Advanced Synthesis of 4'-Hydroxy-4,6'-dimethoxydihydrochalcone for Pharmaceutical Applications
Introduction to Novel Dihydrochalcone Synthesis
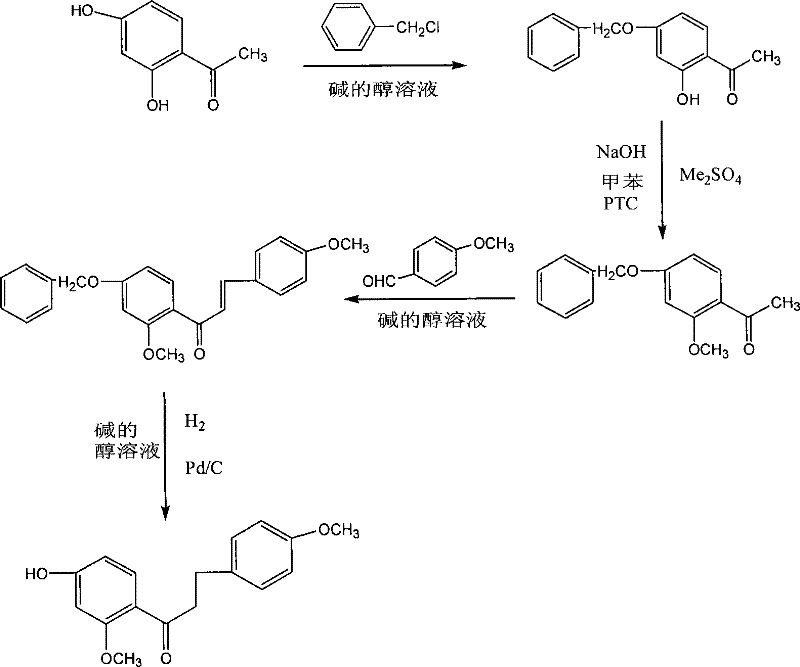
The pharmaceutical landscape continuously demands innovative intermediates that offer superior therapeutic profiles alongside manufacturability. Patent CN101182289B introduces a groundbreaking synthesis method for 4'-hydroxy-4,6'-dimethoxydihydrochalcone, a novel dihydrochalcone derivative with potent anti-platelet aggregation properties. This compound represents a significant advancement in cardiovascular therapeutic research, addressing the critical need for agents that can effectively manage thrombosis without the severe side effects associated with existing treatments. The patent details a robust, three-step synthetic pathway that begins with the methylation of 4-benzyloxy-2-hydroxyacetophenone, proceeds through an aldol condensation, and concludes with a sophisticated catalytic hydrogenation step that simultaneously achieves reduction and deprotection. This strategic approach not only ensures high chemical purity but also streamlines the production workflow, making it an attractive candidate for industrial adoption by reliable pharmaceutical intermediates suppliers seeking to optimize their portfolios.
From a structural perspective, the target molecule features a specific arrangement of methoxy and hydroxyl groups on the aromatic rings that are crucial for its biological activity. The synthesis described in the patent leverages modern organic synthesis techniques to construct this scaffold efficiently. By utilizing phase-transfer catalysis (PTC) in the initial step, the process overcomes solubility barriers often encountered in biphasic systems, ensuring high conversion rates. Furthermore, the final step employs palladium on carbon (Pd/C) under controlled hydrogen pressure, a method renowned for its selectivity and cleanliness in fine chemical manufacturing. This comprehensive methodology underscores a commitment to green chemistry principles by minimizing waste and avoiding toxic heavy metal reductants like zinc, which were common in historical precedents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of similar dihydrochalcone derivatives has been plagued by inefficiencies that hinder commercial viability. Literature reviews, such as those by Richard P. Duffley et al., reveal methodologies relying on methyl iodide for methylation, a process that notoriously requires extended reaction times of up to 72 hours to reach completion. Such prolonged durations drastically reduce reactor turnover rates and increase energy consumption, creating bottlenecks in production schedules. Other approaches, like those documented by V. K. Sharma, utilize anhydrous potassium carbonate in acetone under reflux for 24 hours, which, while slightly faster, still represents a significant time investment for a single transformation. Moreover, purification in these traditional routes often necessitates labor-intensive silica gel column chromatography, as seen in the work of Asit K. Chakraborti. This reliance on chromatographic separation is a major drawback for cost reduction in API manufacturing, as it generates substantial solvent waste and requires expensive stationary phases, rendering the process economically unfeasible for large-scale operations.
