high-purity intermediates. Additionally, the lack of regioselectivity in some conventional cyclization reactions necessitates difficult separation processes, further eroding profit margins and extending lead times for commercial batches.
The Novel Approach
In stark contrast, the methodology outlined in patent CN112174880B offers a streamlined solution that circumvents these historical inefficiencies. By leveraging the inherent ring strain of cyclopropenone derivatives and the nucleophilic character of sulfur ylides, this novel approach facilitates a smooth ring-expansion and cyclization sequence. The reaction proceeds with excellent regioselectivity, ensuring that the carbonyl carbon-carbon bond of the cyclopropenone breaks preferentially, followed by the alkenyl bond cleavage, to form the desired pyridone skeleton cleanly. This specificity drastically reduces the burden on downstream purification, allowing for simpler workup procedures involving basic extraction and chromatography. Moreover, the compatibility of this method with a wide range of solvents, including the option for solvent-free conditions, underscores its versatility and potential for green chemistry applications. For supply chain managers, this translates to a more resilient manufacturing process that is less dependent on specialized reagents and more adaptable to varying production scales.
Mechanistic Insights into Cyclopropenone-Sulfur Ylide Condensation
Understanding the mechanistic underpinnings of this transformation is crucial for optimizing the process for industrial application. The reaction initiates with the nucleophilic attack of the sulfur ylide on the electrophilic centers of the cyclopropenone derivative. The unique electronic structure of the cyclopropenone ring, characterized by significant anti-aromatic character and ring strain, makes it highly susceptible to ring-opening events. Upon interaction with the ylide, the system undergoes a concerted rearrangement where the three-membered ring expands to form the six-membered pyridone core. This pathway is energetically favorable and avoids the high-energy intermediates often associated with traditional cyclization methods. The presence of electron-withdrawing groups, such as the trifluoromethyl moiety on the sulfur ylide, further stabilizes the transition states, driving the reaction to completion with high efficiency. This mechanistic clarity allows chemists to fine-tune reaction parameters, such as temperature and stoichiometry, to maximize yield and minimize side reactions.
Impurity control is another critical aspect where this mechanism offers distinct advantages. The high specificity of the bond-breaking and bond-forming events ensures that side products are minimized. In conventional syntheses, competing pathways often lead to isomeric byproducts that are structurally similar to the target molecule, making them difficult to remove. However, in this novel route, the regioselectivity is intrinsic to the reaction design. The preferential cleavage of the carbonyl carbon-carbon bond dictates the substitution pattern on the resulting pyridone ring, effectively preventing the formation of unwanted regioisomers. This level of control is paramount for meeting the stringent purity specifications required by regulatory bodies for pharmaceutical intermediates. Consequently, the final product exhibits a cleaner impurity profile, reducing the need for extensive recrystallization or preparative HPLC, which are costly and time-consuming operations in a commercial setting.
How to Synthesize 1,3,4,6-Tetra-Substituted Pyridone Derivatives Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific operational protocols to ensure reproducibility and safety. The process begins with the precise weighing and mixing of the cyclopropenone derivative and the sulfur ylide in a suitable reaction vessel. While a variety of polar aprotic solvents like DMSO or DMF can be used, the patent highlights the feasibility of solvent-free conditions or the use of greener alternatives, aligning with modern sustainability goals. Once the reagents are combined, the mixture is heated to a controlled temperature range of 60°C to 100°C. Maintaining this thermal window is essential; temperatures that are too low may result in incomplete conversion, while excessive heat could potentially degrade the sensitive ylide reagent. The reaction typically proceeds over a period of 12 to 24 hours, after which the progress is monitored via TLC or HPLC to confirm full consumption of the starting materials.
- Mix cyclopropenone derivatives, sulfur ylides, and a suitable solvent in a reaction vessel.
- Heat the mixture to 60-100°C and maintain reaction for 12-24 hours to ensure complete conversion.
