Scaling High-Performance Halogen-Free Flame Retardant Intermediates for Global Polymer Markets
Introduction to Advanced Flame Retardant Intermediate Synthesis
The global demand for high-performance, halogen-free flame retardants in engineering thermoplastics has necessitated a shift towards more sophisticated chemical intermediates. Patent CN102164932A introduces a groundbreaking process for preparing monocarboxy-functionalized dialkylphosphinic acids, esters, and salts, which serve as critical precursors for next-generation polymer additives. This technology addresses the long-standing industry challenge of achieving high fire resistance without compromising the mechanical integrity of polymers like polybutylene terephthalate (PBT) and polyamide 66 (PA66). By leveraging a multi-step catalytic sequence involving phosphinic acid sources, olefins, and alkylene oxides, this method offers a robust pathway to produce compounds that deliver exceptional thermal stability and flame retardancy.
The core innovation lies in the strategic functionalization of the phosphinic acid backbone. Unlike traditional methods that often rely on harsh halogenated reagents or result in complex mixtures difficult to separate, this process utilizes alkylene oxides to introduce carboxy-functionality with high precision. This specificity is paramount for R&D directors seeking consistent impurity profiles and reliable batch-to-batch reproducibility. The resulting intermediates can be further converted into various metal salts (such as Aluminum, Zinc, or Titanium salts), which are highly effective in promoting char formation during combustion, thereby protecting the underlying polymer matrix from thermal degradation.
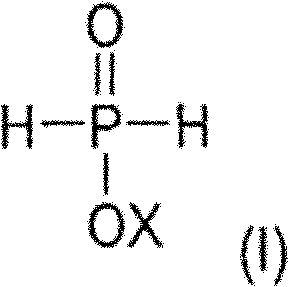
Furthermore, the versatility of this synthetic route allows for the customization of the organic groups attached to the phosphorus atom. By varying the olefin and alkylene oxide inputs, manufacturers can tailor the solubility, compatibility, and migration resistance of the final flame retardant additive. This adaptability makes the technology particularly valuable for supply chain heads who need to manage a diverse portfolio of polymer grades, from transparent films to reinforced molding compounds. The ability to synthesize these high-value intermediates efficiently positions suppliers to meet the stringent regulatory and performance requirements of the automotive, electronics, and construction sectors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of phosphorus-based flame retardants has been plagued by significant processing inefficiencies and environmental concerns. Conventional routes often involve the use of halogenated compounds as reactants, which introduce corrosive byproducts that require expensive neutralization and waste treatment protocols. These halogenated pathways not only increase the operational expenditure (OpEx) due to the need for specialized corrosion-resistant equipment but also pose risks to supply chain continuity regarding the disposal of hazardous waste. Moreover, traditional methods frequently suffer from low space-time yields, meaning that reactors must be operated for extended periods to achieve modest conversion rates, thereby limiting overall production capacity.
Another critical drawback of older technologies is the difficulty in isolating the desired mono-functionalized products. Without precise control over the reaction conditions, side reactions often lead to di-functionalized or oligomeric byproducts that degrade the mechanical properties of the final polymer. For procurement managers, this translates to higher costs associated with purification steps such as extensive chromatography or recrystallization. The presence of these impurities can also interfere with the polymerization process itself, leading to off-spec resin batches that must be scrapped. Consequently, there has been a persistent market need for a cleaner, more direct synthesis route that minimizes waste and maximizes the yield of the active flame-retarding species.
The Novel Approach
The process disclosed in CN102164932A represents a paradigm shift by eliminating interfering halogen compounds entirely from the reactant list. Instead, it employs a catalytic addition of phosphinic acid sources to olefins, followed by a ring-opening reaction with alkylene oxides. This approach is not only environmentally superior but also economically advantageous due to the high atom economy of the reactions involved. The use of alkylene oxides, which are commodity chemicals available in large volumes, ensures a stable and cost-effective raw material base. This stability is crucial for supply chain heads looking to mitigate risks associated with volatile specialty chemical markets.
