Advanced Catalytic Asymmetric Synthesis of Chiral Alpha-Deuterated Pyrrolidine Compounds for Commercial Scale-up
The pharmaceutical industry is increasingly recognizing the strategic value of deuterated drugs, which offer improved metabolic stability and potentially enhanced therapeutic profiles compared to their non-deuterated counterparts. Patent CN114349585A introduces a groundbreaking catalytic asymmetric synthesis method for producing chiral alpha-deuterated tetrahydropyrrolidine compounds, a structural motif prevalent in numerous bioactive molecules. This technology leverages a dual-strategy approach combining hydrogen-deuterium exchange with catalytic asymmetric 1,3-dipolar cycloaddition, utilizing readily available deuterium water as the deuterium source. For R&D directors and procurement specialists seeking reliable pharmaceutical intermediate suppliers, this innovation represents a significant leap forward in accessing high-purity deuterated building blocks efficiently. The method addresses the critical challenge of introducing deuterium precisely at chiral centers without compromising stereochemical integrity, a feat that has historically required cumbersome multi-step sequences.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of chiral deuterated compounds has been fraught with significant technical and economic hurdles. Conventional strategies often rely on the catalytic asymmetric reduction or protonation of pro-chiral compounds, which limits the structural diversity of accessible products. Furthermore, existing methods for constructing deuterated heterocycles like pyrrolidines frequently involve harsh reaction conditions, expensive deuterated reagents, and complex purification processes that result in low overall yields. The scarcity of efficient protocols for introducing deuterium directly into chiral centers during ring-forming reactions has created a bottleneck in the development of deuterated API intermediates. These limitations not only inflate the cost of goods but also extend lead times for high-purity pharmaceutical intermediates, making it difficult for supply chain heads to ensure consistent availability for clinical and commercial programs.
The Novel Approach
In stark contrast, the methodology disclosed in CN114349585A offers a streamlined and versatile solution. By employing a chiral copper complex catalyst, the process enables the direct incorporation of deuterium from inexpensive deuterium water into the alpha-position of the pyrrolidine ring during the cycloaddition event. This approach eliminates the need for pre-functionalized deuterated starting materials, thereby drastically simplifying the synthetic route. The reaction proceeds under mild conditions, typically between 0°C and 25°C, preserving sensitive functional groups and ensuring high stereocontrol. This novel pathway facilitates the commercial scale-up of complex pharmaceutical intermediates by reducing the number of unit operations and minimizing waste generation. The ability to tune the stereochemistry through the selection of specific chiral ligands further enhances the utility of this platform for diverse drug discovery campaigns.
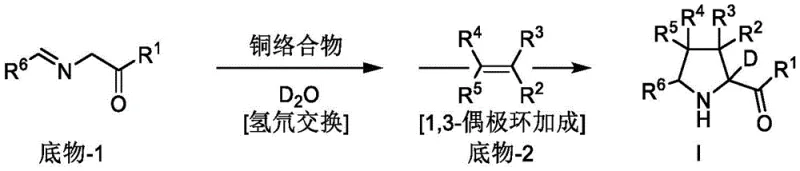
Mechanistic Insights into Copper-Catalyzed Asymmetric 1,3-Dipolar Cycloaddition
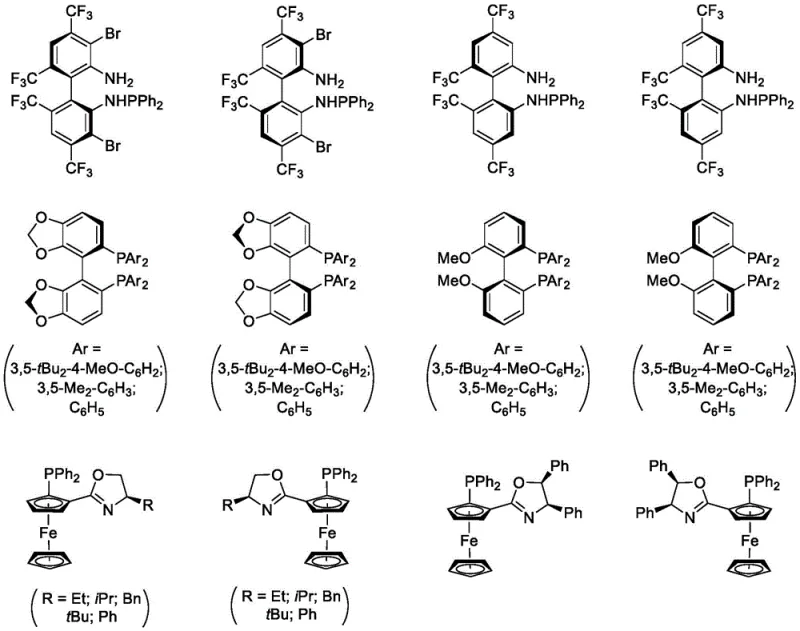
The core of this technological advancement lies in the sophisticated design of the chiral copper catalyst system. The reaction initiates with the formation of an active copper-azomethine ylide species from the imine ester substrate. In the presence of a base and deuterium water, a rapid hydrogen-deuterium exchange occurs at the alpha-position of the ylide precursor, effectively labeling the reactive intermediate with deuterium before the ring closure takes place. The chiral environment provided by the ligand, such as the bis-phosphine or ferrocene-based structures illustrated in the patent, dictates the facial selectivity of the subsequent cycloaddition with the dipolarophile. This precise control ensures that the deuterium atom is installed with high fidelity at the desired stereocenter. The mechanistic elegance of this process allows for the simultaneous construction of the pyrrolidine skeleton and the installation of the isotopic label in a single operational sequence.
