Advanced Synthesis of Naproxen Nitroxyalkylesters: A Breakthrough in Chiral Stability and Commercial Scalability
The pharmaceutical industry continuously seeks robust synthetic routes that guarantee high stereochemical purity, particularly for non-steroidal anti-inflammatory drug (NSAID) derivatives. Patent CN1367773A introduces a transformative process for the preparation of naproxene nitroxyalkylesters, specifically targeting the 2-(S)-(6-methoxy-2-naphthyl)-propanoic acid scaffold. This innovation addresses a critical bottleneck in prior art by replacing traditional organic bases with inorganic mineral alkalis, thereby securing an enantiomeric excess (ee) greater than or equal to 98%. For R&D directors and procurement specialists, this shift represents a significant leap in process reliability, ensuring that the biologically active (S)-enantiomer is preserved throughout the esterification with aliphatic nitroxyalkanols. The method not only achieves superior optical purity compared to previous benchmarks but also delivers high chemical yields exceeding 85%, making it a cornerstone technology for the reliable pharmaceutical intermediate supplier market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this invention, the synthesis of nitroxyalkylesters of naproxen relied heavily on organic nitrogenous bases such as 4-dimethylaminopyridine (DMAP), morpholine, or triethylamine, as documented in earlier filings like WO 98/25,918. While these catalysts facilitate acylation, they suffer from severe limitations regarding stereochemical retention. Experimental data from the patent indicates that when using standard organic bases, the enantiomeric excess often drops precipitously, ranging from merely 56% to 80%. Even under optimized conditions with DMAP, the ee rarely surpasses 94%, which is insufficient for high-grade pharmaceutical applications requiring near-perfect chiral fidelity. Furthermore, the removal of these organic amines post-reaction often necessitates complex purification protocols, adding unnecessary cost and time to the manufacturing cycle while risking product degradation.
The Novel Approach
The novel approach detailed in CN1367773A fundamentally alters the reaction landscape by employing inorganic bases such as potassium carbonate, zinc oxide, or calcium aluminosilicate. This strategic substitution eliminates the nucleophilic interference associated with organic amines, allowing the reaction between the acid halide and the nitroxyalkanol to proceed with exceptional stereochemical control. By operating within a mild temperature range of 0°C to 20°C in inert solvents like methylene dichloride or toluene, the process consistently yields products with an ee of ≥98% and HPLC purity >99%. This method simplifies the downstream processing significantly, as the inorganic salts can be easily removed via filtration, leaving a crude product that requires minimal purification to meet stringent quality standards for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Inorganic Base Catalyzed Esterification
The core mechanistic advantage of this process lies in the non-nucleophilic nature of the inorganic base, which acts primarily as a proton scavenger rather than a catalytic nucleophile. In the reaction between 2-(S)-(6-methoxy-2-naphthyl)-propionyl chloride and aliphatic nitroxyalkanol, the preservation of the chiral center at the alpha-position is paramount. Organic bases can sometimes form reactive acyl-ammonium intermediates that are prone to racemization via enolization, especially under prolonged reaction times or elevated temperatures. In contrast, the inorganic base facilitates the deprotonation of the alcohol moiety gently, promoting a direct nucleophilic attack on the acid chloride without forming unstable chiral intermediates. This ensures that the configuration of the starting material, which typically possesses an ee of 98%, is transferred quantitatively to the final ester product without erosion.
Furthermore, the structural versatility of the aliphatic nitroxyalkanol component allows for the creation of diverse prodrug variants. The patent defines the 'Y' group in the nitroxyalkanol HO-Y-ONO2 as potentially containing linear or branched alkylene chains, cycloalkylene rings, or even aromatic moieties. 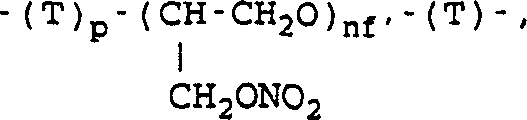 As illustrated in the structural representation, the nitrate ester functionality (CH2ONO2) is crucial for the NO-donating properties of the final molecule, which contributes to its gastrointestinal safety profile compared to traditional NSAIDs. The robustness of the inorganic base method ensures that even with complex 'Y' groups, the esterification proceeds efficiently, maintaining the integrity of the sensitive nitrate group which might otherwise decompose under harsher basic conditions.