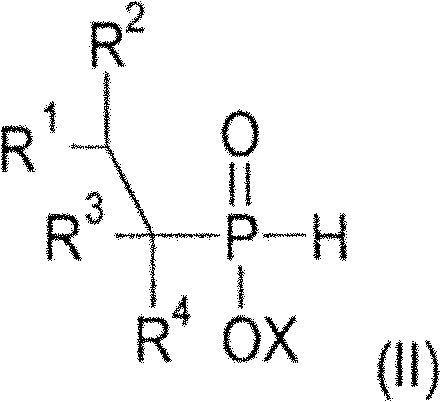
Moreover, this novel approach allows for the final product to be readily obtained or isolated under specific reaction conditions, such as transesterification, in a targeted manner. The ability to directly synthesize esters or salts without intermediate isolation steps significantly streamlines the manufacturing workflow. For instance, the process can be tuned to produce aluminum or zinc salts directly in the reaction medium, which can then be filtered and dried. This reduction in unit operations translates to substantial capital expenditure (CapEx) savings for manufacturing facilities. Additionally, the high selectivity of the catalysts used ensures that the final product meets the rigorous purity specifications required for high-end engineering applications, reducing the burden on quality control laboratories.
Mechanistic Insights into Transition Metal and Lewis Acid Catalysis
The success of this synthesis relies on a sophisticated interplay of three distinct catalytic systems, each optimized for a specific transformation. In the first step, the reaction between the phosphinic acid source and the olefin is mediated by Catalyst A, which typically comprises transition metals from groups 7, 8, 9, or 10, such as Palladium, Platinum, Rhodium, or Nickel. These metals, often coordinated with phosphine ligands like triphenylphosphine or bidentate ligands like BINAP, facilitate the formation of the P-C bond with high regioselectivity. The choice of ligand is critical; for example, bulky phosphines can prevent the formation of unwanted di-alkylated byproducts, ensuring that the mono-alkylphosphonous acid is the dominant species. This level of control is essential for maintaining the stoichiometry required for subsequent functionalization steps.
Following the initial alkylation, the intermediate alkylphosphonous acid reacts with an alkylene oxide in the presence of Catalyst B, a Lewis acid. Common Lewis acids for this step include metal halides such as Zinc Chloride, Aluminum Chloride, or Iron(III) Chloride. The Lewis acid activates the epoxide ring of the alkylene oxide, making it susceptible to nucleophilic attack by the phosphorus center. This ring-opening reaction is highly exothermic and requires precise temperature control, typically between -78°C and 100°C, to prevent polymerization of the epoxide. The mechanistic precision here ensures that the hydroxyl group is introduced at the correct position, setting the stage for the final oxidation. This step exemplifies the kind of chemical engineering rigor that R&D directors look for when evaluating process feasibility, as it demonstrates a clear understanding of reaction kinetics and thermodynamics.
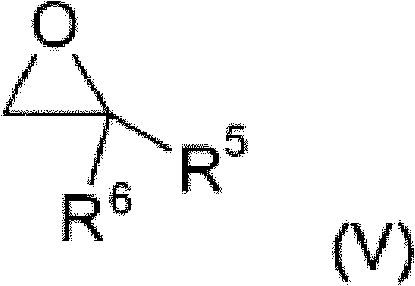
The final transformation involves the selective oxidation of the hydroxyl-containing intermediate to the corresponding carboxylic acid derivative. This is achieved using Catalyst C, which can be similar to Catalyst A (e.g., Platinum or Palladium on carbon) but operates under oxidative conditions using molecular oxygen or hydrogen peroxide. The mechanism likely involves the formation of a metal-peroxo species that abstracts hydrogen from the alcohol, converting it to an aldehyde and subsequently to a carboxylic acid. The patent highlights that this oxidation can be performed in the presence of water, which simplifies the solvent system and reduces the need for anhydrous conditions. This tolerance to water is a significant practical advantage, as it lowers the energy consumption associated with solvent drying and allows for the use of more economical aqueous workup procedures. The result is a monocarboxy-functionalized dialkylphosphinic acid that is ready for salt formation or esterification.
How to Synthesize Monocarboxy-Functionalized Dialkylphosphinic Acids Efficiently
Implementing this synthesis route requires a thorough understanding of the catalytic cycles and safety protocols associated with handling alkylene oxides and transition metals. The process is designed to be scalable, moving from laboratory glassware to industrial reactors with minimal modification. Key parameters such as pressure, temperature, and catalyst loading must be strictly controlled to ensure safety and maximize yield. For example, the oxidation step involving oxygen requires careful monitoring to stay outside the explosive limits of the gas mixture. Detailed standardized operating procedures are essential for training plant operators and ensuring consistent product quality across different production sites.
- React a phosphinic acid source with olefins in the presence of a transition metal catalyst (Catalyst A) to form alkylphosphonous acid derivatives.