Impurity control is inherently managed through the high selectivity of the catalyst. The use of molecular sieves in the reaction mixture helps to sequester water generated during the exchange or present in the solvent, driving the equilibrium towards the deuterated product and preventing back-exchange with protic impurities. The robustness of the copper catalyst system tolerates a wide range of electron-withdrawing and electron-donating substituents on both the imine ester and the dipolarophile. This broad substrate scope means that potential side reactions, such as polymerization of the dipolarophile or hydrolysis of the imine, are minimized under the optimized conditions. Consequently, the crude reaction profiles are clean, facilitating easier downstream processing and ensuring that the final API intermediates meet stringent purity specifications required by regulatory agencies.
How to Synthesize Chiral Alpha-Deuterated Pyrrolidine Efficiently
The practical implementation of this synthesis is designed for ease of operation in standard laboratory and pilot plant settings. The general procedure involves the in-situ generation of the chiral copper catalyst by mixing a copper salt, such as Cu(CH3CN)4BF4, with the chosen chiral ligand in a dry organic solvent like dichloromethane or tetrahydrofuran. Once the catalyst is formed, the imine ester substrate is introduced along with deuterium water and an organic base to effect the H/D exchange. Following a brief incubation period at low temperature, the dipolarophile is added to trigger the cycloaddition. The detailed standardized synthesis steps are provided in the guide below.
- Prepare the chiral copper catalyst by mixing a copper salt like Cu(CH3CN)4BF4 with a chiral ligand in an organic solvent under inert atmosphere.
- Conduct hydrogen-deuterium exchange on the imine ester substrate using deuterium water (D2O) and an organic base at low temperature.
- Add the dipolarophile (e.g., maleate) to the reaction mixture to perform the asymmetric 1,3-dipolar cycloaddition, yielding the deuterated pyrrolidine product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented technology offers compelling advantages that directly impact the bottom line and supply chain resilience. The substitution of costly specialized deuterated reagents with commodity-grade deuterium water represents a fundamental shift in cost structure for deuterated intermediate manufacturing. This change not only lowers the direct material costs but also simplifies the logistics of raw material sourcing, as D2O is widely available and stable. For procurement managers, this translates into a more predictable pricing model and reduced exposure to supply volatility associated with niche chemical reagents. The simplified workflow also reduces the demand on production capacity, allowing for faster turnaround times.
- Cost Reduction in Manufacturing: The elimination of expensive deuterated starting materials and the reduction in synthetic steps lead to substantial cost savings in pharmaceutical intermediate manufacturing. By integrating the deuteration step directly into the ring-forming reaction, the process avoids the need for separate protection and deprotection sequences often required in traditional routes. This streamlining reduces solvent consumption, energy usage, and labor hours, contributing to a leaner and more economical production process. The high yield and selectivity further minimize the loss of valuable materials, maximizing the overall process efficiency.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions and the use of common reagents enhance the reliability of the supply chain. The method's tolerance to various substrates means that a single platform can be adapted to produce a library of different deuterated analogs without requiring significant process re-development. This flexibility is crucial for responding to changing demands in drug development pipelines. Furthermore, the scalability of the copper-catalyzed system ensures that production can be ramped up from gram to kilogram scales without encountering significant engineering challenges, securing a continuous supply for clinical trials and commercial launch.
- Scalability and Environmental Compliance: The process aligns well with green chemistry principles by reducing waste and utilizing safer reagents. The absence of harsh conditions and toxic heavy metals beyond the catalytic amount of copper simplifies waste treatment and disposal. This environmental compliance is increasingly important for maintaining operational licenses and meeting corporate sustainability goals. The ability to scale this reaction safely in large reactors ensures that the technology is viable for industrial production, supporting the long-term commercial viability of deuterated drug candidates.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthesis technology. Understanding these details is essential for evaluating the feasibility of this route for specific project needs. The answers are derived directly from the experimental data and claims presented in the patent documentation.
Q: What is the primary advantage of using D2O in this synthesis?
A: Using deuterium water (D2O) as the deuterium source significantly reduces raw material costs compared to expensive specialized deuterated reagents, while achieving high deuteration rates at the alpha position.
Q: Can this method be applied to complex drug molecules like Idasanutlin?
A: Yes, the patent explicitly demonstrates the successful application of this methodology for the precise alpha-position deuteration of the chiral drug molecule Idasanutlin (RG7388).
Q: What level of stereoselectivity can be expected?
A: The method achieves excellent stereoselectivity, with reported enantiomeric excess (ee) values often exceeding 90% and diastereomeric ratios (dr) greater than 20:1 depending on the specific substrate and ligand used.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Alpha-Deuterated Pyrrolidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this copper-catalyzed deuteration technology for the next generation of therapeutics. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to market supply is seamless. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of chiral alpha-deuterated pyrrolidine meets the highest international standards. We are equipped to handle the complexities of deuterated chemistry, providing you with a secure and compliant source for your critical API intermediates.
We invite you to collaborate with us to leverage this innovative synthesis route for your drug development programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating how this method can optimize your budget. Please contact us to request specific COA data and route feasibility assessments, and let us help you accelerate the delivery of your deuterated drug candidates to patients.
