Optimizing Pitavastatin Intermediate Production: A Novel Synthetic Route for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust synthetic pathways for statin intermediates that balance high stereoselectivity with industrial feasibility. Patent CN111875538A discloses a groundbreaking synthetic method for pitavastatin tert-butyl ester, a critical precursor in the manufacturing of pitavastatin calcium, a potent HMG-CoA reductase inhibitor. This technical insight report analyzes the novel four-step sequence which replaces traditional, waste-intensive methodologies with a streamlined process involving nucleophilic substitution, oxidation, Julia-type olefination, and acid deprotection. By leveraging a heterocyclic sulfone strategy, this route addresses the persistent challenge of cis-isomer formation that has plagued previous generations of synthesis. For R&D directors and procurement specialists, understanding this shift is vital, as it represents a transition from laboratory-scale curiosity to a commercially viable process capable of delivering high-purity pharmaceutical intermediates with significantly reduced environmental impact and operational complexity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
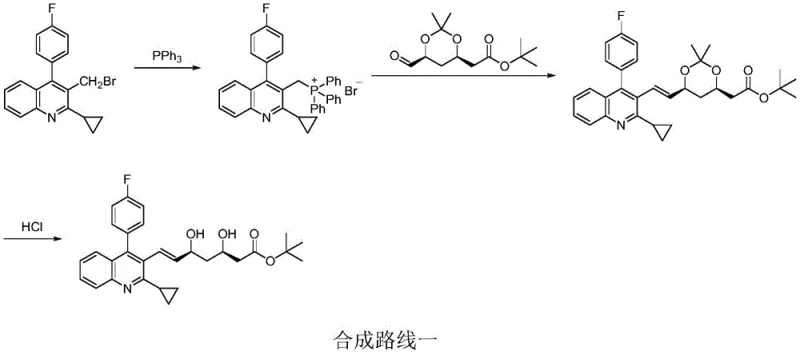
Historically, the synthesis of pitavastatin intermediates has relied heavily on the Wittig reaction, a method fraught with stereochemical inefficiencies. As illustrated in the legacy pathway, the reaction of a quinoline phosphonium salt with a side-chain aldehyde typically yields a mixture of E and Z isomers, with the undesirable cis-isomer content reaching as high as 20%. This stereochemical impurity necessitates rigorous and costly purification steps to meet pharmacopeial standards. Furthermore, the Wittig reaction generates stoichiometric amounts of triphenylphosphine oxide, a solid waste byproduct that complicates downstream processing and increases the burden on waste treatment facilities. Another conventional approach involves Mitsunobu chemistry, which, while effective for inversion of configuration, often requires ultra-low temperature conditions and expensive reagents like diisopropyl azodicarboxylate. These harsh conditions and the generation of hydrazine byproducts create significant safety hazards and cost barriers for large-scale manufacturing, rendering these traditional routes suboptimal for modern supply chains demanding both efficiency and sustainability.
The Novel Approach
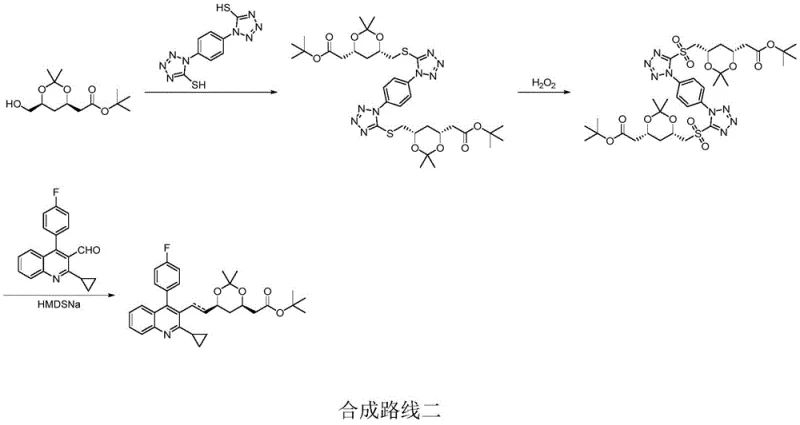
In stark contrast to the legacy methodologies, the novel approach detailed in the patent data utilizes a Julia-Kocienski olefination strategy that fundamentally alters the reaction landscape. By employing a heterocyclic thiol, such as a thiadiazole derivative, to form a sulfone intermediate, the synthesis achieves exceptional control over the double bond geometry. This method operates under mild and controllable reaction conditions, specifically avoiding the ultra-low temperatures required by previous sulfone-based routes. The oxidation step, utilizing a hydrogen peroxide system catalyzed by ammonium molybdate, ensures complete conversion of the sulfide to the sulfone without the steric hindrance issues that often lead to incomplete oxidation in other methods. The result is a process that not only simplifies the operational workflow but also drastically improves the overall yield and purity profile, making it an ideal candidate for reliable pharmaceutical intermediate supplier networks aiming to optimize their production capabilities.
