Advanced Manufacturing of Dopexamine Hydrochloride: Overcoming Traditional Demethylation Limitations via Catalytic Hydrogenation
Advanced Manufacturing of Dopexamine Hydrochloride: Overcoming Traditional Demethylation Limitations via Catalytic Hydrogenation
The pharmaceutical industry continuously seeks robust synthetic routes that balance high purity with operational safety, particularly for cardiovascular agents like Dopexamine Hydrochloride (FPL-60278AR). Patent CN102336676A introduces a transformative approach utilizing an arylmethyl protection strategy, fundamentally shifting the paradigm from harsh chemical demethylation to elegant catalytic hydrogenation. This innovation addresses critical pain points in the manufacturing of adrenomimetic drugs, specifically targeting the instability of the catechol moiety during deprotection. By employing bis-arylmethyl protected intermediates, manufacturers can achieve selective removal of protecting groups under mild conditions, thereby preserving the integrity of the sensitive 1,2-dihydroxy-benzene structure. This technical breakthrough not only enhances the chemical purity of the final active pharmaceutical ingredient but also streamlines the downstream processing required to obtain the pharmacologically active hydrochloride salt form.
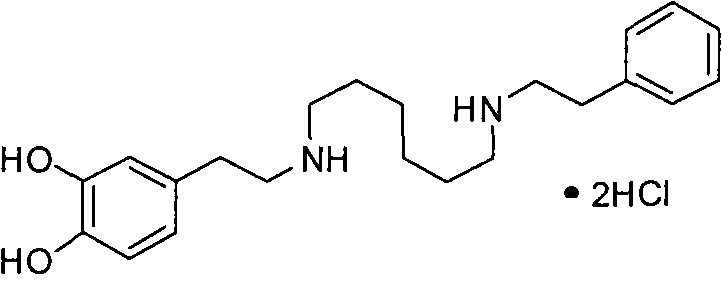
The core of this invention lies in the strategic substitution of traditional methyl ethers with arylmethyl ethers, which serve as robust protecting groups during the construction of the molecular backbone. Unlike methyl groups that require aggressive Lewis acids or strong mineral acids for cleavage, arylmethyl groups can be cleanly removed via hydrogenolysis. This distinction is pivotal for large-scale production, as it mitigates the formation of quinoid byproducts that typically arise from the oxidation of catechols under alkaline neutralization conditions found in older methods. The versatility of the aryl group (Ar), which can range from phenyl to various substituted naphthyl or heterocyclic systems, offers process chemists significant flexibility in tuning the physicochemical properties of the intermediate to optimize solubility and crystallization behavior during purification stages.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Dopexamine Hydrochloride has relied heavily on methyl protection strategies, such as those described in early literature by Fisons Company and academic researchers. These conventional routes typically involve the formation of a bis-methoxy protected intermediate, which subsequently requires demethylation using concentrated hydrobromic acid at elevated temperatures for extended periods. This harsh chemical environment poses severe challenges, including significant equipment corrosion, safety hazards associated with handling hot concentrated acids, and notoriously low overall yields, often reported as low as 16.8% to 57.6%. Furthermore, the demethylation step invariably produces the hydrobromide salt of the drug, necessitating an additional, cumbersome salt exchange process involving alkaline liberation and subsequent re-acidification with hydrochloric acid. This multi-step manipulation exposes the sensitive catechol ring to oxidative stress, frequently resulting in the formation of colored quinoid impurities that are difficult to remove and compromise the quality of the final API.
The Novel Approach
In stark contrast, the novel arylmethyl protection strategy detailed in the patent data offers a streamlined pathway that bypasses these detrimental conditions entirely. By utilizing arylmethyl groups, the deprotection can be achieved through catalytic hydrogenation, a reaction that proceeds efficiently at room temperature or slightly elevated temperatures under hydrogen pressure. This method eliminates the need for hydrobromic acid, thereby removing the requirement for a salt conversion step; when the reaction is conducted in a solvent containing hydrogen chloride, the product precipitates directly as the desired hydrochloride salt. This direct formation not only simplifies the isolation procedure but also drastically reduces the potential for oxidative degradation, as the reaction environment remains reducing throughout the critical deprotection phase. The result is a process with near-quantitative yields in the deprotection step, significantly higher than the fractional yields observed in traditional methyl-based routes, ensuring a more economical and sustainable manufacturing process.
