Scalable Production of 4-Hydroxy-1-Indanone via Acid-Catalyzed Cyclization for Global Supply Chains
Introduction to Advanced Manufacturing of 4-Hydroxy-1-indanone
The global demand for high-purity pharmaceutical intermediates continues to surge, driven by the development of novel therapeutic agents requiring complex aromatic scaffolds. A pivotal breakthrough in this domain is detailed in patent CN113248356A, which discloses a robust industrial production method for 4-hydroxy-1-indanone (CAS: 40731-98-4). This molecule serves as a critical building block in medicinal chemistry, possessing both phenolic hydroxyl and ketone functionalities that allow for diverse nucleophilic additions and derivatizations. Historically, the synthesis of this compound has been plagued by severe operational bottlenecks, including extreme reaction temperatures and poor scalability. The disclosed technology addresses these challenges by introducing a concise two-step sequence starting from readily available dihydrocoumarin. By shifting from harsh Lewis acid melts to a controlled Brønsted acid system utilizing polyphosphoric acid and strong acid resins, this innovation offers a pathway to reliable pharmaceutical intermediate supplier status, ensuring consistent quality and supply continuity for downstream drug manufacturers.
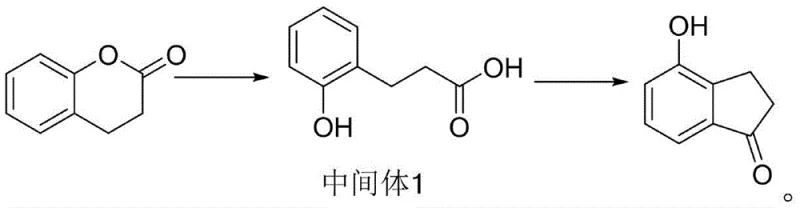
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
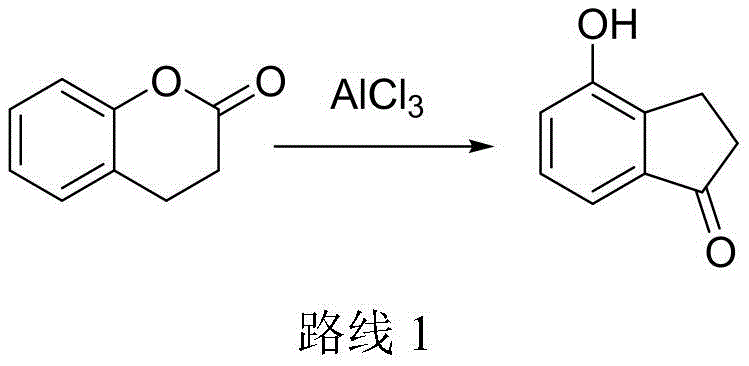
Prior art methodologies for synthesizing 4-hydroxy-1-indanone have consistently failed to meet the rigorous demands of modern industrial chemistry. For instance, Route 1 (WO2014082379) relies on a rearrangement reaction using sodium chloride and aluminum trichloride at excessive temperatures ranging from 180°C to 220°C. While laboratory yields might appear acceptable, scaling this process reveals catastrophic engineering flaws; the reaction mixture becomes a viscous solid melt that cannot be effectively stirred in vessels larger than 1kg, leading to localized overheating, severe carbonization, and significant safety hazards. Furthermore, the quenching of massive amounts of aluminum salts generates voluminous quantities of strong-acid wastewater, creating an untenable environmental burden. Alternatively, Route 2 (WO2010127855) attempts a multi-step approach involving alkaline hydrolysis and protection groups, but it suffers from abysmal overall yields of approximately 37% and requires cryogenic conditions of -50°C, which drastically increases energy consumption and capital expenditure for specialized refrigeration equipment.
