Scalable Synthesis of Rosuvastatin Tert-Butyl Ester via Modified Julia Olefination for Global Pharma Supply Chains
Introduction to Advanced Statin Intermediate Manufacturing
The pharmaceutical industry continuously seeks robust, scalable, and environmentally sustainable pathways for producing high-value Active Pharmaceutical Ingredients (APIs) and their precursors. Patent CN110483412B introduces a groundbreaking synthetic methodology for Rosuvastatin tert-butyl ester, a critical intermediate in the production of the blockbuster cardiovascular drug Rosuvastatin Calcium. This innovation addresses long-standing inefficiencies in statin manufacturing by replacing hazardous, low-yield processes with a streamlined, high-selectivity approach. By leveraging a modified Julia olefination strategy, the technology ensures superior stereochemical control while operating under significantly milder conditions than legacy methods. For global supply chain leaders, this represents a pivotal shift towards more reliable and cost-effective pharmaceutical intermediate sourcing, minimizing the risks associated with complex cryogenic operations and difficult purification steps.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Rosuvastatin tert-butyl ester has relied heavily on the Wittig reaction, as depicted in early synthetic routes like US20050124639A1. While the raw materials for the Wittig approach are accessible, the reaction inherently suffers from poor stereoselectivity, typically generating a mixture containing approximately 25% of the undesired cis-isomer. This necessitates rigorous and wasteful purification protocols to isolate the pharmacologically active trans-isomer, resulting in an overall yield of only about 60% after separation. Furthermore, the stoichiometric generation of triphenylphosphine oxide byproducts creates substantial solid waste disposal challenges, inflating both material costs and environmental compliance burdens. Alternative Julia olefination routes attempted to solve the isomer issue but introduced new bottlenecks, such as the requirement for ultra-low temperatures (-60°C) and incomplete oxidation of side chains, rendering them economically unviable for large-scale commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
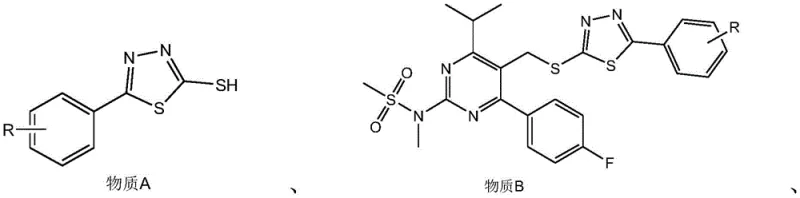
The methodology disclosed in CN110483412B fundamentally reengineers the carbon-carbon bond formation step to overcome these historical barriers. Instead of phosphonium salts or unstable tetrazole sulfones, this route utilizes a stable 2-mercapto-5-phenyl-1,3,4-thiadiazole derivative (Substance A) which couples efficiently with the pyrimidine core. The subsequent oxidation to the sulfone (Substance C) is achieved using a green catalytic system involving hydrogen peroxide and molybdic acid or sodium tungstate, ensuring complete conversion without harsh reagents. Crucially, the final olefination with the chiral aldehyde proceeds with exceptional trans-selectivity, virtually eliminating the formation of cis-isomers that plague the Wittig process. This results in a much shorter, cleaner synthetic sequence that operates at ambient or mildly elevated temperatures, offering a distinct advantage for cost reduction in pharmaceutical intermediate manufacturing by simplifying reactor requirements and waste treatment.
Mechanistic Insights into Modified Julia-Kocienski Olefination
The core chemical innovation lies in the precise execution of the Julia-Kocienski type olefination, which dictates the stereochemical outcome of the statin side chain. The process begins with the nucleophilic substitution of the bromomethyl pyrimidine by the thiolate anion of the thiadiazole, forming the sulfide linkage in Substance B. This intermediate is then carefully oxidized to the corresponding sulfone (Substance C), a critical activation step that increases the acidity of the alpha-protons adjacent to the sulfur atom. Upon treatment with a strong base such as sodium tert-butoxide, the sulfone forms a stabilized carbanion which attacks the carbonyl carbon of the chiral dioxane aldehyde. Unlike traditional Julia olefinations that require reductive elimination, this heteroaryl sulfone variant undergoes a spontaneous syn-elimination of the sulfinate group, driven by the electronic properties of the thiadiazole ring. This mechanism inherently favors the formation of the thermodynamically stable E-alkene (trans-isomer), thereby solving the stereocontrol issue at the molecular level without needing chiral catalysts or extensive chromatography.
Impurity control is another pillar of this mechanistic design, particularly regarding the oxidation state of the sulfur and the integrity of the chiral centers. The use of a catalytic amount of molybdic acid tetrahydrate or sodium tungstate with hydrogen peroxide allows for a controlled, selective oxidation that prevents over-oxidation or degradation of the sensitive pyrimidine ring. Furthermore, the reaction conditions for the coupling step are maintained between -20°C and 40°C, a range that is wide enough to ensure kinetic control over the addition step while preventing epimerization of the chiral centers on the dioxane ring. This tight control over reaction parameters ensures that the final product, Rosuvastatin tert-butyl ester, exhibits extremely low levels of diastereomers and non-enantiomeric impurities, meeting the stringent purity specifications required for downstream API synthesis and regulatory approval.