Mechanistic Insights into Catalytic Hydrogenation Deprotection
The mechanistic elegance of this process centers on the transition metal-catalyzed hydrogenolysis of the benzylic carbon-oxygen bond. When the bis-arylmethyl protected intermediate is exposed to hydrogen gas in the presence of a catalyst such as palladium on carbon (Pd/C), the metal surface facilitates the adsorption and activation of hydrogen molecules. These activated hydrogen species then attack the benzylic position, cleaving the ether linkage to release the free phenolic hydroxyl groups and the corresponding hydrocarbon byproduct (e.g., toluene if benzyl protection is used). The presence of hydrogen chloride in the solvent system plays a dual role: it protonates the amine functionalities to prevent catalyst poisoning by basic nitrogen atoms, and it ensures that the liberated amine immediately forms the stable hydrochloride salt upon completion of the reaction. This synergy between the catalyst and the acidic medium is crucial for maintaining high catalytic activity and preventing the formation of side products.
Impurity control is inherently built into this mechanistic pathway due to the mildness of the reaction conditions. In traditional base-mediated workups, the electron-rich catechol ring is highly susceptible to oxidation by atmospheric oxygen, leading to complex mixtures of quinones and polymers. However, the hydrogenation environment is strictly anaerobic and reducing, effectively suppressing any oxidative pathways. Furthermore, the selectivity of the hydrogenolysis ensures that other functional groups within the molecule, such as the secondary amines in the polyamine chain, remain intact without undergoing unwanted hydrogenation or reduction. This high degree of chemoselectivity simplifies the purification profile, allowing for the production of high-purity Dopexamine Hydrochloride that meets stringent regulatory specifications for cardiovascular medications without the need for extensive chromatographic purification.
How to Synthesize Dopexamine Hydrochloride Efficiently
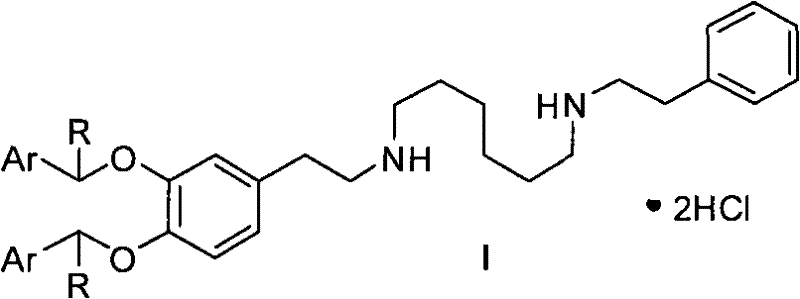
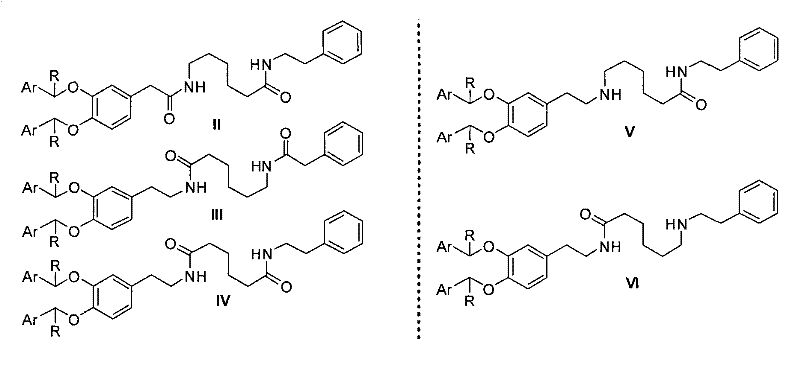
The synthesis of Dopexamine Hydrochloride via this advanced route involves a logical sequence of condensation, reduction, and deprotection steps that are highly amenable to scale-up. Initially, the protected precursors (Compounds II through VI) are assembled using standard amide coupling or substitution reactions, utilizing readily available starting materials like 6-aminocaproic acid and substituted phenylethylamines. These intermediates are then subjected to reduction using hydride reagents such as sodium borohydride or lithium aluminum hydride, often in combination with Lewis acids to enhance reactivity, yielding the fully protected amine (Compound I). The final and most critical transformation is the catalytic hydrogenation, which removes the protecting groups to reveal the active drug substance. For a detailed breakdown of the specific reaction parameters, stoichiometry, and workup procedures required to execute this synthesis successfully, please refer to the standardized guide below.
