Advanced Saddle-Type Perylene Imide Tetramers: Scalable Synthesis for High-Efficiency Organic Photovoltaics
The rapid evolution of the renewable energy sector has placed immense pressure on the development of next-generation organic photovoltaic materials that can surpass the limitations of traditional silicon-based technologies. Patent CN109553625B introduces a groundbreaking saddle-type perylene imide tetramer that addresses critical stability and efficiency bottlenecks inherent in current non-fullerene acceptor systems. This innovation leverages a cyclooctatetraene core to create a distorted three-dimensional geometry, fundamentally altering the molecular packing behavior to prevent the severe aggregation that typically plagues planar perylene derivatives. For R&D directors and procurement specialists alike, this patent represents a significant leap forward in material design, offering a pathway to higher photoelectric conversion efficiencies without compromising on synthetic feasibility. The technical breakthrough lies not just in the final structure, but in the robust multi-step synthesis that allows for precise modulation of electronic properties through heterocyclic fusion. As the industry seeks reliable electronic chemical suppliers capable of delivering complex molecular architectures, this technology stands out as a prime candidate for commercial scale-up and integration into high-performance organic solar cell modules.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional non-fullerene acceptor materials, particularly those based on planar perylene bisimides, have long suffered from intrinsic structural drawbacks that limit their practical application in large-area photovoltaic devices. The high planarity of these conventional molecules leads to strong intermolecular pi-pi stacking interactions, which inevitably cause severe molecular aggregation and the formation of large crystalline domains within the active layer. This excessive crystallinity results in increased phase separation dimensions that are often mismatched with the exciton diffusion length, thereby restricting the efficiency of exciton separation and diffusion processes. Furthermore, the rigid planar structure makes it difficult to modulate the absorption spectra and energy levels without complex and costly chemical modifications, leading to high production costs and limited tunability. From a supply chain perspective, the purification of these highly aggregating materials is notoriously difficult, often requiring extensive chromatographic separation that reduces overall yield and increases solvent waste. These technical hurdles have historically slowed the adoption of perylene-based acceptors in favor of fullerene derivatives, despite the latter's own issues with cost and spectral absorption limitations.
The Novel Approach
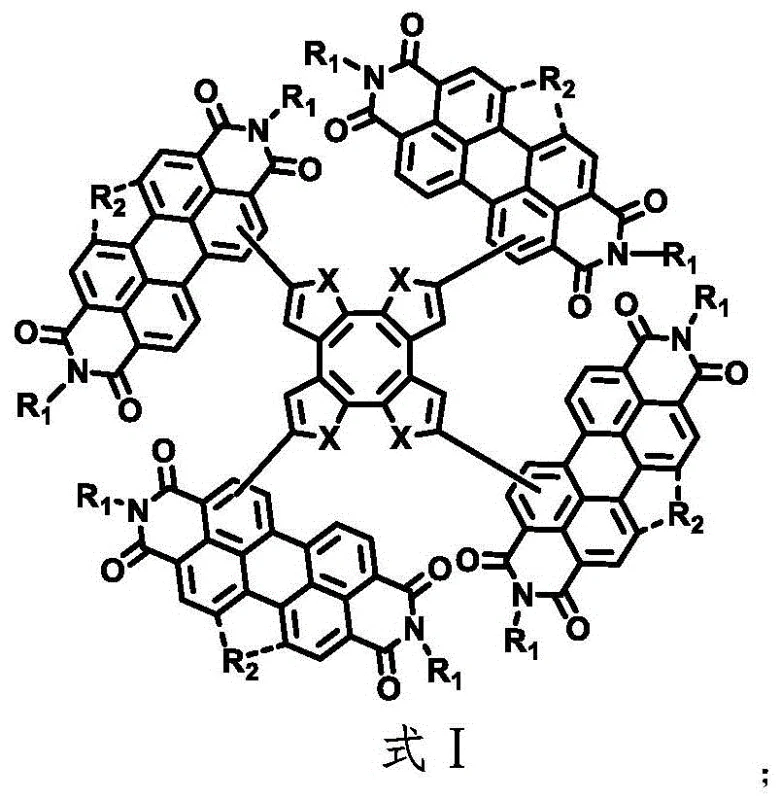
The novel approach detailed in the patent data circumvents these aggregation issues by introducing a saddle-shaped topology centered around a cyclooctatetraene unit, which forces the molecule into a non-planar conformation. By condensing heterocyclic groups at the carbon-carbon double bonds of the cyclooctatetraene core and subsequently coupling four perylene imide units at the active sites, the synthesis creates a sterically hindered architecture that physically prevents tight molecular packing. This distorted three-dimensional geometry effectively inhibits the crystallinity of the material, maintaining a more amorphous morphology in the blend film that is ideal for charge transport. The result is a significant improvement in the photoelectric conversion efficiency, with reported values reaching up to 9.0% in specific derivatives, alongside enhanced short-circuit current and fill factor metrics. This structural innovation not only solves the technical problem of low efficiency but also simplifies the downstream processing requirements, as the material exhibits better solubility and film-forming properties compared to its planar counterparts. For manufacturers, this means a more reliable specialty chemical production process with fewer defects related to phase separation.
