Scalable Biocatalytic Production of High-Purity Hydroxytyrosol for Pharmaceutical Applications
The global demand for potent natural antioxidants has placed Hydroxytyrosol, a phenolic compound primarily derived from olive oil, at the forefront of pharmaceutical and nutraceutical research. Recognized for its exceptional ability to prevent low-density lipoprotein (LDL) oxidation and suppress carcinogenic processes, the commercial viability of this molecule hinges on the development of efficient, high-yield synthesis routes. Patent CN101624607A introduces a groundbreaking biocatalytic methodology that addresses the longstanding challenges of selectivity and purity in Hydroxytyrosol production. Unlike traditional chemical oxidation methods that often suffer from poor regioselectivity and harsh reaction conditions, this invention leverages the specificity of polyphenol oxidase (PPO) coupled with a strategic inhibition mechanism using ascorbic acid. The core innovation lies in the precise control of the enzymatic oxidation of p-hydroxyphenylethanol, ensuring the reaction halts at the desired dihydroxy stage rather than proceeding to unwanted quinone byproducts. 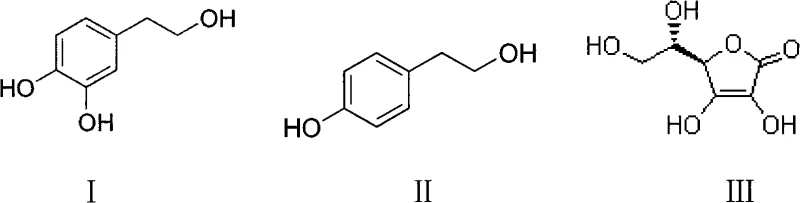 This structural transformation, depicted in the accompanying diagram, highlights the conversion of the mono-phenolic precursor (II) into the valuable di-phenolic target (I), facilitated by the protective presence of ascorbic acid (III). For R&D directors and procurement specialists seeking a reliable hydroxytyrosol supplier, this patent represents a paradigm shift towards greener, more sustainable manufacturing protocols that align with modern regulatory standards for food and drug safety.
This structural transformation, depicted in the accompanying diagram, highlights the conversion of the mono-phenolic precursor (II) into the valuable di-phenolic target (I), facilitated by the protective presence of ascorbic acid (III). For R&D directors and procurement specialists seeking a reliable hydroxytyrosol supplier, this patent represents a paradigm shift towards greener, more sustainable manufacturing protocols that align with modern regulatory standards for food and drug safety.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of catechol derivatives like Hydroxytyrosol has been plagued by significant technical hurdles inherent to non-enzymatic chemical oxidation. Traditional approaches often rely on strong oxidizing agents or transition metal catalysts, which frequently lack the necessary specificity to distinguish between the initial hydroxylation and subsequent over-oxidation. This lack of control results in the formation of complex mixtures containing o-quinones and polymerized tarry substances, necessitating laborious and costly purification steps to isolate the target molecule. Furthermore, chemical methods typically require extreme pH levels or elevated temperatures that can degrade sensitive functional groups, leading to reduced overall yields and the generation of hazardous waste streams. From a supply chain perspective, the reliance on heavy metal catalysts introduces stringent requirements for residual metal testing and removal, adding layers of complexity and cost to the final quality control process. These inefficiencies make conventional chemical routes less attractive for the cost reduction in pharmaceutical intermediates manufacturing, particularly when scaling up to meet the rigorous purity specifications demanded by the global market.
The Novel Approach
The methodology disclosed in CN101624607A offers a sophisticated solution by mimicking biological pathways to achieve unparalleled selectivity under mild conditions. By utilizing polyphenol oxidase as a biocatalyst, the process operates efficiently at a neutral pH of roughly 6.5 and moderate temperatures around 50°C, significantly reducing energy consumption and thermal stress on the product. The critical breakthrough is the dual-function role of ascorbic acid, which not only serves as a reactant but acts as a kinetic regulator to suppress the enzyme's bis-phenolase activity. This clever manipulation of enzyme kinetics prevents the rapid conversion of the newly formed Hydroxytyrosol into reactive quinones, allowing the desired product to accumulate in the reaction broth to high concentrations. Additionally, the option to use immobilized enzymes enables a continuous flow process, where the biocatalyst can be retained within a reactor column while the substrate flows through, facilitating easy separation and catalyst reuse. This approach not only enhances the commercial scale-up of complex phenolic compounds but also ensures a consistent, high-quality output that is free from the toxic residues associated with synthetic chemistry.
