Advanced Manufacturing of Naratriptan Impurity B: A Strategic Breakthrough for Pharmaceutical Quality Control
Advanced Manufacturing of Naratriptan Impurity B: A Strategic Breakthrough for Pharmaceutical Quality Control
The pharmaceutical industry faces relentless pressure to ensure the highest standards of drug safety and efficacy, particularly for complex small molecules like triptans used in migraine treatment. Patent CN103554081A introduces a transformative preparation method for Naratriptan Impurity B, a critical reference standard and process contaminant that must be strictly monitored according to United States Pharmacopeia (USP) guidelines. This innovation addresses the longstanding challenges associated with synthesizing this specific indole derivative, offering a pathway that is not only chemically robust but also commercially viable for large-scale operations. By shifting away from hazardous high-pressure conditions and expensive noble metal catalysts, this technology provides a reliable pharmaceutical intermediates supplier with a distinct competitive edge in the global market. The method leverages a sophisticated yet operationally simple two-step sequence involving catalytic hydrogenation and subsequent condensation, ensuring that manufacturers can achieve superior purity profiles while minimizing environmental impact and production costs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Naratriptan and its associated impurities has been fraught with technical complexities that hinder efficient commercial scale-up of complex pharmaceutical intermediates. Prior art, such as the methodologies disclosed in patent WO95/09166, often relied on the use of homogeneous noble metal catalysts like palladium acetate or palladium oxide, which necessitate rigorous removal steps to prevent heavy metal contamination in the final active pharmaceutical ingredient. Furthermore, alternative routes described in documents like CN200410093115.6 frequently required high-pressure catalytic hydrogenation, demanding specialized reactor infrastructure that significantly increases capital expenditure and operational risk. These conventional approaches often suffered from prolonged reaction times, difficult downstream processing, and the generation of multiple by-products that complicated purification efforts. For procurement managers and supply chain heads, these factors translated into higher costs, longer lead times, and potential bottlenecks in securing high-purity materials essential for regulatory compliance and quality control laboratories worldwide.
The Novel Approach
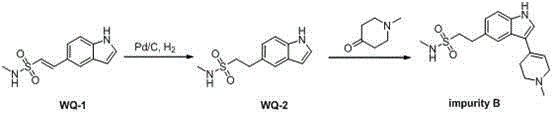
In stark contrast to these legacy techniques, the novel approach detailed in CN103554081A utilizes a streamlined strategy that dramatically simplifies the manufacturing landscape. The core innovation lies in the use of atmospheric pressure hydrogenation coupled with a heterogeneous palladium on carbon (Pd/C) catalyst, which eliminates the need for expensive high-pressure vessels and facilitates easy catalyst recovery through simple filtration. This method starts with (E)-N-methyl-2-(1H-indole-5) vinyl sulfonamide as a readily available starting material, subjecting it to mild reduction conditions that preserve the integrity of the sensitive indole ring system. Following reduction, the intermediate undergoes a condensation reaction with N-methylpiperidone under alkaline reflux conditions, a process that is both rapid and highly selective. This strategic shift not only reduces the reaction time significantly but also ensures a cleaner reaction profile with fewer side products, thereby enhancing the overall yield and purity of the target Naratriptan Impurity B without compromising on safety or environmental standards.
Mechanistic Insights into Pd/C-Catalyzed Hydrogenation and Condensation
The chemical elegance of this synthesis lies in its precise control over reaction parameters to maximize efficiency and minimize impurity formation. The first stage involves the catalytic hydrogenation of the vinyl sulfonamide double bond, where the Pd/C catalyst acts as a surface for hydrogen adsorption and activation. Unlike homogeneous catalysts that can be difficult to separate, the heterogeneous nature of Pd/C allows for complete removal via filtration, preventing any residual palladium from carrying over into subsequent steps or the final product. The choice of solvent plays a pivotal role in this mechanism; while methanol alone is effective, the patent highlights that mixed solvent systems, particularly methanol combined with acetic acid, can further accelerate the reaction rate and improve conversion efficiency at ambient temperatures ranging from 25°C to 40°C. This mild thermal requirement is crucial for preventing thermal degradation of the indole scaffold, ensuring that the structural fidelity of the molecule is maintained throughout the reduction process.
Following the successful reduction to the ethyl sulfonamide intermediate (WQ-2), the process transitions to a base-mediated condensation reaction. In this step, the intermediate reacts with N-methylpiperidone in an ethanol solvent system under the influence of potassium hydroxide (KOH). The alkaline conditions facilitate the formation of a carbanion or enolate species that attacks the electrophilic center, forging the critical carbon-carbon bond that links the piperidine ring to the indole core. This condensation is conducted under reflux for approximately five hours, a duration optimized to drive the reaction to completion while avoiding excessive thermal stress. The result is a highly pure final product, Naratriptan Impurity B, which is isolated as an off-white solid after simple aqueous workup and washing. This mechanistic pathway effectively mitigates the formation of regioisomers or over-alkylated by-products, providing a robust solution for producing reference standards needed for rigorous analytical validation.