Mechanistic Insights into Stille Coupling and Photochemical Oxidation
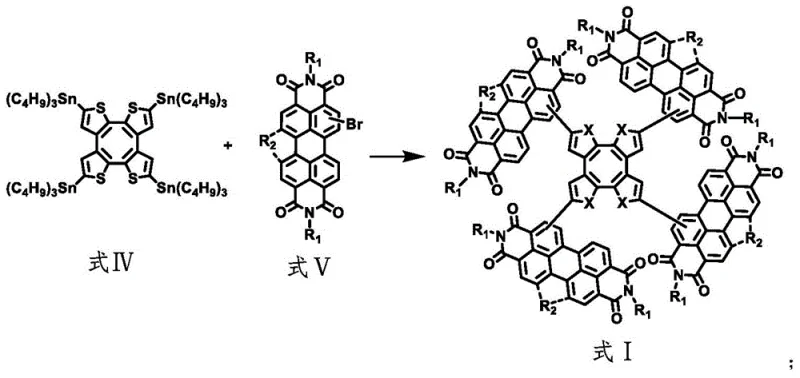
The synthesis mechanism relies on a sophisticated sequence of organometallic transformations beginning with the lithiation of a tetrabromo-cyclooctatetraene derivative at cryogenic temperatures ranging from -90°C to -60°C. This initial step is critical for generating the reactive organolithium intermediate, which is subsequently trapped with tributyltin chloride to form the key organotin species required for the cross-coupling reaction. The precision required in temperature control during this phase ensures high regioselectivity and prevents side reactions that could compromise the integrity of the cyclooctatetraene core. Following the formation of the organotin intermediate, the process employs a palladium-catalyzed Stille coupling reaction with brominated perylene imide derivatives, utilizing catalysts such as tetrakis(triphenylphosphine)palladium or palladium acetate. This step is the cornerstone of the molecular construction, allowing for the attachment of four large perylene imide wings to the central core in a single pot, which is a remarkable feat of synthetic efficiency. The reaction conditions, typically involving heating to 100°C to 120°C in inert organic solvents like toluene, facilitate the formation of the carbon-carbon bonds necessary to assemble the tetramer structure without degrading the sensitive imide functionalities.
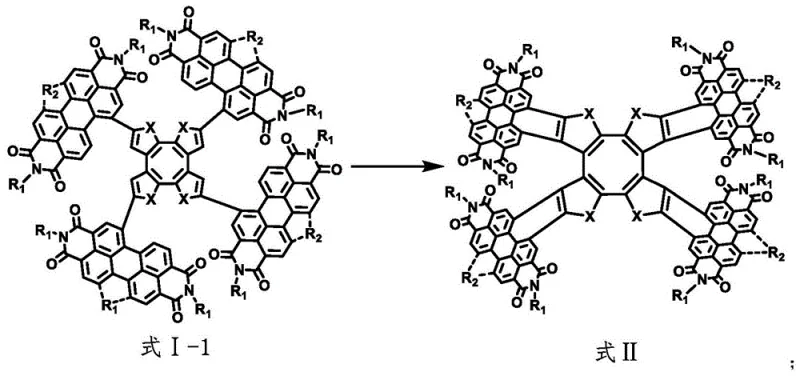
Following the assembly of the precursor tetramer, the synthesis employs a photochemical oxidation step to induce ring closure, locking the molecule into its final saddle-shaped conformation as seen in Formula II. This photochemical transformation is initiated by irradiation under a high-pressure mercury lamp in the presence of iodine, which acts as an oxidant to facilitate the dehydrogenative cyclization. This step is crucial for stabilizing the three-dimensional geometry and optimizing the electronic conjugation across the molecule, which directly correlates to the observed improvements in electron mobility and absorption properties. The mechanism effectively converts the flexible precursor into a rigid, shape-persistent macrocycle that resists conformational changes during device operation, thereby enhancing long-term stability. From an impurity control perspective, the distinct chemical shift between the precursor and the oxidized product allows for efficient separation via column chromatography using standard silica gel and solvent systems. This mechanistic clarity provides R&D teams with a clear roadmap for commercial scale-up of complex optoelectronic materials, ensuring that the critical quality attributes of the final product can be consistently maintained across different production batches.
How to Synthesize Saddle-Type Perylene Imide Tetramer Efficiently
The synthesis of this high-performance acceptor material involves a carefully orchestrated sequence of reactions that balance reactivity with selectivity to ensure high purity and yield. The process begins with the preparation of the organotin intermediate, followed by the palladium-catalyzed coupling to form the open-chain tetramer, and concludes with the photochemical ring-closing step. Each stage requires specific attention to solvent choice, temperature gradients, and stoichiometric ratios to maximize the formation of the desired saddle-shaped isomer while minimizing byproducts. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating these results in a pilot or production environment.
- Perform lithiation of tetrabromo-cyclooctatetraene derivative at low temperature followed by stannylation to generate the key organotin intermediate.
- Execute a Stille coupling reaction between the organotin intermediate and brominated perylene imide using a palladium catalyst system.