The Novel Approach
In stark contrast, the novel approach outlined in CN101182289B revolutionizes the production workflow by integrating efficiency at every stage. The utilization of dimethyl sulfate in the presence of a phase-transfer catalyst (benzyltriethylammonium chloride) and sodium hydroxide allows the methylation reaction to proceed rapidly at mild temperatures between 25 and 40 ℃, completing within just 3 to 6 hours. This dramatic reduction in reaction time directly translates to enhanced throughput. Furthermore, the subsequent aldol condensation is conducted in an alkaline alcoholic solution, yielding the chalcone intermediate with high precision. The true brilliance of this method lies in the final step, where catalytic hydrogenation serves a dual purpose: it reduces the olefinic double bond of the chalcone to form the dihydrochalcone backbone while simultaneously cleaving the benzyl protecting group to reveal the phenolic hydroxyl moiety. This telescoped operation eliminates the need for separate deprotection steps and avoids the use of zinc dust, thereby simplifying the post-reaction workup to mere filtration and recrystallization.
Mechanistic Insights into Pd/C-Catalyzed Hydrogenation and Deprotection
The core of this synthetic strategy relies on the sophisticated interplay between catalytic hydrogenation and hydrogenolysis. In the final stage, the intermediate 4'-benzyloxy-4,6'-dimethoxychalcone is subjected to hydrogen gas in the presence of a Pd/C catalyst within an alkaline alcoholic medium. Mechanistically, the palladium surface adsorbs molecular hydrogen, dissociating it into atomic hydrogen species which are then transferred to the substrate. The conjugated double bond of the chalcone is susceptible to reduction, accepting hydrogen atoms to form the saturated dihydrochalcone structure. Concurrently, the benzyl ether protecting group at the 4'-position undergoes hydrogenolysis. The benzylic carbon-oxygen bond is cleaved via a similar surface-catalyzed mechanism, releasing toluene as a byproduct and regenerating the free phenolic hydroxyl group. This dual functionality is critical because it allows for the installation of the hydroxyl group late in the synthesis, protecting it from unwanted side reactions during the earlier aldol condensation step, thus ensuring high-purity dihydrochalcone formation without the need for orthogonal protection strategies.
Impurity control is inherently managed through the selectivity of the Pd/C catalyst and the reaction conditions. By maintaining the hydrogen pressure between 5 and 15 mmHg and controlling the temperature between 20 and 60 ℃, the process minimizes over-reduction or hydrogenolysis of the methoxy groups, which are generally more stable under these specific conditions. The use of an alkaline environment during hydrogenation further aids in preventing acid-catalyzed side reactions that could degrade the sensitive ketone functionality. Additionally, the initial PTC methylation step is highly selective for the phenolic oxygen due to the specific pKa differences and the phase-transfer mechanism, preventing O-alkylation of other potential sites. This rigorous control over reaction parameters ensures that the final product, after simple ethanol recrystallization, achieves purity levels exceeding 99%, meeting the stringent specifications required for clinical-grade pharmaceutical intermediates.
How to Synthesize 4'-Hydroxy-4,6'-dimethoxydihydrochalcone Efficiently
The synthesis of this high-value intermediate is designed for operational simplicity and scalability, making it accessible for both laboratory optimization and industrial production. The process begins with the precise methylation of the starting acetophenone derivative, followed by a condensation reaction that builds the carbon skeleton, and concludes with the critical hydrogenation step. Each stage has been optimized to balance reaction kinetics with product quality, ensuring that operators can achieve consistent results. For detailed procedural specifics regarding reagent ratios, temperature controls, and workup procedures, please refer to the standardized synthesis guide below which outlines the exact experimental conditions validated in the patent examples.
- Perform phase-transfer catalyzed methylation of 4-benzyloxy-2-hydroxyacetophenone using dimethyl sulfate and NaOH in toluene.
