Scalable Synthesis of 2-Nitroindole Derivatives via Copper-Catalyzed Transformation for Commercial API Production
Introduction to Advanced 2-Nitroindole Synthesis Technology
The pharmaceutical industry continuously seeks robust and scalable pathways for constructing heterocyclic scaffolds that serve as critical building blocks for bioactive molecules. Patent CN108689907B discloses a groundbreaking method for preparing 2-nitroindole derivatives, which are pivotal precursors for synthesizing 2-aminoindole compounds used as monoamine oxidase inhibitors in treating neurodegenerative disorders like Alzheimer's disease. This technology represents a significant leap forward in fine chemical synthesis, addressing long-standing challenges regarding raw material availability and reaction safety. By utilizing pyridine formyl indole derivatives as initiators, the process achieves high yields under remarkably mild conditions, offering a compelling alternative to legacy synthetic routes. For R&D directors and procurement specialists, understanding the nuances of this copper-catalyzed transformation is essential for securing a reliable supply of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-nitroindole derivatives has been plagued by significant operational hazards and supply chain bottlenecks. Traditional routes often rely on the use of sodium azide and hydrogen sulfide, reagents that pose severe risks of explosion and toxicity, thereby complicating regulatory compliance and increasing insurance costs for manufacturing facilities. Furthermore, existing methods frequently suffer from narrow substrate applicability and difficult-to-obtain starting materials, which restricts the chemical diversity available for drug discovery programs. These legacy processes typically involve multiple steps and harsh reaction conditions, leading to lower overall yields and generating substantial hazardous waste that requires costly disposal protocols. Consequently, the industry has faced persistent challenges in achieving cost reduction in pharmaceutical intermediate manufacturing while maintaining strict safety standards.
The Novel Approach
In stark contrast, the innovative methodology described in the patent utilizes a pyridine formyl indole derivative as a versatile initiator, which is easily accessible and available in a wide variety of substituted forms. This new route eliminates the need for dangerous azides or sulfides, replacing them with tert-butyl nitrite and a copper catalyst in a straightforward one-pot transformation. The reaction proceeds efficiently at temperatures ranging from room temperature to 100°C, often requiring only simple solvents like 1,4-dioxane or methanol. This shift not only drastically simplifies the post-treatment process but also expands the scope of accessible derivatives, allowing for the rapid generation of diverse chemical libraries. 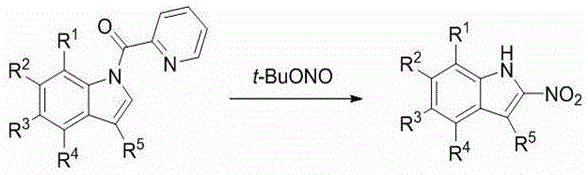 The structural versatility is further highlighted by the broad range of substituents tolerated on the indole ring, as depicted in the general formulas for both the starting materials and the final products.
The structural versatility is further highlighted by the broad range of substituents tolerated on the indole ring, as depicted in the general formulas for both the starting materials and the final products.
Mechanistic Insights into Copper-Catalyzed Nitrosation and Cyclization
The core of this technological advancement lies in the efficient copper-catalyzed transformation that converts the N-pyridine-2-formyl group into a nitro functionality at the 2-position of the indole ring. The reaction mechanism likely involves the activation of tert-butyl nitrite by the copper species, generating a reactive nitrosating agent that facilitates the cleavage of the amide bond and subsequent nitration. The use of catalysts such as copper acetate, cuprous iodide, or copper chloride at low molar loadings (0.05 to 0.15 equivalents) ensures high atom economy and minimizes metal contamination in the final product. This catalytic cycle operates effectively under an air atmosphere, removing the need for inert gas protection and further simplifying the operational setup for large-scale reactors. The tolerance of various functional groups, including halogens, cyano, and ester moieties, suggests a robust catalytic system that maintains activity despite electronic variations on the substrate.
From an impurity control perspective, the mild reaction conditions play a crucial role in minimizing side reactions such as over-nitration or decomposition of sensitive functional groups. The high selectivity observed in the examples, with yields consistently exceeding 75% and reaching up to 96%, indicates a clean reaction profile that simplifies downstream purification. By avoiding harsh acidic or basic conditions often associated with traditional nitration methods, the integrity of the indole core is preserved, ensuring that the resulting 2-nitroindole derivatives meet stringent purity specifications required for API synthesis. This level of control is vital for reducing lead time for high-purity pharmaceutical intermediates, as it reduces the burden on analytical quality control and purification teams.