Mechanistic Insights into Polyphenol Oxidase Catalyzed Oxidation
To fully appreciate the technical depth of this invention, one must understand the intricate catalytic cycle of polyphenol oxidase and how it is modulated to favor Hydroxytyrosol accumulation. Polyphenol oxidase typically exhibits two distinct activities: monophenolase activity, which hydroxylates mono-phenols to o-diphenols, and diphenolase activity, which oxidizes o-diphenols to o-quinones. In a standard reaction without intervention, the enzyme would rapidly convert the intermediate Hydroxytyrosol into its corresponding o-quinone, leading to product loss and the formation of dark pigments. 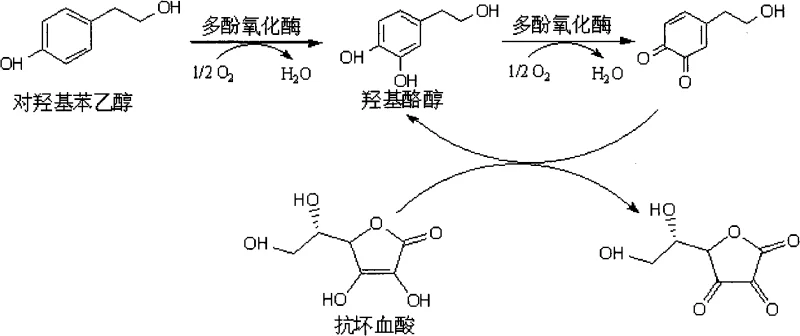 The reaction mechanism illustrated above demonstrates how the introduction of ascorbic acid disrupts this cycle. Ascorbic acid acts as a preferential reducing agent for the o-quinone species, chemically reducing them back to the stable o-diphenol (Hydroxytyrosol) form faster than they can undergo further polymerization or side reactions. Simultaneously, ascorbic acid competitively inhibits the diphenolase active site of the enzyme, effectively slowing down the oxidation rate of the product while allowing the hydroxylation of the substrate to proceed. This creates a 'kinetic trap' where the concentration of Hydroxytyrosol builds up significantly, transforming what would normally be a transient intermediate into the major endpoint of the reaction. For technical teams, understanding this redox balance is crucial for optimizing the molar ratios of substrate to inhibitor to maximize space-time yield in large-scale bioreactors.
The reaction mechanism illustrated above demonstrates how the introduction of ascorbic acid disrupts this cycle. Ascorbic acid acts as a preferential reducing agent for the o-quinone species, chemically reducing them back to the stable o-diphenol (Hydroxytyrosol) form faster than they can undergo further polymerization or side reactions. Simultaneously, ascorbic acid competitively inhibits the diphenolase active site of the enzyme, effectively slowing down the oxidation rate of the product while allowing the hydroxylation of the substrate to proceed. This creates a 'kinetic trap' where the concentration of Hydroxytyrosol builds up significantly, transforming what would normally be a transient intermediate into the major endpoint of the reaction. For technical teams, understanding this redox balance is crucial for optimizing the molar ratios of substrate to inhibitor to maximize space-time yield in large-scale bioreactors.
Beyond the primary catalytic cycle, the patent details a robust downstream processing strategy designed to handle the complexities of aqueous enzymatic reactions. The use of nanofiltration membranes with a molecular weight cut-off between 200Da and 1000Da allows for the precise separation of the low molecular weight product from the high molecular weight enzyme and buffer salts. This membrane step is critical for concentrating the product stream prior to chromatographic purification, reducing the volume load on subsequent resin columns. The employment of macroporous adsorption resins, specifically styrene or polyamide types, leverages hydrogen bonding and hydrophobic interactions to selectively retain Hydroxytyrosol while washing away polar impurities. This multi-stage purification protocol ensures that the final product meets the stringent purity specifications required for pharmaceutical applications, effectively removing trace proteins, residual sugars, and any minor oxidative byproducts that might have escaped the enzymatic control mechanisms.
How to Synthesize Hydroxytyrosol Efficiently
The practical implementation of this biocatalytic route involves a streamlined sequence of unit operations that bridge the gap between laboratory discovery and industrial reality. The process begins with the preparation of a buffered reaction system where p-hydroxyphenylethanol is dissolved alongside a stoichiometric amount of ascorbic acid to establish the necessary inhibitory environment. Following the addition of the polyphenol oxidase catalyst, the mixture is subjected to controlled aeration to supply the oxygen required for the hydroxylation reaction, maintaining a temperature of approximately 50°C to optimize enzyme turnover rates. Once the conversion reaches completion, the reaction broth undergoes a series of separation steps including nanofiltration to remove the biocatalyst, followed by adsorption on macroporous resin to capture the product. The detailed standardized synthesis steps, including specific solvent ratios for elution and crystallization parameters, are outlined below to guide process engineers in replicating this high-efficiency protocol.