How to Synthesize Naratriptan Impurity B Efficiently
Implementing this synthesis protocol requires careful attention to solvent selection and catalyst loading to replicate the high yields reported in the patent data. The process begins with the dissolution of the vinyl sulfonamide starting material in a suitable alcohol solvent, followed by the addition of the Pd/C catalyst and the introduction of hydrogen gas at atmospheric pressure. Operators must monitor the reaction progress, typically via TLC or HPLC, to ensure complete consumption of the starting olefin before proceeding to the workup phase. Once the intermediate is isolated, it is immediately subjected to the condensation conditions with N-methylpiperidone and KOH in ethanol. The detailed standardized synthesis steps, including specific molar ratios, temperature controls, and isolation procedures, are outlined below to ensure reproducibility and safety in your laboratory or pilot plant settings.
- Perform catalytic hydrogenation of (E)-N-methyl-2-(1H-indole-5) vinyl sulfonamide using Pd/C under atmospheric pressure in methanol or mixed solvents.
- Filter the reaction mixture to remove the catalyst and concentrate the filtrate to obtain the reduced intermediate WQ-2.
- Condense the intermediate with N-methylpiperidone in ethanol under reflux with KOH to yield the final Naratriptan Impurity B.
Commercial Advantages for Procurement and Supply Chain Teams
For decision-makers focused on the bottom line and operational continuity, this patented method offers substantial commercial advantages that directly address key pain points in pharmaceutical manufacturing. By eliminating the need for high-pressure reactors and expensive homogeneous catalysts, the process drastically lowers the barrier to entry for production, allowing for more flexible and cost-effective manufacturing strategies. The use of commodity chemicals such as methanol, ethanol, and acetic acid ensures that raw material sourcing is stable and unaffected by the volatility often seen with specialized reagents. Furthermore, the simplified workup procedure, which relies on filtration and evaporation rather than complex chromatographic separations, significantly reduces solvent consumption and waste generation, aligning with modern green chemistry principles and reducing disposal costs. These factors combine to create a supply chain that is not only more resilient but also capable of delivering high-quality materials with greater speed and reliability.
- Cost Reduction in Manufacturing: The transition to atmospheric pressure hydrogenation removes the necessity for costly high-pressure autoclaves and the associated safety maintenance protocols, leading to significant capital expenditure savings. Additionally, the use of recoverable heterogeneous catalysts like Pd/C minimizes the loss of precious metals, while the high selectivity of the reaction reduces the need for expensive purification steps such as preparative HPLC. This holistic reduction in operational complexity translates directly into a lower cost of goods sold (COGS), enabling competitive pricing for high-purity pharmaceutical intermediates without sacrificing margin.
- Enhanced Supply Chain Reliability: Dependence on specialized equipment or rare catalysts often creates single points of failure in a supply chain; however, this method utilizes widely available reagents and standard glass-lined or stainless-steel reactors found in most multipurpose facilities. The robustness of the reaction conditions means that production schedules are less likely to be disrupted by equipment downtime or reagent shortages. Consequently, suppliers can offer more consistent lead times and guarantee continuous availability of Naratriptan Impurity B, which is critical for pharmaceutical companies managing tight regulatory submission timelines and quality control inventories.
- Scalability and Environmental Compliance: The simplicity of the reaction design facilitates seamless scale-up from laboratory grams to multi-ton commercial production without the need for re-engineering the core process. The reduced solvent load and the absence of toxic heavy metal residues in the final product simplify wastewater treatment and environmental compliance reporting. This scalability ensures that as demand for Naratriptan and its related quality control standards grows, the manufacturing capacity can be expanded rapidly to meet market needs while adhering to increasingly stringent global environmental regulations regarding chemical discharge and worker safety.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its adoption for their specific applications. The following questions address common inquiries regarding the practical implementation, quality attributes, and regulatory implications of this manufacturing route. These answers are derived directly from the experimental data and technical specifications provided in the patent literature, ensuring that the information is accurate and relevant for technical due diligence processes.
Q: What are the critical advantages of this synthesis method over prior art?
A: Unlike previous methods requiring high-pressure equipment or noble metal catalysts like Pd(OAc)2, this novel approach utilizes atmospheric pressure hydrogenation with Pd/C, significantly reducing equipment costs and operational hazards while maintaining high yields.
Q: How is purity controlled during the manufacturing of Naratriptan Impurity B?
A: The process minimizes by-product formation through optimized solvent systems (such as methanol/acetic acid mixtures) and straightforward workup procedures involving simple filtration and recrystallization, ensuring the final product meets stringent USP quality standards.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the use of common solvents like ethanol and methanol, combined with atmospheric pressure conditions and robust heterogeneous catalysis, makes this route highly scalable and ideal for industrial manufacturing without specialized high-pressure reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Naratriptan Impurity B Supplier
At NINGBO INNO PHARMCHEM, we recognize that the integrity of your final drug product depends on the quality of every component in your supply chain, including critical impurities and reference standards. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in the early stages of method development or full-scale commercial manufacturing. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the identity and purity of every batch of Naratriptan Impurity B we produce. Our commitment to excellence ensures that you receive materials that are fully compliant with international pharmacopeial standards, supporting your regulatory filings and quality assurance programs with confidence.
We invite you to collaborate with us to optimize your supply chain for Naratriptan-related projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs and logistical constraints. Please contact us today to request specific COA data, discuss route feasibility assessments, or inquire about our current inventory levels. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable pharmaceutical intermediates supplier dedicated to driving innovation and efficiency in your drug development pipeline.