- React the resulting alkylphosphonous acid with alkylene oxides using a Lewis acid catalyst (Catalyst B) to generate monofunctionalized dialkylphosphinic acid derivatives.
- Oxidize the monofunctionalized derivative using an oxidizing agent or oxygen/water with a catalyst (Catalyst C) to yield the final monocarboxy-functionalized product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this technology offers compelling strategic benefits beyond mere technical performance. The primary advantage is the drastic simplification of the supply chain for raw materials. By relying on commodity olefins and alkylene oxides rather than exotic halogenated precursors, manufacturers can leverage existing global logistics networks to secure feedstock at competitive prices. This reduces exposure to supply disruptions caused by regulatory crackdowns on hazardous chemical transport. Furthermore, the elimination of halogenated waste streams significantly lowers the cost of environmental compliance and waste disposal, contributing to a lower total cost of ownership for the manufacturing process.
- Cost Reduction in Manufacturing: The streamlined nature of this process, which avoids complex purification steps and utilizes high-yield catalytic reactions, leads to significant operational efficiencies. By reducing the number of unit operations and minimizing solvent usage, facilities can lower their energy consumption and labor costs. The high selectivity of the catalysts means that less raw material is wasted on byproducts, improving the overall material balance. Additionally, the ability to produce various metal salts directly in the reactor eliminates the need for separate neutralization and precipitation tanks, further reducing capital investment and footprint requirements for new production lines.
- Enhanced Supply Chain Reliability: The reliance on widely available industrial chemicals like ethylene oxide and propylene oxide ensures a stable supply base that is less susceptible to geopolitical fluctuations compared to specialty halogenated reagents. This stability allows for longer-term contracting and better inventory planning. Moreover, the robustness of the catalyst systems, which can often be recovered and reused or sourced from multiple vendors, mitigates the risk of single-source dependency. For supply chain heads, this translates to improved business continuity and the ability to meet sudden spikes in demand from the automotive or electronics sectors without compromising delivery schedules.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, with reaction conditions that are compatible with standard stainless steel reactors and continuous flow technologies. The absence of corrosive halogens extends the lifespan of equipment and reduces maintenance downtime. From an environmental perspective, the production of halogen-free flame retardants aligns with global trends towards sustainability and circular economy principles. This compliance facilitates easier market access in regions with strict environmental regulations, such as the European Union, and enhances the brand reputation of downstream polymer producers who market their products as eco-friendly.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is critical for stakeholders evaluating its integration into their production portfolios. The following questions address common concerns regarding catalyst selection, product purity, and application performance. These answers are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Clear communication of these technical details helps bridge the gap between R&D innovation and commercial implementation.
Q: What are the primary advantages of this alkylene oxide route over conventional halogenated methods?
A: This process eliminates the need for interfering halogen compounds as reactants, significantly reducing corrosive waste streams and simplifying downstream purification. It offers higher space-time yields and allows for the targeted production of specific esters or salts without complex separation steps.
Q: Which catalysts are essential for the selective oxidation step in this synthesis?
A: The selective oxidation (Step C) typically utilizes transition metal catalysts such as Platinum, Palladium, or Ruthenium, often supported on carbon or metal oxides. These catalysts enable the use of molecular oxygen or hydrogen peroxide, ensuring high selectivity for the carboxy-functional group while preserving the phosphinic acid core.
Q: Can these intermediates achieve V-0 ratings in engineering plastics like PBT and PA66?
A: Yes, experimental data within the patent demonstrates that polymers formulated with these monocarboxy-functionalized dialkylphosphinic acid salts can achieve UL94 V-0 ratings at thicknesses of 1.5mm. They also exhibit high Limiting Oxygen Index (LOI) values, often exceeding 36, indicating superior flame retardancy compared to untreated polymers.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Monocarboxy-Functionalized Dialkylphosphinic Acids Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the chemistry described in CN102164932A for the global flame retardant market. As a leading CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring these advanced intermediates to market. Our state-of-the-art facilities are equipped to handle the specific catalytic requirements of this process, including high-pressure hydrogenation and controlled oxidation steps. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch of monocarboxy-functionalized dialkylphosphinic acid meets the exacting standards of the polymer industry.
We invite you to collaborate with us to optimize this synthesis for your specific application needs. Whether you require custom metal salts for polyamide compounding or specific esters for polyester modification, our technical team is ready to assist. Please contact our technical procurement team to request a Customized Cost-Saving Analysis. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how partnering with us can accelerate your time-to-market for next-generation halogen-free flame retardant solutions.
