Advanced Purification Technology for High-Purity Telavancin Hydrochloride Production
Introduction to Advanced Telavancin Purification Technologies
The pharmaceutical industry continuously seeks robust methodologies to ensure the highest quality of second-generation glycopeptide antibiotics, particularly for treating resistant bacterial infections. Patent CN107629115B introduces a groundbreaking purification method for Telavancin, addressing critical challenges related to impurity profiles and product stability that have long plagued manufacturers. This technology leverages a sophisticated combination of octadecyl-bonded silica gel chromatography and advanced nanofiltration techniques to isolate high-purity Telavancin hydrochloride from complex reaction mixtures. For R&D directors and procurement specialists, understanding this process is vital as it represents a significant leap forward in reliable telavancin supplier capabilities, ensuring that the final active pharmaceutical ingredient meets rigorous global regulatory standards. The method specifically targets the removal of difficult-to-separate dimers and structural analogues that typically co-elute in traditional processes.
Furthermore, the integration of this purification strategy into commercial manufacturing lines offers substantial advantages in terms of process control and yield consistency. By optimizing the interaction between the target molecule and the stationary phase through precise pH and solvent gradients, the process minimizes product degradation often seen during prolonged processing times. This technical insight report delves deep into the mechanistic underpinnings of this innovation, providing a comprehensive analysis for stakeholders interested in cost reduction in antibiotic manufacturing and the secure sourcing of critical antimicrobial agents. The ability to consistently produce material with purity levels exceeding established thresholds is a key differentiator in the competitive landscape of generic and branded antibiotic production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of semi-synthetic glycopeptide antibiotics like Telavancin has relied heavily on liquid-liquid extraction and ion exchange resin chromatography, methods that are increasingly recognized for their inefficiencies in handling complex molecular architectures. Traditional ion exchange resins often lack the specific selectivity required to distinguish between the target Telavancin molecule and its closely related impurities, such as dimers formed during the reductive amination and Mannich reaction steps. These dimers possess similar charge properties to the target compound, making them exceptionally difficult to remove using standard electrostatic interactions alone, which frequently results in final products that fail to meet the stringent purity specifications required for clinical application. Moreover, the multi-step extraction processes involve extensive use of organic solvents and aqueous phases, leading to significant operational complexity, higher waste generation, and increased risk of product loss during phase transfers.
Another critical drawback of conventional methods is the instability of the glycopeptide backbone under prolonged processing conditions. Vancomycin derivatives are known to be susceptible to oxidation, particularly at the phenolic and diphenolic moieties within their structure, which can lead to undesirable color changes ranging from yellow to red upon exposure to light and air over time. Conventional crystallization and drying steps, such as lyophilization, can exacerbate these stability issues due to thermal stress or extended exposure to suboptimal pH environments. Consequently, manufacturers often face batch-to-batch variability and reduced shelf-life, creating significant supply chain vulnerabilities. The inability to effectively separate these degradation products and synthetic by-products necessitates additional reprocessing steps, further driving up production costs and extending lead times for high-purity glycopeptide antibiotics.
The Novel Approach
The innovative approach detailed in patent CN107629115B fundamentally shifts the purification paradigm by employing octadecyl-bonded silica gel (ODS) as the primary chromatographic medium, coupled with a tailored solvent system designed to exploit hydrophobic interactions rather than just ionic ones. This method begins with the dissolution of the crude reaction mixture in a specific methanol-formic acid-water solution, which effectively solubilizes the target compound while precipitating certain insoluble impurities, setting the stage for high-resolution separation. The core of this novelty lies in the use of ODS media, which possesses a hydrophobic surface capable of engaging in specific steric and hydrophobic interactions with the unique lipophilic side chain of Telavancin. This allows for the precise resolution of the target molecule from structural analogues and dimers that conventional resins cannot differentiate, achieving a mass concentration of Telavancin greater than 95% in the collected fractions.
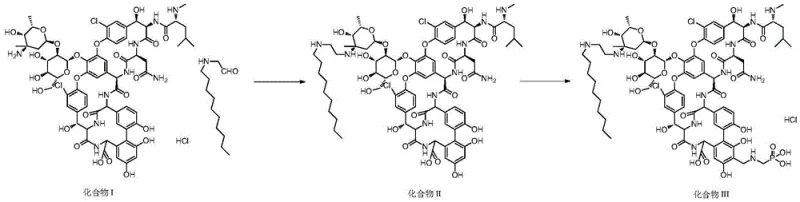
Following chromatography, the process utilizes a polyethersulfone nanofiltration membrane for concentration and desalting, a step that replaces energy-intensive freeze-drying and significantly enhances product stability. By maintaining the process at lower temperatures and controlling the pH environment through diafiltration with dilute hydrochloric acid, the method prevents the oxidative degradation that typically causes discoloration in glycopeptide antibiotics. This streamlined workflow not only simplifies the commercial scale-up of complex API intermediates but also ensures a more consistent and robust supply of the final drug substance. The combination of selective chromatography and gentle membrane filtration represents a holistic solution to the purity and stability challenges that have historically limited the efficiency of Telavancin production.