- Dissolve p-hydroxyphenylethanol in a buffered solution (pH 4-7) and add ascorbic acid to inhibit bis-phenol activity.
- Introduce polyphenol oxidase (free or immobilized) and oxygen to catalyze the selective oxidation reaction at 50°C.
- Purify the reaction mixture through nanofiltration, macroporous resin adsorption, ethyl acetate extraction, and final column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this enzymatic manufacturing route offers compelling economic and logistical benefits that extend far beyond simple yield improvements. The elimination of toxic heavy metal catalysts and harsh organic solvents traditionally used in chemical oxidation significantly simplifies the waste treatment infrastructure required at the production site. This reduction in hazardous waste generation translates directly into lower disposal costs and a diminished environmental footprint, aligning perfectly with the increasingly strict sustainability mandates imposed by global regulatory bodies. Furthermore, the ability to operate under mild aqueous conditions reduces the energy intensity of the process, as there is no need for high-pressure reactors or extreme heating and cooling cycles, contributing to substantial cost savings in utility consumption over the lifecycle of the plant.
- Cost Reduction in Manufacturing: The implementation of immobilized enzyme technology allows for the repeated reuse of the biocatalyst over multiple batches, effectively amortizing the cost of the enzyme over a much larger production volume. By avoiding the expensive scavenging steps required to remove trace metals from the final API intermediate, manufacturers can streamline their purification workflow and reduce the consumption of specialized filtering media. The high selectivity of the enzymatic reaction minimizes the formation of difficult-to-remove impurities, thereby increasing the overall recovery rate of the valuable product and reducing the raw material input required per kilogram of output.
- Enhanced Supply Chain Reliability: The reliance on bio-based catalysts derived from renewable sources such as potatoes or mushrooms mitigates the supply risks associated with petrochemical-derived reagents and rare earth metals. The continuous flow capability demonstrated in the patent embodiments ensures a steady, uninterrupted output of product, which is critical for maintaining just-in-time inventory levels for downstream pharmaceutical customers. Additionally, the stability of the immobilized enzyme formulation simplifies storage and logistics, as the catalyst can be stored for extended periods without significant loss of activity, ensuring production readiness at all times.
- Scalability and Environmental Compliance: The process design inherently supports linear scale-up from pilot plant to full commercial production without the need for complex re-engineering of the reaction kinetics. The use of water as the primary solvent and ethanol or ethyl acetate for extraction ensures that the volatile organic compound (VOC) emissions are kept to a minimum, facilitating easier compliance with air quality regulations. This green chemistry approach not only future-proofs the manufacturing asset against tightening environmental laws but also enhances the brand value of the final product as a sustainably sourced ingredient for health-conscious consumers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the biocatalytic production of Hydroxytyrosol, providing clarity on the operational nuances and quality assurances of this patented method. These insights are derived directly from the experimental data and process descriptions found in the source intellectual property, offering a transparent view into the feasibility of adopting this technology for large-scale operations. Understanding these details is essential for stakeholders evaluating the technical risk and potential return on investment associated with switching from conventional chemical synthesis to this advanced enzymatic platform.
Q: How does ascorbic acid improve the yield of Hydroxytyrosol in enzymatic synthesis?
A: Ascorbic acid acts as a specific inhibitor of the bis-phenol activity of polyphenol oxidase. It prevents the over-oxidation of the desired Hydroxytyrosol into o-quinones, effectively recycling the intermediate back to the catechol form and significantly increasing accumulation.
Q: What are the advantages of using immobilized enzymes in this process?
A: Immobilized enzymes offer superior stability and can be easily separated from the reaction mixture via centrifugation or filtration. This allows for enzyme recovery and repeated use, which drastically reduces catalyst costs and facilitates continuous flow processing.
Q: Is this biocatalytic method suitable for large-scale industrial production?
A: Yes, the process is designed for industrial scalability. It utilizes continuous operation modes, such as packed-bed reactors for immobilized enzymes, and employs standard downstream processing techniques like nanofiltration and macroporous resin chromatography that are easily scaled from kilograms to tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Hydroxytyrosol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of high-value antioxidants like Hydroxytyrosol requires more than just a laboratory recipe; it demands a partner with deep expertise in process engineering and quality assurance. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale optimization to industrial manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, utilizing state-of-the-art analytical instrumentation to verify identity, potency, and impurity profiles against the highest international standards.
We invite forward-thinking organizations to collaborate with us to leverage this innovative biocatalytic technology for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and sustainability in your product portfolio.