Mechanistic Insights into Julia-Kocienski Olefination
The core of this technological breakthrough lies in the mechanistic elegance of the Julia-Kocienski olefination, which facilitates the construction of the critical carbon-carbon double bond with high E-selectivity. The reaction begins with the formation of a sulfone intermediate (Substance C) from a chloromethyl dioxolane precursor. Upon treatment with a strong base such as sodium hydride in tetrahydrofuran, the sulfone generates a carbanion that attacks the quinoline aldehyde. Unlike the betaine intermediates formed in Wittig reactions which can revert and equilibrate to form cis-isomers, the sulfone anion addition leads to a beta-alkoxy sulfone intermediate that undergoes a concerted syn-elimination. This elimination step is driven by the expulsion of the heterocyclic sulfinate leaving group, a process that is thermodynamically favored to produce the trans-alkene. The use of specific heterocycles like thiadiazoles enhances the acidity of the alpha-proton and stabilizes the transition state, ensuring that the reaction proceeds with minimal formation of the Z-isomer, thereby securing the stereochemical integrity of the final pitavastatin scaffold.
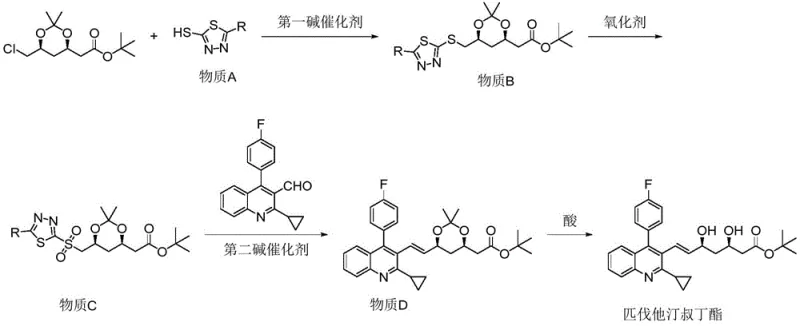
Beyond stereocontrol, the mechanism offers profound advantages in impurity management, a key concern for R&D directors overseeing quality control. The oxidative step converting the sulfide to the sulfone is highly selective, utilizing a catalytic amount of ammonium molybdate with hydrogen peroxide to prevent over-oxidation or side reactions on the sensitive dioxolane ring. The subsequent olefination step proceeds with such high fidelity that the resulting Substance D contains negligible amounts of cis-isomers, often below the detection limit of standard HPLC methods. This high level of purity is carried through to the final acid deprotection step, where mild hydrochloric acid conditions remove the acetonide protecting group without inducing epimerization or degradation of the sensitive polyene chain. The cumulative effect of these mechanistic features is a final product with purity exceeding 99.9%, eliminating the need for resource-intensive chromatographic purification and ensuring a consistent, high-quality supply of the API intermediate.
How to Synthesize Pitavastatin Tert-Butyl Ester Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize the benefits of the novel pathway. The process is divided into four distinct stages: the initial nucleophilic substitution to install the heterocyclic handle, the oxidation to activate the sulfone, the key olefination to build the carbon skeleton, and the final deprotection. Each step has been optimized to use commercially available solvents and reagents, such as 1,4-dioxane for the substitution and isopropanol for the oxidation, facilitating easy technology transfer. The reaction temperatures are maintained within a practical range, typically between 0°C and 80°C depending on the specific step, which allows for standard reactor equipment to be used without the need for specialized cryogenic infrastructure. For a detailed breakdown of the specific molar ratios, addition rates, and workup procedures required to replicate this high-yield process, please refer to the standardized technical guide below.