As illustrated in the structural representation, the nitrate ester functionality (CH2ONO2) is crucial for the NO-donating properties of the final molecule, which contributes to its gastrointestinal safety profile compared to traditional NSAIDs. The robustness of the inorganic base method ensures that even with complex 'Y' groups, the esterification proceeds efficiently, maintaining the integrity of the sensitive nitrate group which might otherwise decompose under harsher basic conditions.
How to Synthesize Naproxen Nitroxyalkylesters Efficiently
To implement this synthesis effectively, operators must adhere to strict stoichiometric controls and temperature management. The process begins with the suspension of the chosen inorganic base in an inert solvent, followed by the addition of the nitroxyalkanol. The acid chloride is then added dropwise to maintain the exotherm within the optimal 10°C to 20°C window. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by dissolving aliphatic nitroxyalkanol in an inert organic solvent such as methylene dichloride or toluene.
- Add a stoichiometric amount of inorganic base, such as potassium carbonate or calcium aluminosilicate, to the mixture under agitation.
- Introduce the 2-(S)-(6-methoxy-2-naphthyl)-propionyl chloride solution slowly while maintaining temperature between 0°C and 20°C to preserve stereochemistry.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this inorganic base protocol offers substantial operational benefits beyond mere chemical yield. The elimination of expensive and potentially toxic organic catalysts like DMAP directly translates to raw material cost optimization. Additionally, the simplified workup procedure—often requiring only filtration and solvent evaporation—reduces the consumption of purification media such as silica gel and decreases the overall cycle time per batch. This efficiency is critical for maintaining competitive pricing in the high-purity pharmaceutical intermediate sector while ensuring consistent supply continuity.
- Cost Reduction in Manufacturing: The replacement of proprietary or high-cost organic amines with commodity inorganic salts like potassium carbonate drastically lowers the bill of materials. Since these inorganic bases are globally available and inexpensive, the volatility of raw material pricing is significantly mitigated. Moreover, the high yield (>85%) and high purity (>99%) reduce the loss of valuable chiral starting materials, ensuring that every kilogram of input generates maximum output value without the need for costly recycling of racemic mixtures.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which tolerate a broad temperature range from -20°C to 50°C, makes the process less susceptible to minor equipment fluctuations or environmental variations. This tolerance enhances batch-to-batch consistency, a key metric for supply chain reliability. By avoiding sensitive catalytic systems that require strict anhydrous conditions or specialized handling, the manufacturing process becomes more resilient, reducing the risk of production delays caused by failed batches or complex troubleshooting.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the heterogeneous nature of the inorganic base reaction. The ease of filtration allows for efficient separation of solids on a large scale, avoiding the emulsion issues often encountered with organic base extractions. Furthermore, the reduction in organic waste streams associated with amine removal aligns with increasingly stringent environmental regulations, facilitating smoother regulatory approvals and reducing the carbon footprint of the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this patented synthesis route. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity for technical teams evaluating this technology for adoption.
Q: Why does the inorganic base method yield higher enantiomeric excess than organic bases?
A: According to patent CN1367773A, organic bases like DMAP or triethylamine can induce racemization or fail to maintain the chiral integrity of the S-enantiomer during acylation, resulting in ee values below 94%. In contrast, inorganic bases provide a milder, non-nucleophilic environment that preserves the stereocenter, consistently achieving ee values ≥98%.
Q: What are the preferred solvents for this nitroxyalkylester synthesis?
A: The patent specifies inert organic solvents including aromatic hydrocarbons like toluene, chlorinated solvents such as methylene dichloride, and aliphatic esters like ethyl acetate. These solvents ensure optimal solubility of both the acid halide and the nitroxyalkanol while facilitating easy product isolation.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process is highly scalable due to the use of inexpensive, commercially available inorganic bases and mild reaction temperatures (-20°C to 50°C). The workup involves simple filtration and solvent evaporation, avoiding complex purification steps required by organic base methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Naproxen Nitroxyalkylesters Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of chiral purity and process efficiency in the development of next-generation NSAID prodrugs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to full-scale manufacturing is seamless. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced HPLC and chiral chromatography to verify enantiomeric excess and impurity profiles.
We invite global partners to collaborate with us to leverage this advanced synthetic technology for their pipeline projects. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized processes can enhance your supply chain efficiency and product quality.
