Scalable Manufacturing of High-Purity 3-(2-hydroxy-4-methoxyphenyl)-1-propanol for Cardiovascular Applications
Scalable Manufacturing of High-Purity 3-(2-hydroxy-4-methoxyphenyl)-1-propanol for Cardiovascular Applications
The pharmaceutical industry is constantly seeking robust and scalable synthetic routes for bioactive intermediates, particularly those targeting cardiovascular health. Patent CN101691324B introduces a significant advancement in the preparation of 3-(2-hydroxy-4-methoxyphenyl)-1-(4-methoxyphenyl)-1-propanol, a compound exhibiting potent anti-platelet aggregation activity. This technical insight report analyzes the novel methodology disclosed in the patent, highlighting its superiority over legacy synthetic pathways. By leveraging a strategic combination of aldol condensation and palladium-catalyzed hydrogenation, this process offers a streamlined approach to generating high-purity intermediates essential for developing next-generation antithrombotic therapies. The structural integrity and biological potential of this molecule make it a critical candidate for reliable pharmaceutical intermediates supplier networks aiming to support the growing demand for cardiovascular medications.
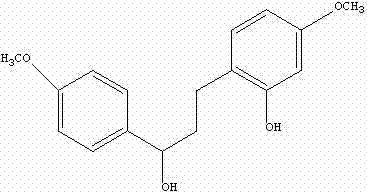
The core innovation lies in the efficient construction of the carbon skeleton and the simultaneous execution of reduction and deprotection steps. Traditional methods often suffer from multi-step sequences that introduce impurities and lower overall throughput. In contrast, the methodology outlined in CN101691324B utilizes readily available starting materials such as 2-benzyloxy-4-methoxybenzaldehyde and 4-methoxyacetophenone. The initial formation of the chalcone intermediate via base-catalyzed condensation sets the stage for a highly selective hydrogenation process. This approach not only simplifies the operational workflow but also ensures that the final product meets rigorous quality specifications required for downstream API synthesis. For R&D directors and process chemists, understanding the nuances of this catalytic system is key to implementing cost-effective manufacturing strategies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art techniques for synthesizing 3-(2-hydroxy-4-methoxyphenyl)-1-(4-methoxyphenyl)-1-propanol have historically been plagued by inefficiencies and operational complexities. For instance, early literature describes routes involving the heating of dimethoxyflavan-2-ene under acidic vapor conditions, followed by reduction with sodium borohydride. These methods are not only limited by the availability of specialized starting materials like flavan derivatives but also involve harsh reaction environments that can compromise product stability. Another notable conventional pathway involves hydroboration-oxidation sequences using boron trifluoride etherate and subsequent oxidation with hydrogen peroxide. This multi-step procedure is notoriously difficult to control, often resulting in low isolated yields, reported as low as 24% in some studies. Furthermore, the use of stoichiometric amounts of boron reagents generates significant waste and requires extensive purification efforts to remove residual inorganic salts, thereby increasing the environmental footprint and production costs.
The Novel Approach
The patented method revolutionizes this landscape by introducing a concise two-step sequence that maximizes atom economy and operational safety. The first step involves a classic aldol condensation between 2-benzyloxy-4-methoxybenzaldehyde and 4-methoxyacetophenone in an alcoholic solvent containing a base such as potassium hydroxide. This reaction proceeds smoothly at temperatures between 60°C and 100°C under an inert nitrogen atmosphere, yielding the chalcone intermediate with high conversion rates. The true breakthrough occurs in the second step, where the chalcone intermediate undergoes catalytic hydrogenation in the presence of Pd/C. Remarkably, this single operation achieves both the reduction of the double bond and the removal of the benzyl protecting group. This tandem transformation eliminates the need for separate deprotection steps, drastically reducing processing time and solvent consumption. The result is a streamlined process that delivers the target alcohol with exceptional purity, typically exceeding 96% as confirmed by HPLC analysis.
Mechanistic Insights into Pd/C-Catalyzed Hydrogenation and Deprotection
The success of this synthesis hinges on the precise mechanistic interplay during the catalytic hydrogenation phase. When the 2-benzyloxy-4,4’-dimethoxychalcone intermediate is exposed to hydrogen gas in the presence of a palladium on carbon catalyst, several concurrent reactions occur on the metal surface. Initially, the alkene double bond of the chalcone is hydrogenated to form the saturated ketone or alcohol precursor. Simultaneously, the benzyl ether moiety at the ortho-position undergoes hydrogenolysis. The palladium catalyst facilitates the cleavage of the carbon-oxygen bond in the benzyl group, releasing toluene as a byproduct and revealing the free phenolic hydroxyl group. This dual functionality is critical because it avoids the use of strong Lewis acids or harsh demethylating agents that could otherwise degrade the sensitive methoxy groups or the alcohol functionality on the side chain. The presence of a base in the reaction medium further assists in neutralizing any acidic byproducts and stabilizing the phenolic intermediate, ensuring a clean reaction profile.