How to Synthesize 2-Nitroindole Derivatives Efficiently
To implement this synthesis effectively, operators must adhere to precise stoichiometric ratios and reaction parameters outlined in the patent data. The standard protocol involves dissolving the pyridine formyl indole substrate, tert-butyl nitrite, and the copper catalyst in a selected solvent, followed by heating to the optimal temperature determined by the specific substrate's reactivity. Monitoring the reaction progress via Thin Layer Chromatography (TLC) is essential to determine the endpoint, ensuring complete conversion before proceeding to workup. The detailed standardized synthesis steps below provide a comprehensive guide for replicating this high-yielding transformation in a laboratory or pilot plant setting.
- Dissolve the pyridine formyl indole derivative, tert-butyl nitrite, and a copper catalyst (such as copper acetate or cuprous iodide) in a suitable solvent like 1,4-dioxane or methanol.
- Heat the reaction mixture to a temperature between room temperature and 100°C, preferably around 70°C, while stirring under an air atmosphere until TLC indicates completion.
- Separate the crude product via column chromatography using an ethyl acetate and petroleum ether system to isolate the high-purity 2-nitroindole derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits that extend beyond mere technical feasibility. The elimination of hazardous reagents like sodium azide directly translates to reduced safety compliance costs and lower insurance premiums, contributing to significant cost reduction in fine chemical manufacturing. Additionally, the reliance on commodity chemicals such as tert-butyl nitrite and common copper salts ensures a stable and resilient supply chain, mitigating the risks associated with sourcing specialized or controlled precursors. The simplicity of the workup procedure, which often requires only standard column chromatography, reduces solvent consumption and waste generation, aligning with modern green chemistry initiatives and environmental regulations.
- Cost Reduction in Manufacturing: The use of inexpensive copper catalysts at low loadings significantly lowers the raw material cost per kilogram of product compared to precious metal-catalyzed alternatives. Furthermore, the high yields reported across diverse substrates minimize material loss, enhancing the overall process efficiency and profitability. By streamlining the synthesis into fewer steps with milder conditions, energy consumption is also reduced, providing additional operational savings that improve the bottom line for commercial production.
- Enhanced Supply Chain Reliability: Since the starting pyridine formyl indole derivatives are easily obtained and available in wide varieties, manufacturers can secure raw materials from multiple sources, preventing bottlenecks. The robustness of the reaction conditions means that production schedules are less likely to be disrupted by sensitive parameter fluctuations, ensuring consistent delivery timelines for downstream clients. This reliability is critical for maintaining the continuity of API production lines that depend on these key intermediates.
- Scalability and Environmental Compliance: The method's suitability for industrial production is underscored by its simple operation and post-treatment process, which facilitates easy scale-up from gram to ton quantities without complex engineering modifications. The avoidance of toxic gases and explosive reagents simplifies waste treatment protocols, making it easier for facilities to meet increasingly strict environmental discharge standards. This combination of scalability and safety makes the process an ideal candidate for long-term commercial partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the process capabilities and limitations. Understanding these details helps stakeholders make informed decisions about integrating this methodology into their existing manufacturing portfolios.
Q: What are the advantages of this copper-catalyzed method over traditional synthesis routes?
A: Unlike conventional methods that require hazardous reagents like sodium azide or hydrogen sulfide, this novel approach utilizes mild conditions, inexpensive copper catalysts, and readily available tert-butyl nitrite, significantly enhancing operational safety and reducing environmental impact.
Q: What is the typical yield and purity profile for these 2-nitroindole derivatives?
A: The patent data demonstrates consistently high yields ranging from 75% to 96% across various substrates. The simple post-treatment process involving column chromatography ensures high purity specifications suitable for downstream pharmaceutical applications.
Q: Can this process be scaled for industrial production of API intermediates?
A: Yes, the method is explicitly designed for industrial suitability due to its simple operation, mild reaction temperatures (40-100°C), and the use of common organic solvents, making the commercial scale-up of complex pharmaceutical intermediates highly feasible.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Nitroindole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving medications. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We are committed to delivering products that adhere to stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify every batch. Our expertise in copper-catalyzed transformations allows us to optimize this specific route for maximum efficiency and cost-effectiveness.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your supply chain goals and accelerate your time to market.