- Conduct aldol condensation between the resulting 4-benzyloxy-2-methoxyacetophenone and 4-methoxybenzaldehyde in alkaline ethanol.
- Execute catalytic hydrogenation using Pd/C in alkaline alcohol to simultaneously reduce the double bond and remove the benzyl protecting group.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route offers transformative benefits that extend beyond mere chemical yield. The elimination of column chromatography is perhaps the most significant economic driver, as it removes a major cost center associated with silica gel procurement, solvent recovery, and waste disposal. This streamlined purification process, relying instead on crystallization, drastically simplifies the manufacturing workflow and reduces the overall environmental footprint of the production facility. Furthermore, the replacement of hazardous zinc dust with catalytic hydrogenation aligns with modern safety standards and regulatory requirements, mitigating the risks associated with handling pyrophoric metals and heavy metal waste streams. These factors collectively contribute to a more resilient and cost-effective supply chain for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The transition from stoichiometric reductants like zinc to catalytic hydrogenation fundamentally alters the cost structure of production. By utilizing a reusable heterogeneous catalyst (Pd/C), the process eliminates the continuous consumption of expensive metal powders and the subsequent costs of removing metal residues from the final product. The ability to perform deprotection and reduction in a single pot reduces the number of unit operations, saving on labor, energy, and equipment usage time. Additionally, the use of common solvents like toluene and ethanol, rather than specialized chlorinated solvents often required for chromatography, further drives down raw material expenses and simplifies solvent recycling protocols.
- Enhanced Supply Chain Reliability: The raw materials for this synthesis, specifically 4-benzyloxy-2-hydroxyacetophenone and 4-methoxybenzaldehyde, are commercially available and structurally simple, reducing the risk of supply disruptions associated with exotic or custom-synthesized precursors. The robustness of the reaction conditions, which tolerate a range of temperatures and pressures without compromising yield, ensures consistent batch-to-batch quality. This reliability is crucial for maintaining uninterrupted production schedules for downstream API manufacturers, allowing for better inventory planning and reduced safety stock requirements. The simplified process flow also means shorter lead times from order to delivery, enhancing the agility of the supply chain.
- Scalability and Environmental Compliance: Scaling this process from kilogram to multi-ton quantities is straightforward due to the absence of complex purification steps that often fail upon scale-up. The hydrogenation step is a standard unit operation in the fine chemical industry, easily accommodated by existing infrastructure. From an environmental perspective, the process generates significantly less hazardous waste compared to traditional methods. The avoidance of zinc salts and silica gel waste simplifies effluent treatment and reduces the burden on waste management systems. This alignment with green chemistry principles not only lowers compliance costs but also enhances the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 4'-hydroxy-4,6'-dimethoxydihydrochalcone. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for potential partners. Understanding these details is essential for evaluating the feasibility of integrating this intermediate into your specific drug development pipeline or manufacturing portfolio.
Q: What are the advantages of this synthesis over conventional methods?
A: Unlike conventional methods requiring prolonged reaction times (up to 72 hours) and complex column chromatography purification, this novel route utilizes efficient phase-transfer catalysis and one-pot hydrogenation/deprotection, significantly simplifying operations and improving purity.
Q: Is the process suitable for large-scale manufacturing?
A: Yes, the process avoids hazardous reagents like zinc dust and eliminates the need for silica gel column separation, making it highly amenable to commercial scale-up with standard industrial equipment.
Q: What is the biological activity of the target compound?
A: The compound demonstrates significant anti-platelet aggregation activity, superior to acetylsalicylic acid in specific concentration ranges, and exhibits low acute toxicity in animal models.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4'-Hydroxy-4,6'-dimethoxydihydrochalcone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of next-generation cardiovascular therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our expertise in catalytic hydrogenation and phase-transfer catalysis allows us to optimize this specific synthesis route for maximum efficiency and minimal environmental impact, providing you with a sustainable sourcing solution.
We invite you to collaborate with us to explore the full potential of this novel compound for your research and development needs. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project requirements, demonstrating how our optimized manufacturing processes can reduce your overall procurement costs. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and sample availability. Let us be your partner in advancing pharmaceutical innovation through superior chemical synthesis and supply chain excellence.