Impurity control is inherently built into this mechanism due to the selectivity of the Pd/C catalyst. Unlike chemical reducing agents like sodium borohydride which might reduce other functional groups non-selectively, heterogeneous catalysis allows for fine-tuning of reaction parameters such as hydrogen pressure and temperature. The patent specifies a low hydrogen pressure range of 5 to 15 mmHg and a mild temperature window of 25°C to 40°C. These mild conditions prevent over-reduction or side reactions such as the hydrogenolysis of the methoxy groups, which are more stable under these specific parameters. Consequently, the crude product obtained after filtration of the catalyst contains minimal impurities, allowing for high recovery rates upon simple recrystallization from solvents like ethyl acetate. This mechanistic elegance translates directly into a robust manufacturing process capable of producing high-purity pharmaceutical intermediates consistently.
How to Synthesize 3-(2-hydroxy-4-methoxyphenyl)-1-propanol Efficiently
Implementing this synthesis requires careful attention to reaction stoichiometry and atmospheric control to ensure optimal yields and safety. The process begins with the preparation of the chalcone intermediate, where the molar ratio of aldehyde to ketone is carefully balanced, often with a slight excess of the aldehyde to drive the condensation to completion. Following the isolation of the intermediate, the hydrogenation step must be conducted in a sealed system equipped with pressure monitoring to maintain the specified low-pressure hydrogen environment.
- Perform aldol condensation between 2-benzyloxy-4-methoxybenzaldehyde and 4-methoxyacetophenone in an alkaline alcohol solution at 60-100°C to form the chalcone intermediate.
- Subject the intermediate to catalytic hydrogenation using Pd/C catalyst in an alkaline alcohol solution at 25-40°C under low hydrogen pressure to achieve reduction and deprotection simultaneously.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers substantial benefits for procurement managers and supply chain leaders focused on cost reduction in pharmaceutical intermediates manufacturing. The elimination of expensive and hazardous reagents such as boron trifluoride and organic peroxides significantly lowers the raw material costs and reduces the complexity of waste disposal. Furthermore, the use of common solvents like ethanol and propanol, along with widely available starting materials, ensures a stable supply chain that is less susceptible to market volatility. The simplified workflow, which combines reduction and deprotection into a single unit operation, reduces the overall equipment footprint and labor hours required per batch. This efficiency gain allows manufacturers to scale up production from pilot scales to multi-ton annual capacities without proportional increases in operational expenditure.
- Cost Reduction in Manufacturing: The primary driver for cost savings is the replacement of low-yielding, multi-step legacy processes with this high-efficiency route. By avoiding the use of stoichiometric reducing agents and complex workup procedures associated with hydroboration, the process minimizes reagent consumption. Additionally, the recovery of the palladium catalyst, although used in small quantities, adds value through potential recycling protocols. The high purity of the crude product reduces the need for extensive chromatographic purification, relying instead on cost-effective crystallization techniques. These factors collectively contribute to a significantly reduced cost of goods sold (COGS) for the final intermediate.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as 4-methoxyacetophenone and basic inorganic bases ensures that raw material sourcing is robust and geographically diverse. Unlike specialized flavan derivatives required by older methods, these precursors are produced by numerous global suppliers, mitigating the risk of supply disruptions. The mild reaction conditions also reduce the wear and tear on reactor vessels and ancillary equipment, leading to higher asset utilization rates and fewer unplanned maintenance downtimes. This reliability is crucial for maintaining continuous production schedules and meeting tight delivery windows for downstream API manufacturers.
- Scalability and Environmental Compliance: The process is inherently green, aligning with modern environmental regulations and sustainability goals. The absence of heavy metal waste (other than the recoverable Pd/C) and toxic boron residues simplifies effluent treatment and lowers environmental compliance costs. The ability to run the hydrogenation at near-atmospheric pressure enhances safety profiles, making it easier to obtain regulatory approvals for large-scale facilities. This scalability ensures that the supply of high-purity pharmaceutical intermediates can be rapidly expanded to meet surging market demand for cardiovascular therapeutics without compromising on quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of this cardiovascular intermediate. These answers are derived directly from the experimental data and claims presented in patent CN101691324B, providing a factual basis for decision-making.
Q: What are the advantages of this synthesis method over traditional hydroboration routes?
A: Unlike traditional methods requiring complex hydroboration-oxidation steps with low yields (around 24%), this patented route utilizes a straightforward aldol condensation followed by one-pot hydrogenation, significantly simplifying the operation and improving overall yield and purity.
Q: What is the purity level achievable with this Pd/C catalyzed process?
A: The process described in patent CN101691324B consistently achieves high purity levels, with HPLC analysis confirming product purity between 96% and 98% after simple recrystallization, meeting stringent pharmaceutical intermediate standards.
Q: Is this intermediate suitable for large-scale cardiovascular drug manufacturing?
A: Yes, the method uses common raw materials like 4-methoxyacetophenone and operates under mild conditions (25-40°C for hydrogenation), making it highly scalable and safe for commercial production of anti-platelet aggregation agents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-(2-hydroxy-4-methoxyphenyl)-1-propanol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving cardiovascular drugs. Our team of expert process chemists has extensively evaluated the route described in CN101691324B and possesses the technical capability to optimize it for industrial-scale production. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instruments to guarantee stringent purity specifications for every batch of 3-(2-hydroxy-4-methoxyphenyl)-1-propanol we deliver. We are committed to being a partner that not only supplies chemicals but also provides technical solutions that enhance your overall project viability.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this advanced synthetic technology. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact us today to request specific COA data and route feasibility assessments for your upcoming projects. Let us help you accelerate your drug development timeline with a reliable supply of high-performance intermediates that meet the highest global standards.
