Optimizing Relugolix Intermediate Production: A Technical Analysis of Patent CN113563303B
Introduction to Advanced Relugolix Intermediate Manufacturing
The pharmaceutical landscape for gonadotropin-releasing hormone (GnRH) antagonists has been significantly advanced by the development of Relugolix, a potent small molecule therapeutic. However, the commercial viability of such complex active pharmaceutical ingredients (APIs) hinges entirely on the efficiency and robustness of their synthetic pathways. Patent CN113563303B, published in April 2022, introduces a groundbreaking preparation method for Relugolix intermediates that addresses critical bottlenecks in existing manufacturing technologies. This patent discloses a novel sequence involving compounds A through I, culminating in the final API with exceptional purity profiles and minimal heavy metal residues. For R&D directors and procurement specialists, this represents a pivotal shift towards more sustainable and cost-effective production methodologies. The invention specifically targets the elimination of harsh reaction conditions, offering a pathway that is not only environmentally friendlier but also operationally safer for large-scale facilities. By leveraging mild reaction parameters and streamlined post-treatment steps, this technology promises to redefine the supply chain dynamics for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art synthetic routes, such as those detailed in patent CN104703992, typically rely on a strategy of coupling functional groups prior to ring closure. This conventional approach necessitates rigorous reaction conditions, often requiring elevated temperatures and significant pressurization to drive the coupling reactions to completion. Such demanding parameters impose severe constraints on manufacturing infrastructure, mandating the use of specialized high-pressure reactors that entail substantial capital expenditure and rigorous safety protocols. Furthermore, the harsh thermal environments inherent in these legacy processes frequently promote the formation of complex impurity profiles, complicating downstream purification and potentially trapping heavy metal catalysts within the crystal lattice of the product. These factors collectively inflate the cost of goods sold (COGS) and extend lead times, creating vulnerabilities in the supply chain for critical API intermediates. The reliance on aggressive conditions also limits the scalability of the process, as heat transfer and pressure management become increasingly difficult to control as batch sizes increase.
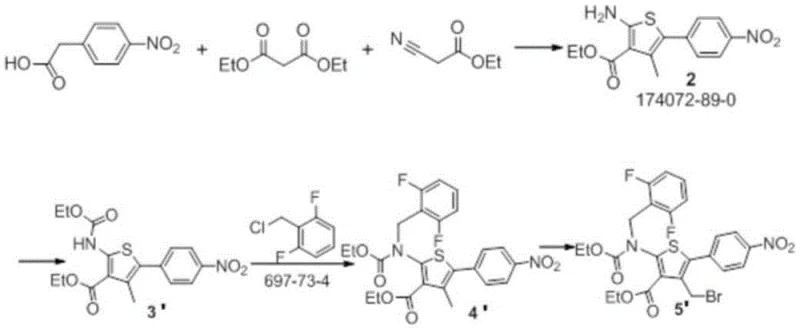
The Novel Approach
In stark contrast, the methodology outlined in CN113563303B adopts a divergent strategy that prioritizes mild conditions and sequential functionalization. As illustrated in the comprehensive reaction scheme below, the new route initiates with a gentle catalytic reduction and proceeds through a series of well-controlled condensation and cyclization steps. This approach effectively decouples the ring-closing events from the high-energy coupling stages, allowing each transformation to occur under optimized, ambient, or moderately heated conditions. The result is a process that is inherently safer and more amenable to standard stainless steel reactor setups, removing the barrier of high-pressure equipment requirements. By avoiding extreme thermal stress, the novel pathway minimizes degradation byproducts, thereby enhancing the overall yield and simplifying the purification workflow. This strategic redesign of the synthetic tree not only improves the chemical efficiency but also aligns perfectly with modern green chemistry principles, reducing the environmental footprint of the manufacturing process while ensuring the final product meets stringent raw material medicine standards.
