Advanced Synthesis of Fused Ring Indole Compounds for High-Performance OLED Applications
Introduction to Next-Generation Fused Indole Architectures
The rapid evolution of the organic electronics sector demands materials with superior optical properties and structural stability. Patent CN115124535A introduces a groundbreaking methodology for the synthesis of fused ring indole compounds, specifically targeting high-performance applications in Organic Light-Emitting Diodes (OLEDs) and advanced pharmaceutical intermediates. This technology addresses the critical need for efficient construction of complex polycyclic indole skeletons, which are notoriously difficult to synthesize with high regioselectivity using conventional organic transformations. The core innovation lies in the generation of compounds defined by general formula (I) and their subsequent oxidative cyclization into formula (II), creating rigid, planar structures essential for charge transport and fluorescence.
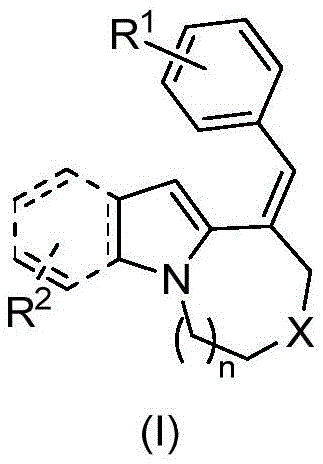
The structural versatility of these molecules is defined by the substituents R1 and R2, which can be tuned from simple hydrogen atoms to alkyl, alkoxy, halogen, amino, or acetyl groups, allowing for precise modulation of electronic properties. Furthermore, the linker X offers flexibility, being selectable from oxygen, nitrogen-benzenesulfonyl, or methylene bridges. This modularity is crucial for a reliable OLED material supplier aiming to customize emission wavelengths and energy levels for specific device architectures. The ability to systematically vary these positions while maintaining the integrity of the fused core represents a significant leap forward in material design capabilities.
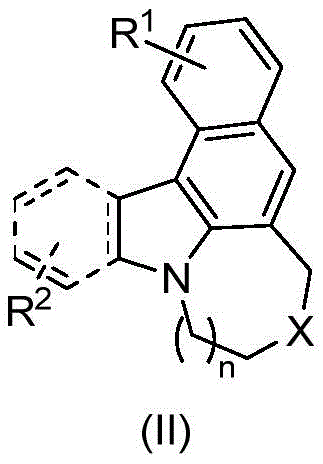
From a supply chain perspective, the availability of such tunable scaffolds reduces the lead time for high-purity electronic chemicals development. Traditional routes often require multi-step protections and deprotections that inflate costs and extend timelines. By contrast, the pathway disclosed in this patent streamlines the assembly of these complex cores, offering a direct route to materials that exhibit strong fluorescence absorption, particularly in the 360-500nm range. This spectral profile is highly desirable for blue and green emissive layers in display technologies, positioning this chemistry as a key enabler for next-generation optoelectronic devices.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of fused indole polycyclic systems has relied heavily on Fischer indole synthesis or transition-metal mediated cross-couplings that often suffer from poor atom economy and harsh reaction conditions. Conventional methods frequently require stoichiometric amounts of toxic heavy metals or strong acids, leading to significant waste generation and challenging purification processes. Moreover, achieving the specific 7-exo-dig cyclization required to form these fused rings is kinetically disfavored in many standard protocols, often resulting in mixtures of regioisomers that are difficult to separate. This lack of site selectivity not only lowers the overall yield but also introduces impurities that can act as quenchers in OLED applications, severely degrading device efficiency and lifetime.
The Novel Approach
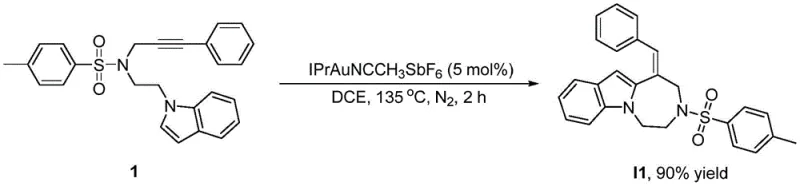
The methodology presented in CN115124535A overcomes these hurdles through a sophisticated dual-catalytic strategy involving Palladium/Copper coupling followed by Gold(I) catalysis. This novel approach fundamentally changes the skeleton structure of the raw material molecules, driving the formation of the fused ring system with high fidelity. The process initiates with the assembly of an alkyne-tethered indole precursor, which then undergoes a gold-catalyzed activation of the carbon-carbon triple bond. This activation lowers the energy barrier for the nucleophilic attack by the indole nitrogen or carbon, facilitating the critical ring-closing step.