How to Synthesize Rosuvastatin Tert-Butyl Ester Efficiently
Implementing this synthesis requires strict adherence to the optimized reaction parameters defined in the patent to maximize yield and stereochemical purity. The process is divided into four distinct operational stages: the initial coupling to form the sulfide, the catalytic oxidation to the sulfone, the base-mediated olefination with the chiral aldehyde, and the final acidic deprotection. Each step utilizes common industrial solvents like acetone, ethanol, and tetrahydrofuran, avoiding exotic or highly toxic reagents. The following guide outlines the standardized workflow derived from the patent examples, designed for seamless translation from laboratory bench to pilot plant.
- React 5-bromomethyl-4-(4-fluorophenyl)-6-isopropyl-2-[methyl(methylsulfonyl)amino]pyrimidine with 2-mercapto-5-phenyl-1,3,4-thiadiazole using a base catalyst to form the sulfide intermediate.
- Oxidize the sulfide intermediate using hydrogen peroxide and a molybdate or tungstate catalyst to generate the corresponding sulfone species.
- Perform Julia olefination by reacting the sulfone with (4R-Cis)-6-aldehyde-2,2-dimethyl-1,3-dioxane-4-acetic acid tert-butyl ester under basic conditions.
- Execute acid-catalyzed deprotection using dilute hydrochloric acid to remove the acetonide protecting group and yield the final Rosuvastatin tert-butyl ester.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible strategic benefits beyond mere technical elegance. By shifting away from the Wittig reaction, manufacturers can eliminate the procurement and disposal costs associated with tons of triphenylphosphine oxide waste, directly impacting the bottom line through reduced waste treatment fees and raw material consumption. The transition to a catalytic oxidation system using hydrogen peroxide further aligns with green chemistry principles, reducing the environmental footprint and simplifying regulatory compliance for facility operations. Additionally, the reliance on readily available starting materials, such as substituted thiadiazoles and standard pyrimidine derivatives, mitigates supply chain risks associated with specialized or monopolized reagents, ensuring a more resilient and continuous supply of this critical high-purity pharmaceutical intermediate.
- Cost Reduction in Manufacturing: The elimination of cryogenic equipment requirements is a major driver for operational expenditure (OpEx) savings. Traditional routes necessitating -60°C reactions demand specialized refrigeration infrastructure and high energy inputs to maintain such low temperatures over long reaction times. In contrast, this novel process operates effectively between 0°C and 50°C, allowing the use of standard jacketed reactors and conventional cooling water systems. This drastic simplification of thermal management translates to significantly lower utility costs and reduced capital expenditure (CapEx) for new production lines, making the manufacturing process far more economically attractive for high-volume production.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions directly correlates to supply continuity. Processes that operate at extreme temperatures or require sensitive reagents are prone to batch failures and delays due to equipment malfunction or reagent instability. By utilizing stable intermediates and mild conditions, this method reduces the probability of batch rejection and ensures consistent cycle times. The high stereoselectivity also means that less time is spent on recycling off-spec material or performing complex recrystallizations, leading to faster throughput and more predictable delivery schedules for downstream API manufacturers who rely on just-in-time inventory models.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental hazards, but this route is inherently designed for scalability. The exothermic profiles of the reactions are manageable, and the use of aqueous workups and common organic solvents facilitates efficient solvent recovery and recycling. The reduction in heavy metal usage (avoiding certain oxidation catalysts used in other routes) and phosphorus waste simplifies wastewater treatment protocols. This alignment with environmental, social, and governance (ESG) goals not only reduces compliance costs but also enhances the marketability of the final API to global pharmaceutical partners who prioritize sustainable supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on performance metrics and operational feasibility. Understanding these details is crucial for technical teams evaluating the integration of this route into existing manufacturing portfolios.
Q: How does this synthesis method improve stereoselectivity compared to the Wittig reaction?
A: Unlike the traditional Wittig reaction which generates approximately 25% cis-isomers requiring difficult purification, this modified Julia olefination route achieves exceptional trans-selectivity, virtually eliminating cis-byproducts and significantly simplifying downstream purification.
Q: What are the operational temperature advantages of this new route?
A: Previous methods required cryogenic conditions down to -60°C for the olefination step, demanding specialized equipment and high energy consumption. This novel process operates under mild conditions ranging from 0°C to 50°C, drastically reducing energy costs and facilitating easier industrial scale-up.
Q: Does this method address the oxidation challenges found in earlier Julia olefination routes?
A: Yes, earlier routes struggled with incomplete side-chain oxidation. This method utilizes a specific catalytic oxidation system with hydrogen peroxide and molybdic acid or sodium tungstate, ensuring complete conversion to the sulfone intermediate with high atom economy and minimal waste.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rosuvastatin Tert-Butyl Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a GMP-compliant environment. Our facilities are equipped with state-of-the-art reactor systems capable of handling the specific thermal and mixing requirements of the Julia olefination process, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest international standards for cardiovascular drug intermediates.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this superior synthesis technology for their Rosuvastatin supply chains. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical needs. We encourage you to contact us today to request specific COA data, route feasibility assessments, and comprehensive project timelines that demonstrate how we can drive efficiency and reliability in your API manufacturing operations.