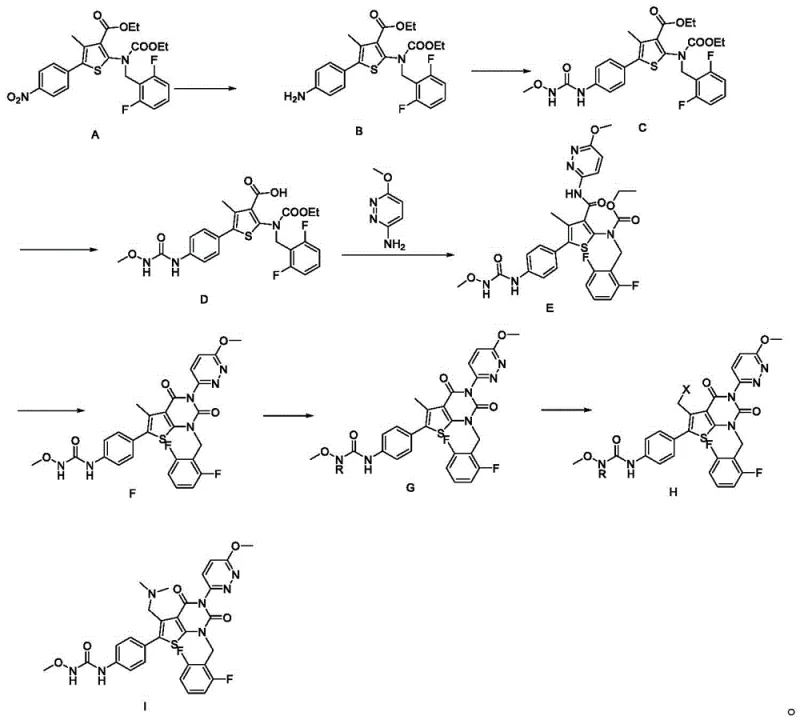
Mechanistic Insights into the Multi-Step Synthesis
The core of this technological advancement lies in the precise orchestration of reaction mechanisms, particularly during the formation of the thieno[2,3-d]pyrimidine core. A critical juncture in this synthesis is the cyclization of Compound E to Compound F, which is achieved using an alkali metal alkoxide, preferably sodium methoxide, in an alcohol solvent. This base-mediated intramolecular cyclization is highly selective, occurring at moderate temperatures between 40 to 80 ℃, which prevents the epimerization or decomposition of sensitive chiral centers or labile ester groups. The mechanism likely involves the deprotonation of an active methylene or amide nitrogen, followed by nucleophilic attack on the adjacent carbonyl or electrophilic center, closing the ring with high fidelity. Following this, the protection of the amine functionality in Compound F using di-tert-butyl dicarbonate (Boc2O) and a catalyst like DMAP ensures that subsequent halogenation steps proceed with regioselectivity. The use of radical initiators such as AIBN in conjunction with N-bromosuccinimide (NBS) allows for the precise installation of a halogen atom at the benzylic position (Compound H), setting the stage for the final nucleophilic substitution. This level of mechanistic control is paramount for maintaining the structural integrity of the molecule throughout the synthesis.
Furthermore, the final amination step converting Compound H to Compound I demonstrates exceptional control over impurity generation. By employing dimethylamine hydrochloride in the presence of an organic base like triethylamine within a nitrile solvent, the reaction proceeds via a clean SN2 mechanism at temperatures as low as 0 to 60 ℃. This mild nucleophilic substitution avoids the formation of quaternary ammonium salts or over-alkylation byproducts that often plague similar transformations in acidic or high-temperature environments. The subsequent removal of the Boc protecting group to yield the final Relugolix API is equally refined, utilizing hydrochloric acid in ethyl acetate under controlled conditions to ensure complete deprotection without hydrolyzing the adjacent amide or ester linkages. The cumulative effect of these mechanistic optimizations is a product with remarkably low heavy metal content, as the avoidance of harsh conditions prevents the leaching of reactor materials and facilitates the efficient removal of palladium residues from the initial reduction step. This rigorous attention to chemical detail ensures that the intermediate and final API consistently achieve the high purity specifications required for global regulatory approval.
How to Synthesize Relugolix Intermediate Efficiently
The synthesis of Relugolix Intermediate I requires a disciplined adherence to the specific reaction parameters outlined in the patent to maximize yield and purity. The process begins with the catalytic hydrogenation of the nitro precursor, followed by a cascade of condensation, hydrolysis, and cyclization reactions that build the complex heterocyclic framework. Each step demands precise control over stoichiometry, temperature, and solvent choice to prevent the accumulation of impurities that could compromise the final drug substance. For example, the hydrolysis of Compound C must be carefully monitored to ensure complete conversion to the acid without saponifying other ester groups, while the cyclization of Compound E requires exact pH adjustment during workup to isolate the product effectively. The detailed standardized synthetic steps for executing this high-efficiency pathway are provided in the technical guide below, offering a roadmap for laboratory and pilot-scale replication.
- Perform catalytic hydrogenation on Compound A using Pd/C in methanol to obtain Compound B.