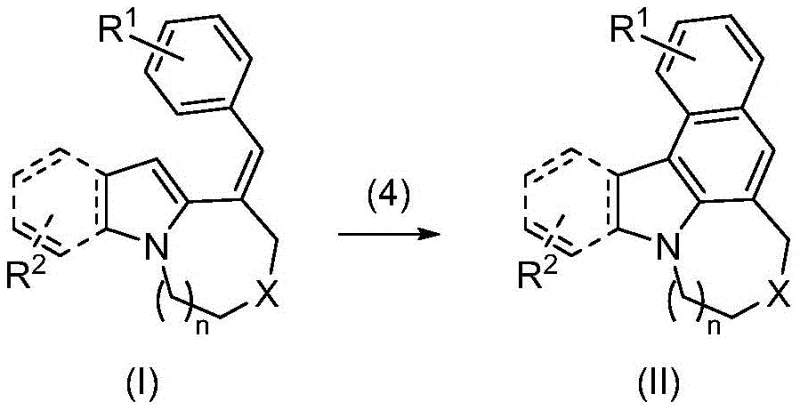
Following the initial cyclization to form compound (I), the process employs a mild oxidative cyclization step using [Hydroxy(tosyloxy)iodo]benzene (PhI(OH)OTs). This hypervalent iodine reagent acts as a powerful yet selective oxidant, promoting the final ring closure to yield the fully fused system (II) without degrading sensitive functional groups. This two-stage sequence ensures cost reduction in electronic chemical manufacturing by minimizing side reactions and maximizing the conversion of starting materials into the desired high-value product. The result is a robust synthetic platform capable of producing diverse derivatives simply by altering the initial aryl iodide or indole starting materials.
Mechanistic Insights into Au(I)-Catalyzed 7-exo-dig Cyclization
The heart of this synthetic breakthrough is the Gold(I)-catalyzed activation of the alkyne moiety. In this mechanism, the cationic Gold(I) species, such as [(IPr)Au(CH3CN)][SbF6], coordinates to the pi-system of the carbon-carbon triple bond. This coordination significantly increases the electrophilicity of the alkyne carbons, rendering them susceptible to nucleophilic attack by the proximal indole ring. The reaction proceeds through a 7-exo-dig cyclization pathway, which is typically challenging due to entropic factors, but the gold catalyst effectively templates this transition state. Following the cyclization, a proton transfer and protodeauration step release the gold catalyst back into the cycle, generating the exocyclic double bond characteristic of compound (I).
Impurity control is inherently built into this mechanism due to the high specificity of the gold-alkyne interaction. Unlike radical-based cyclizations that can lead to random polymerization or oligomerization, the coordination chemistry of gold ensures that the reaction occurs exclusively at the activated triple bond. Subsequent oxidative cyclization with PhI(OH)OTs further refines the molecular architecture. This step likely involves the formation of an iodonium intermediate or a radical cation species that facilitates the final C-C or C-N bond formation to close the second ring. The use of tetrahydrofuran (THF) as a solvent in this step provides a polar environment that stabilizes charged intermediates while remaining inert to the oxidant, ensuring a clean reaction profile suitable for stringent purity specifications required in the semiconductor industry.
How to Synthesize Fused Ring Indole Compounds Efficiently
The practical implementation of this technology involves a streamlined workflow that balances reaction efficiency with operational simplicity. The process begins with the preparation of the alkyne-functionalized indole intermediate, typically achieved via Sonogashira coupling or Mitsunobu reaction depending on the nature of the linker X. Once the precursor is secured, the gold-catalyzed step is performed in 1,2-dichloroethane (DCE) at elevated temperatures (135°C) to drive the cyclization to completion. The final oxidative step is conducted at room temperature, highlighting the mildness of the overall protocol. For detailed standardized operating procedures and safety guidelines regarding the handling of gold catalysts and hypervalent iodine reagents, please refer to the technical documentation below.