- Synthesize bis-arylmethyl protected precursors (Compound I) via reduction of bis-amide or mono-amide intermediates using hydride reagents.
- Perform catalytic hydrogenation using transition metal catalysts (e.g., Pd/C) in acidic alcoholic solvents to remove arylmethyl groups.
- Isolate the final Dopexamine Hydrochloride directly from the reaction mixture through crystallization, avoiding salt conversion steps.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this arylmethyl protection strategy represents a significant opportunity to optimize the cost structure and reliability of the Dopexamine Hydrochloride supply chain. The elimination of hydrobromic acid from the process removes a major hazardous material from the facility, reducing costs associated with specialized corrosion-resistant equipment, hazardous waste disposal, and safety compliance protocols. Moreover, the direct formation of the hydrochloride salt eliminates an entire unit operation—the salt exchange step—which traditionally consumes significant amounts of solvents, bases, and acids while incurring yield losses. This simplification translates directly into reduced manufacturing cycle times and lower variable costs per kilogram, making the final API more cost-competitive in the global market without compromising on quality standards.
- Cost Reduction in Manufacturing: The transition to catalytic hydrogenation significantly lowers operational expenditures by removing the need for expensive and corrosive hydrobromic acid reagents. Additionally, the near-quantitative yield of the deprotection step minimizes raw material waste, ensuring that expensive precursors are converted into saleable product with maximum efficiency. The ability to use standard stainless steel reactors for hydrogenation, rather than glass-lined or Hastelloy vessels required for hot acid digestion, further reduces capital expenditure requirements for production facilities.
- Enhanced Supply Chain Reliability: By simplifying the synthetic route and reducing the number of isolation steps, the risk of batch failure due to process deviations is substantially mitigated. The robustness of the hydrogenation step ensures consistent batch-to-batch quality, which is critical for maintaining regulatory approval and avoiding supply disruptions. Furthermore, the starting materials for the arylmethyl protected intermediates are commercially available and stable, reducing the risk of raw material shortages that can plague more exotic synthetic pathways.
- Scalability and Environmental Compliance: Catalytic hydrogenation is a well-established unit operation in the fine chemical industry, known for its excellent scalability from pilot plant to commercial tonnage. The process generates benign byproducts such as toluene, which can be easily recovered and recycled, aligning with green chemistry principles. The absence of heavy metal waste streams associated with certain demethylation reagents and the reduction in aqueous waste from salt exchange operations contribute to a lower environmental footprint, facilitating easier permitting and long-term sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering a clear perspective on why this technology is becoming the preferred choice for modern pharmaceutical manufacturing. Understanding these nuances is essential for technical teams evaluating process transfers or seeking to qualify new suppliers for critical cardiovascular intermediates.
Q: Why is the arylmethyl protection strategy superior to traditional methyl protection for Dopexamine?
A: Traditional methyl protection requires harsh hydrobromic acid conditions for demethylation, leading to low yields and oxidation byproducts. The arylmethyl strategy allows mild catalytic hydrogenation, directly yielding the stable hydrochloride salt with higher purity.
Q: What catalysts are suitable for the deprotection step in this novel process?
A: Transition metal catalysts such as palladium on carbon (Pd/C), ruthenium, rhodium, or nickel are effective. Palladium on carbon is particularly preferred for its high activity and ease of removal under standard hydrogenation conditions.
Q: How does this process improve supply chain stability for cardiovascular API intermediates?
A: By eliminating the need for corrosive hydrobromic acid and complex salt exchange procedures, the process reduces equipment corrosion risks and processing time, ensuring more consistent batch-to-batch quality and reliable delivery schedules.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dopexamine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic technologies to meet the evolving demands of the global pharmaceutical market. Our technical team has extensively analyzed the arylmethyl protection strategy and possesses the expertise to implement this route effectively, ensuring the delivery of high-purity Dopexamine Hydrochloride that exceeds industry standards. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, leveraging our state-of-the-art hydrogenation facilities and stringent purity specifications to guarantee product consistency. Our rigorous QC labs are equipped to detect and quantify trace impurities, ensuring that every batch meets the strict requirements for cardiovascular API intermediates.
We invite procurement leaders and R&D directors to collaborate with us to leverage these technological advantages for your supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that demonstrates how switching to this improved synthesis route can reduce your total landed costs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your project timelines, ensuring a seamless transition to a more efficient and reliable supply source.