- Condense Compound B with methoxyamine using CDI catalyst to form Compound C, followed by alkaline hydrolysis to yield Compound D.
- React Compound D with 3-amino-6-methoxypyridazine using T3P to form Compound E, then cyclize with sodium methoxide to get Compound F.
- Protect Compound F with Boc anhydride to form G, halogenate to H, and finally substitute with dimethylamine to obtain Intermediate I.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of the synthesis route described in CN113563303B offers transformative economic and logistical benefits. The primary advantage stems from the drastic simplification of the manufacturing infrastructure; by eliminating the need for high-pressure and high-temperature reactors, facilities can utilize existing standard equipment, thereby significantly reducing capital expenditure and maintenance overheads. This compatibility with standard processing units also accelerates the timeline for technology transfer and scale-up, allowing manufacturers to bring products to market faster. Moreover, the simplified post-treatment steps, which often involve straightforward filtration and washing rather than complex chromatographic separations, translate directly into reduced solvent consumption and waste disposal costs. The ability to produce intermediates with low heavy metal content right out of the reactor further reduces the burden on quality control laboratories and minimizes the risk of batch rejection due to specification failures. These factors combine to create a supply chain that is not only more cost-efficient but also more resilient to disruptions, ensuring a steady flow of high-quality materials for downstream API production.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts in later stages and the removal of high-pressure equipment requirements lead to substantial operational savings. By utilizing readily available reagents like T3P and common solvents such as acetonitrile and ethanol, the raw material costs are optimized, and the dependency on specialized, high-cost catalysts is minimized. The streamlined workup procedures reduce labor hours and utility consumption, contributing to a lower overall cost of goods sold without compromising on the quality of the final pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: The use of commercially available starting materials and reagents ensures that the supply chain is not vulnerable to shortages of exotic or proprietary chemicals. The robustness of the reaction conditions means that production can be maintained consistently across different manufacturing sites with varying levels of equipment sophistication. This flexibility enhances supply continuity, reducing the risk of stockouts and enabling manufacturers to respond more agilely to fluctuations in market demand for GnRH antagonists and related therapeutics.
- Scalability and Environmental Compliance: The mild reaction conditions and reduced solvent usage align with increasingly stringent environmental regulations, facilitating easier permitting and compliance auditing. The process generates less hazardous waste, simplifying disposal and lowering environmental fees. Furthermore, the inherent safety of operating at atmospheric pressure and moderate temperatures makes the process highly scalable, allowing for seamless expansion from kilogram to multi-ton production scales without the need for extensive re-engineering of the process safety protocols.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on how this technology compares to established industry standards. Understanding these nuances is essential for stakeholders evaluating the feasibility of adopting this new methodology for their own production lines or sourcing strategies.
Q: How does the new synthesis route improve product purity compared to conventional methods?
A: The novel route described in CN113563303B avoids harsh high-pressure coupling conditions found in prior art, significantly reducing side reactions and heavy metal contamination, resulting in API-grade purity suitable for direct pharmaceutical use.
Q: What are the key safety advantages of this preparation method?
A: By eliminating the need for high-temperature and high-pressure reactors required in traditional coupling-first strategies, this method operates under mild atmospheric conditions, drastically lowering operational risks and equipment maintenance costs.
Q: Is this process scalable for commercial API manufacturing?
A: Yes, the process utilizes common organic solvents like acetonitrile and ethanol, and reagents such as T3P and Pd/C are commercially available, making the transition from laboratory scale to multi-ton commercial production straightforward and reliable.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Relugolix Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic pathways in the development of life-saving medications like Relugolix. Our team of expert chemists has thoroughly analyzed the technology disclosed in CN113563303B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are committed to delivering high-purity pharmaceutical intermediates that meet the most stringent purity specifications, utilizing our state-of-the-art rigorous QC labs to ensure every batch exceeds regulatory expectations. Our capability to implement complex cyclization and protection-deprotection sequences allows us to offer a reliable supply of Relugolix intermediates that support your clinical and commercial timelines effectively.
We invite you to engage with our technical procurement team to discuss how our manufacturing capabilities can align with your specific project requirements. By partnering with us, you gain access to a Customized Cost-Saving Analysis that demonstrates the tangible economic benefits of switching to this optimized synthesis route. We encourage you to request specific COA data and route feasibility assessments to validate the superior quality and consistency of our intermediates, ensuring a secure and efficient supply chain for your API manufacturing needs.