Mechanistic Insights into ODS Chromatographic Separation
The success of this purification method is deeply rooted in the specific molecular recognition mechanisms facilitated by the octadecyl-bonded silica gel stationary phase. Telavancin is structurally distinct from its precursor, vancomycin, due to the introduction of a hydrophobic decylaminoethyl side chain at the 4-position sugar amine and a hydrophilic phosphonic acid aminomethyl group at the 7-position aromatic ring. The ODS medium, with its long carbon chains, creates a hydrophobic environment that interacts strongly with the newly introduced lipophilic tail of the Telavancin molecule. This interaction is significantly stronger and more specific than the non-specific adsorption observed with conventional ion exchange resins, allowing the chromatography column to retain the target compound while allowing more polar impurities and unreacted starting materials to elute earlier or later depending on the gradient profile.
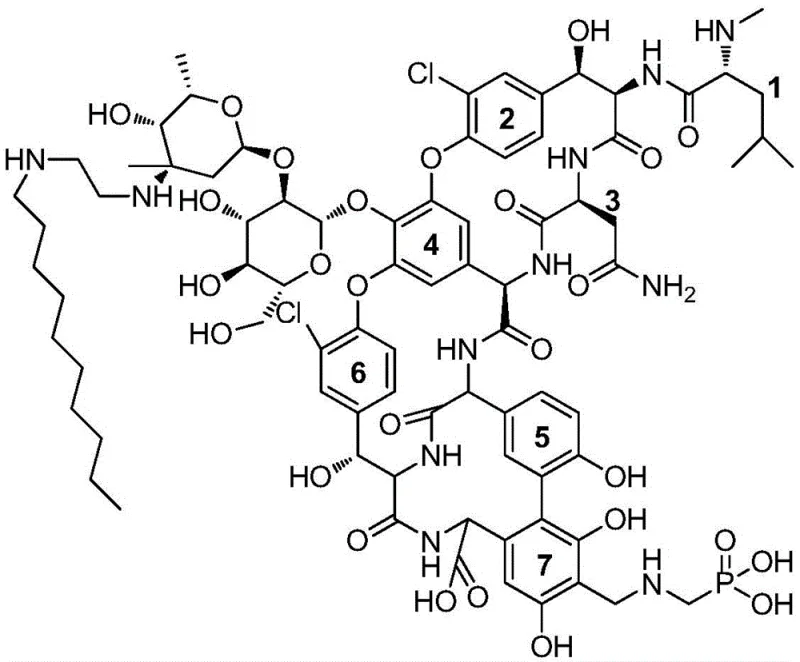
Furthermore, the steric hindrance provided by the bulky ODS ligands plays a crucial role in separating Telavancin from its dimeric impurities. As illustrated in the structural analysis, the spatial arrangement of the substituents on the glycopeptide backbone creates a unique three-dimensional shape that fits optimally within the interstitial spaces of the ODS matrix under specific solvent conditions. The use of a phosphate buffer system adjusted to a pH of 2.0 to 2.5 ensures that the amino groups are protonated while maintaining the integrity of the acid-labile glycosidic bonds. The gradient elution, shifting from low to high concentrations of organic solvents like acetonitrile or ethanol, finely tunes the polarity of the mobile phase to sequentially desorb impurities before eluting the highly pure Telavancin. This precise control over the thermodynamic equilibrium between the mobile and stationary phases is what enables the removal of trace impurities that are critical for meeting pharmacopoeial standards.
In addition to separation efficiency, the mechanism also addresses the chemical stability of the molecule during the process. The rapid passage through the column and the immediate subsequent concentration via nanofiltration minimize the residence time of the compound in solution, reducing the window of opportunity for hydrolytic or oxidative degradation. The nanofiltration membrane, with a molecular weight cutoff between 200 and 1000 daltons, effectively retains the large Telavancin molecule while allowing small buffer salts and solvent molecules to pass through. This diafiltration process not only concentrates the product but also exchanges the buffer system to a final acidic pH of 0.5 to 2, which is the optimal condition for the long-term storage stability of Telavancin hydrochloride. This mechanistic understanding underscores why this specific combination of unit operations is superior to traditional isolation techniques.
How to Synthesize Telavancin Efficiently
The synthesis and subsequent purification of Telavancin require a meticulously controlled sequence of chemical transformations followed by the advanced isolation protocol described in the patent. The process initiates with the reductive amination of vancomycin hydrochloride with a protected decylaminoacetaldehyde derivative, followed by deprotection and a Mannich reaction to introduce the phosphonic acid moiety. However, the true value lies in the downstream processing, where the crude reaction mixture is subjected to the specialized ODS chromatography and nanofiltration steps to ensure pharmaceutical grade quality. Implementing this route requires precise control over solvent ratios, pH levels, and flow rates to maximize yield and purity while minimizing the formation of degradation products. For manufacturing teams, adopting this standardized approach ensures reproducibility and compliance with Good Manufacturing Practices (GMP).