- Dilute with ethyl acetate, wash with water, dry the organic phase, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers substantial benefits that resonate deeply with procurement and supply chain stakeholders. The primary advantage lies in the significant reduction of manufacturing complexity. By eliminating the need for precious metal catalysts and avoiding harsh reaction conditions, the process lowers the barrier to entry for production facilities that may not possess specialized equipment for handling hazardous reagents. This simplification directly correlates to reduced capital expenditure and lower operational costs. Furthermore, the high yields reported in the patent examples, often exceeding 90%, mean that less raw material is wasted per unit of product produced. This efficiency is a key driver for cost reduction in pharmaceutical intermediate manufacturing, allowing companies to offer more competitive pricing to their clients while maintaining healthy profit margins.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound, primarily due to the elimination of expensive catalytic systems and the minimization of waste. Traditional methods often require palladium or rhodium catalysts, which are not only costly to purchase but also require sophisticated recovery systems to meet environmental regulations. By contrast, this metal-free approach utilizes readily available organic starting materials that are commoditized and easy to source. The simplified workup procedure, involving basic aqueous washes and standard drying agents, further reduces the consumption of utilities and consumables. Consequently, the overall cost of goods sold (COGS) is drastically lowered, providing a strategic advantage in price-sensitive markets. This cost efficiency enables manufacturers to absorb fluctuations in raw material prices more effectively, ensuring stable supply contracts for long-term partners.
- Enhanced Supply Chain Reliability: Supply chain resilience is heavily dependent on the availability and stability of raw materials. The precursors used in this synthesis, such as diphenylcyclopropenone and various fluorinated sulfur ylides, are commercially accessible and can be produced in bulk quantities without complex supply chains. This accessibility mitigates the risk of shortages that often plague specialized reagent markets. Additionally, the robustness of the reaction conditions means that the process is less susceptible to variations in ambient conditions or minor deviations in protocol, leading to consistent batch-to-batch quality. For supply chain heads, this reliability translates to predictable lead times and the ability to scale production rapidly in response to market demand. The ability to manufacture under solvent-free conditions also simplifies logistics by reducing the volume of hazardous solvents that need to be transported and stored on-site.
- Scalability and Environmental Compliance: Scaling a chemical process from the bench to the ton-scale is fraught with challenges, particularly regarding heat transfer and safety. The mild thermal profile of this reaction (60-100°C) makes it inherently safer and easier to scale compared to exothermic reactions requiring cryogenic cooling or extreme heating. The absence of toxic heavy metals simplifies the environmental compliance landscape, as there is no need for extensive wastewater treatment to remove metal residues. This "green" attribute is increasingly important as global regulations tighten around pharmaceutical manufacturing emissions. The straightforward purification via column chromatography or crystallization ensures that the process remains viable even at larger scales, where continuous processing techniques can be easily integrated. This scalability ensures that the technology can support the growing demand for pyridone-based APIs without compromising on quality or environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis technology, derived from the detailed specifications and experimental data provided in the patent documentation. These insights are intended to clarify the practical implications of adopting this method for industrial applications. Understanding these nuances helps stakeholders make informed decisions about integrating this chemistry into their existing portfolios. The answers reflect the consensus of technical experts who have analyzed the feasibility and advantages of this specific reaction pathway.
Q: What are the key advantages of this pyridone synthesis method?
A: The method offers excellent regioselectivity, mild reaction conditions (60-100°C), and high yields without requiring transition metal catalysts.
Q: Is this process scalable for industrial production?
A: Yes, the use of readily available raw materials, simple workup procedures, and solvent-free options makes it highly suitable for commercial scale-up.
Q: What types of substituents are tolerated in this reaction?
A: The reaction demonstrates a broad substrate scope, tolerating various groups including halogens, trifluoromethyl, alkyl, and aryl substituents on both reactants.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,3,4,6-Tetra-Substituted Pyridone Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the one described in CN112174880B for the development of next-generation therapeutics. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and high-quality supply of critical intermediates. Our state-of-the-art facilities are equipped to handle the specific requirements of this chemistry, including precise temperature control and efficient purification capabilities. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the highest international standards. Our commitment to excellence extends beyond mere production; we actively collaborate with our partners to optimize processes for maximum efficiency and cost-effectiveness.
We invite pharmaceutical companies and research institutions to explore the possibilities offered by this innovative pyridone synthesis route. Whether you require custom synthesis services for early-stage drug discovery or large-scale manufacturing for clinical trials, our team is ready to support your needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific project requirements. We encourage you to reach out for specific COA data and route feasibility assessments to see how we can accelerate your development timeline. Partnering with us means gaining access to a reliable supply chain and a wealth of technical expertise dedicated to your success.