- Prepare the precursor compound of formula (I) by reacting an alkyne-substituted indole derivative with an aryl iodide using a Palladium/Copper co-catalytic system, followed by Gold(I)-catalyzed cyclization at 135°C in DCE solvent.
- Perform oxidative cyclization on the formula (I) intermediate using [Hydroxy(tosyloxy)iodo]benzene (PhI(OH)OTs) as the oxidant in THF solvent at room temperature to obtain the final fused ring compound of formula (II).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits beyond mere technical performance. The reliance on well-established catalytic systems (Pd/Cu for coupling, Au for cyclization) means that the supply chain for raw materials is robust and less prone to disruption compared to exotic reagent-dependent processes. Furthermore, the high selectivity of the reaction minimizes the formation of difficult-to-remove byproducts, which directly translates to simplified downstream processing and reduced solvent consumption during purification.
- Cost Reduction in Manufacturing: The elimination of multiple protection-deprotection sequences and the use of catalytic rather than stoichiometric amounts of expensive metals significantly lower the bill of materials. Although gold is a precious metal, its usage at low loadings (5 mol%) combined with high turnover numbers ensures that the cost per kilogram of the final API intermediate or OLED material remains competitive. The high yields reported in the patent examples further amplify these savings by reducing waste disposal costs and maximizing throughput per batch.
- Enhanced Supply Chain Reliability: The starting materials, including substituted indoles and aryl iodides, are commodity chemicals available from multiple global suppliers. This diversification of the supply base mitigates the risk of single-source dependency. Additionally, the reaction conditions (e.g., 50-60°C for coupling, 135°C for cyclization) are compatible with standard glass-lined or stainless steel reactors found in most fine chemical manufacturing facilities, removing the need for specialized high-pressure or cryogenic equipment.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability potential, moving seamlessly from gram-scale laboratory optimization to multi-kilogram pilot runs. The use of THF and DCE, while requiring proper recovery systems, is standard in the industry, and the solid byproducts generated are minimal. This aligns with modern green chemistry principles by improving atom economy and reducing the E-factor (mass of waste per mass of product), thereby simplifying environmental compliance and permitting for commercial scale-up of complex electronic chemicals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these fused indole derivatives. These answers are derived directly from the experimental data and scope defined within the patent literature, providing a transparent view of the technology's capabilities and limitations for potential partners and licensees.
Q: What are the primary applications of the fused ring indole compounds described in CN115124535A?
A: These compounds exhibit strong fluorescence properties with quantum yields suitable for Organic Light-Emitting Diode (OLED) materials. Additionally, due to the indole scaffold, they hold significant potential as pharmaceutical intermediates for bioactive alkaloids.
Q: How does the Gold(I) catalytic system improve the synthesis compared to traditional methods?
A: The Au(I) catalyst enables highly selective 7-exo-dig cyclization under relatively mild thermal conditions (135°C), drastically reducing side reactions and improving the overall yield of the complex polycyclic skeleton compared to non-catalytic or harsh acid-mediated routes.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the process utilizes commercially available reagents such as triethylamine, THF, and standard transition metal catalysts (Pd, Cu, Au) at low loadings (e.g., 5 mol% for Gold), making it feasible for scale-up from laboratory to commercial tonnage.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fused Ring Indole Supplier
As the demand for high-performance organic semiconductors and bioactive scaffolds continues to surge, securing a manufacturing partner with deep technical expertise is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging advanced catalytic methodologies like the one described in CN115124535A to deliver superior results. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from R&D to market is seamless. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch meets the exacting standards required for OLED fabrication and pharmaceutical development.
We invite you to collaborate with us to unlock the full potential of these fused ring indole compounds. Whether you require custom synthesis for a specific OLED emitter or large-scale production of a pharmaceutical intermediate, our technical procurement team is ready to assist. Please contact us today to request a Customized Cost-Saving Analysis, specific COA data for our existing inventory, and comprehensive route feasibility assessments tailored to your project's unique requirements. Let us be your trusted partner in driving innovation through chemistry.