- React (4R-Cis)-6-chloromethyl-2,2-dimethyl-1,3-dioxolane-4-tert-butyl acetate with a heterocyclic thiol substance A using a first base catalyst to form substance B.
- Oxidize substance B using a hydrogen peroxide system or m-chloroperoxybenzoic acid to obtain the sulfone intermediate, substance C.
- Condense substance C with 2-cyclopropyl-4-(4-fluorophenyl)-quinoline-3-formaldehyde under a second base catalyst to generate the olefin substance D.
- Perform acid deprotection on substance D using dilute hydrochloric acid to yield the final high-purity pitavastatin tert-butyl ester.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers transformative benefits for procurement managers and supply chain heads focused on cost reduction in pharmaceutical intermediate manufacturing. The elimination of triphenylphosphine removes a major cost driver and waste disposal liability, significantly simplifying the post-reaction workup. The high stereoselectivity means that yield losses associated with isomer separation are virtually non-existent, directly translating to higher throughput per batch. Furthermore, the use of mild reaction conditions reduces energy consumption associated with heating and cooling, contributing to a lower carbon footprint and reduced operational expenditures. The robustness of the chemistry ensures consistent batch-to-batch quality, minimizing the risk of production delays and enhancing supply chain reliability for downstream API manufacturers.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive phosphine reagents and the subsequent removal of phosphine oxide waste, which traditionally requires extensive washing and filtration steps. By avoiding column chromatography and relying on crystallization for purification, the consumption of silica gel and organic solvents is drastically reduced. This streamlined workflow lowers the overall cost of goods sold (COGS) and allows for more competitive pricing structures in the global market for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The reagents used in this pathway, such as sodium carbonate, hydrogen peroxide, and common organic solvents, are commodity chemicals with stable global supply chains. This reduces the risk of raw material shortages that can plague specialized reagent-dependent processes. Additionally, the high yield and purity of the intermediate reduce the need for re-processing or re-running batches, ensuring that delivery schedules are met consistently and that inventory levels can be maintained with greater predictability for long-term contracts.
- Scalability and Environmental Compliance: The absence of heavy metal catalysts and the reduction of hazardous waste streams make this process highly scalable and compliant with increasingly stringent environmental regulations. The oxidation step uses a green oxidant system, and the overall atom economy is superior to the Wittig alternative. This environmental profile facilitates easier permitting for new production lines and aligns with the sustainability goals of major pharmaceutical companies, making the supplier a preferred partner for green chemistry initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic method. They are derived from the specific advantages outlined in the patent data, focusing on the practical implications for scaling up production and ensuring product quality. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for commercial manufacturing of statin intermediates.
Q: How does this novel synthetic route improve stereoselectivity compared to traditional Wittig reactions?
A: Traditional Wittig reactions often generate significant amounts of cis-isomers (up to 20%), requiring difficult separation. This novel method utilizes a Julia-Kocienski type olefination which inherently favors the E-isomer, resulting in a product completely free of cis-isomers and eliminating the need for complex isomer separation.
Q: What are the specific reaction conditions for the key olefination step?
A: The key olefination step reacts the sulfone intermediate with the quinoline aldehyde using a strong base like sodium hydride or sodium tert-butoxide. Crucially, this reaction proceeds efficiently at temperatures between -10°C and 10°C, avoiding the ultra-low temperature requirements of previous methods.
Q: Why is this method considered more suitable for industrial scale-up?
A: The process avoids the use of triphenylphosphine, which generates large amounts of triphenylphosphine oxide waste. Additionally, the oxidation step achieves high conversion without incomplete oxidation byproducts, and the final product purity exceeds 99.9% without the need for repeated column chromatography.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pitavastatin Tert-Butyl Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the competitive landscape of statin production. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel patent are fully realized in an industrial setting. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of verifying the absence of cis-isomers and other critical impurities at every stage of the synthesis. We are committed to delivering high-purity pharmaceutical intermediates that meet the exacting standards of global regulatory bodies, providing our partners with a secure and high-quality supply chain foundation.
We invite procurement leaders and R&D teams to engage with us for a Customized Cost-Saving Analysis tailored to your specific production volumes. By leveraging our technical expertise in Julia olefination and sulfone chemistry, we can help you transition to this more efficient manufacturing process. Please contact our technical procurement team to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced capabilities can optimize your supply chain for pitavastatin production.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