- Conduct photochemical oxidation under high-pressure mercury lamp irradiation to induce ring closure and finalize the saddle-shaped structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this saddle-type perylene imide technology offers substantial strategic advantages rooted in the simplification of the manufacturing process and the robustness of the supply chain. The synthetic route utilizes commercially available starting materials and standard organometallic reagents, which significantly reduces the risk of raw material shortages and price volatility often associated with exotic catalysts or proprietary precursors. The ability to perform the key coupling steps in common organic solvents like toluene and tetrahydrofuran means that existing chemical infrastructure can be leveraged without the need for costly specialized equipment upgrades. Furthermore, the improved solubility and reduced aggregation of the final product streamline the purification process, leading to a drastic simplification of the downstream processing workflow. This efficiency translates directly into cost reduction in electronic chemical manufacturing, as less time and solvent are required to achieve the high purity specifications demanded by the photovoltaic industry. The qualitative improvement in process robustness ensures a more predictable production schedule, mitigating the risks of batch failures that can disrupt supply continuity.
- Cost Reduction in Manufacturing: The elimination of complex purification steps required for planar aggregates leads to significant operational savings and reduced solvent consumption. By preventing the formation of intractable crystalline masses, the process avoids the need for aggressive recrystallization or extensive washing, which are typically resource-intensive. The use of standard palladium catalysts, which can potentially be recovered and recycled, further contributes to the economic viability of the process on a large scale. Additionally, the higher photoelectric conversion efficiency of the material means that less active material is needed per device to achieve the same power output, effectively lowering the bill of materials for the end customer. These factors combine to create a compelling economic case for switching to this novel acceptor material without compromising on performance metrics.
- Enhanced Supply Chain Reliability: The reliance on widely available chemical building blocks ensures that the supply chain is resilient against geopolitical or logistical disruptions that often affect niche specialty chemicals. The synthetic pathway is modular, allowing for the substitution of different heterocyclic groups or alkyl chains without altering the core reaction conditions, which provides flexibility in sourcing raw materials. This adaptability is crucial for maintaining reducing lead time for high-purity optoelectronic materials, as alternative suppliers can be qualified more easily for standard reagents. The robust nature of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, reducing the rejection rate of incoming shipments and ensuring a steady flow of production. This reliability is a key value proposition for supply chain heads looking to secure long-term contracts for critical photovoltaic components.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are readily transferable from laboratory to industrial reactors without significant re-optimization. The photochemical oxidation step, while requiring specific equipment, is a clean reaction that produces minimal hazardous byproducts compared to traditional chemical oxidants. This aligns with increasingly stringent environmental regulations, reducing the burden of waste treatment and disposal costs associated with heavy metal waste or toxic oxidants. The ability to scale from gram to kilogram quantities while maintaining product consistency demonstrates the commercial scale-up of complex polymer additives and similar fine chemicals is feasible. This environmental and operational efficiency positions the technology as a sustainable choice for manufacturers aiming to reduce their carbon footprint while meeting high-volume demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this saddle-type perylene imide technology in organic photovoltaic applications. These answers are derived directly from the patent specifications and are intended to provide clarity on the material's performance, synthesis, and integration potential. Understanding these details is essential for stakeholders evaluating the feasibility of adopting this new acceptor material in their product lines.
Q: How does the saddle-shaped structure improve device performance?
A: The distorted three-dimensional geometry effectively prevents tight molecular accumulation and inhibits excessive crystallinity, which enhances exciton separation and carrier transmission compared to planar derivatives.
Q: What are the key challenges in scaling this synthesis?
A: The process requires precise temperature control during lithiation and handling of organotin reagents, but the use of standard column chromatography and common solvents facilitates industrial purification.
Q: Is this material compatible with existing organic solar cell architectures?
A: Yes, the material functions as a non-fullerene electron acceptor with adjustable energy levels, making it suitable for integration into standard bulk heterojunction device structures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Perylene Imide Tetramer Supplier
As a leader in the fine chemical industry, NINGBO INNO PHARMCHEM is uniquely positioned to support the commercialization of this advanced saddle-type perylene imide tetramer technology. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We understand the critical importance of stringent purity specifications and rigorous QC labs in the production of optoelectronic materials, where even trace impurities can degrade device performance. Our state-of-the-art facilities are equipped to handle the specific requirements of organometallic synthesis and photochemical processing, guaranteeing a consistent supply of high-quality acceptor materials that meet the demanding standards of the global solar market. By partnering with us, clients gain access to a supply chain that is both robust and responsive, capable of adapting to fluctuating market demands without compromising on quality or delivery timelines.
We invite forward-thinking organizations to collaborate with us to optimize their supply chains and reduce manufacturing costs through the adoption of this superior material technology. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes and performance requirements. We encourage potential partners to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on concrete technical evidence. By leveraging our expertise in high-purity OLED material and related electronic chemicals, we can help you accelerate your product development cycles and secure a competitive advantage in the rapidly evolving renewable energy sector. Let us help you engineer the future of solar technology with materials that deliver both performance and value.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