- Dissolve the crude telavancin reaction mixture in a methanol-formic acid-water solution (2: 1:1 volume ratio) at a weight ratio of 1:5 to 1:10, then filter to remove insoluble solids.
- Purify the filtrate using octadecyl-bonded silica gel (ODS) chromatography with a phosphate buffer and organic solvent gradient elution system to separate target compounds from dimers and impurities.
- Concentrate the purified eluate using a polyethersulfone nanofiltration membrane (MWCO 200-1000 Da) while performing diafiltration with pH 4-5 hydrochloric acid to remove buffer salts.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this advanced purification technology translates directly into enhanced operational efficiency and risk mitigation. The elimination of complex liquid-liquid extraction steps and the replacement of energy-intensive lyophilization with nanofiltration significantly streamline the production workflow. This simplification reduces the overall processing time and lowers the consumption of utilities and solvents, leading to a substantial reduction in the cost of goods sold (COGS). Furthermore, the improved selectivity of the ODS chromatography means that fewer batches are rejected due to failing purity specifications, thereby increasing the effective yield of the manufacturing campaign and ensuring a more reliable supply of raw materials for downstream formulation.
- Cost Reduction in Manufacturing: The transition from traditional ion exchange and extraction methods to this streamlined chromatographic process eliminates the need for multiple solvent recovery and waste treatment stages associated with liquid-liquid extractions. By utilizing a single, highly selective chromatographic step followed by membrane filtration, the process reduces the consumption of expensive chromatography resins and organic solvents over the lifecycle of the production facility. Additionally, the avoidance of freeze-drying in favor of nanofiltration drastically cuts energy costs, as thermal concentration methods are far more economical than sublimation. These cumulative efficiencies result in a leaner manufacturing process that is less capital intensive and more profitable, offering a competitive edge in the pricing of high-purity glycopeptide antibiotics.
- Enhanced Supply Chain Reliability: One of the most significant supply chain risks in antibiotic production is batch failure due to instability or impurity spikes. This purification method mitigates that risk by producing a chemically stable product that resists discoloration and degradation during storage. The robustness of the ODS chromatography ensures consistent removal of dimers and by-products, meaning that every batch meets the same high-quality standards without the need for rework. This consistency allows supply chain planners to forecast inventory with greater accuracy and reduces the safety stock requirements needed to buffer against quality failures. Consequently, partners can rely on a steady flow of compliant material, reducing lead time for high-purity glycopeptide antibiotics and ensuring uninterrupted availability for patients.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from laboratory development to commercial production without the need for fundamental process redesign. The use of standard chromatographic columns and modular nanofiltration units allows for flexible capacity expansion to meet market demand. From an environmental perspective, the reduction in solvent usage and the elimination of harsh extraction chemicals align with modern green chemistry principles and regulatory expectations for sustainable manufacturing. The wastewater generated is easier to treat due to the simplified solvent profile, reducing the environmental footprint of the facility. This alignment with sustainability goals not only ensures regulatory compliance but also enhances the corporate social responsibility profile of the manufacturing organization.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this purification technology. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages for potential partners and licensees. Understanding these details is crucial for evaluating the feasibility of integrating this method into existing production lines or for sourcing high-quality Telavancin from external suppliers.
Q: Why is octadecyl-bonded silica gel preferred over ion exchange resins for Telavancin purification?
A: Octadecyl-bonded silica gel (ODS) provides specific steric hindrance and stronger hydrophobic interactions with the lipophilic side chain of Telavancin, allowing for superior separation of structural analogues and dimers compared to conventional ion exchange methods.
Q: How does the nanofiltration step contribute to product stability?
A: The nanofiltration process allows for low-temperature concentration and efficient removal of buffer salts without the thermal stress associated with freeze-drying, thereby preventing the oxidation of phenolic groups that leads to product discoloration.
Q: What is the expected purity level achievable with this purification method?
A: By utilizing gradient elution with optimized phosphate buffers and organic solvents, this method consistently yields chromatographic solutions with a Telavancin mass concentration exceeding 95%, meeting stringent pharmaceutical standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Telavancin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of advanced purification technologies in delivering safe and effective antibiotic therapies to the global market. Our team of expert chemists and engineers possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex molecules like Telavancin are manufactured with the highest degree of precision and care. We are committed to maintaining stringent purity specifications through our rigorous QC labs, which are equipped with state-of-the-art analytical instrumentation to verify the identity and potency of every batch. Our capability to implement the ODS chromatography and nanofiltration protocols described in patent CN107629115B positions us as a leader in the production of next-generation glycopeptide antibiotics.
We invite pharmaceutical companies and contract manufacturers to collaborate with us to leverage these technological advancements for their supply chains. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that demonstrates how our optimized processes can reduce your overall procurement costs while enhancing product quality. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your project requirements. Together, we can ensure a secure and sustainable supply of high-purity Telavancin, supporting the global fight against resistant bacterial infections.