The Novel Approach
In stark contrast to these legacy methods, the inventive process described in CN113248356A streamlines the synthesis into a highly efficient two-step protocol that eliminates the need for cryogenics or molten salt baths. The first step involves the hydrolysis of dihydrocoumarin under hydrochloric acid catalysis to generate a key phenolic acid intermediate (Intermediate 1) with exceptional efficiency. The second step is the true innovation: a cyclization reaction performed in a toluene solvent system using polyphosphoric acid (PPA) in conjunction with a macroporous strong acid styrene cation exchange resin. This combination allows the reaction to proceed smoothly at a moderate temperature of 115°C, maintaining a homogeneous liquid phase that ensures perfect mixing and heat transfer even at the 100L pilot scale. This approach not only achieves impressive yields of 86% to 88% but also facilitates solvent recovery and catalyst reuse, representing a paradigm shift in cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Resin-Catalyzed Cyclization
The success of this novel route lies in the sophisticated interplay between the polyphosphoric acid and the solid acid resin, which orchestrates the intramolecular Friedel-Crafts acylation with high precision. Unlike the failed Route 3, where the use of thionyl chloride and acetic anhydride led to the reformation of the starting material due to the acetyl group acting as a leaving group, the new mechanism avoids such reversibility. The strong acid resin activates the phenolic oxygen and the aromatic ring, creating a transition state that favors the nucleophilic attack of the benzene ring onto the carbonyl carbon of the side chain. Simultaneously, the polyphosphoric acid acts as a potent dehydrating agent, driving the equilibrium forward by removing water generated during the ring closure. This synergistic catalysis ensures that the cyclization proceeds irreversibly towards the desired indanone skeleton rather than reverting to the lactone form.
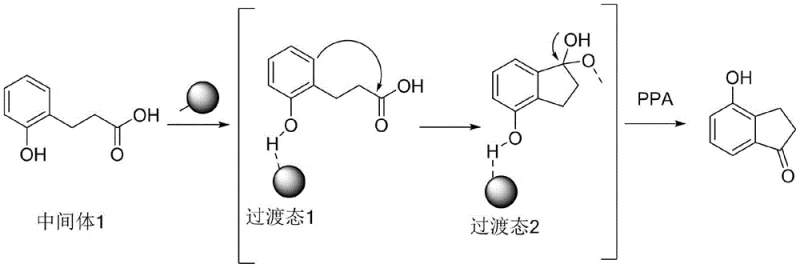
Furthermore, the mechanistic pathway provides inherent advantages regarding impurity control, a critical factor for R&D directors focused on purity specifications. The heterogeneous nature of the strong acid resin minimizes the occurrence of uncontrolled polymerization or over-acylation side reactions that are common with soluble Lewis acids like aluminum chloride. The transition state formed under the influence of the resin exhibits a specific orientation that directs the ring closure to the ortho-position relative to the hydroxyl group, ensuring regioselectivity. Following the formation of the cyclic intermediate, the dehydration step mediated by PPA is clean and efficient, avoiding the formation of chlorinated byproducts often seen when thionyl chloride is employed. This results in a crude product profile that is significantly cleaner, thereby reducing the load on downstream purification units such as activated carbon treatment and crystallization, ultimately delivering high-purity pharmaceutical intermediates suitable for sensitive GMP applications.
How to Synthesize 4-Hydroxy-1-indanone Efficiently
The operational simplicity of this method makes it an ideal candidate for technology transfer and rapid scale-up. The process begins with the acid hydrolysis of dihydrocoumarin, followed by a one-pot cyclization in toluene. The use of standard glass-lined or stainless steel reactors is sufficient, as the corrosion potential is managed by the specific choice of catalysts and solvents. Detailed standard operating procedures regarding stoichiometry, addition rates, and thermal profiles are essential for maximizing the 86-88% yield range observed in pilot studies.
- Hydrolyze dihydrocoumarin using hydrochloric acid at elevated temperatures to obtain the key phenolic acid intermediate.
- Perform cyclization in toluene using polyphosphoric acid and a macroporous strong acid resin catalyst at 115°C.
- Execute aqueous workup, solvent recovery, and crystallization to isolate high-purity 4-hydroxy-1-indanone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers profound strategic benefits beyond mere chemical elegance. The elimination of extreme process conditions directly translates to enhanced operational safety and reduced insurance premiums, while the use of commodity chemicals like hydrochloric acid and toluene ensures raw material security. The ability to recover and recycle toluene significantly lowers the variable cost of goods sold (COGS), and the heterogeneous catalyst can be filtered and potentially regenerated, further driving down long-term operational expenses. This process stability ensures that supply contracts can be honored with greater reliability, mitigating the risk of production stoppages due to equipment fouling or safety incidents associated with older, more volatile chemistries.
- Cost Reduction in Manufacturing: The economic impact of replacing the aluminum chloride melt process with this solvent-based system is substantial. By eliminating the need for specialized high-temperature reactors capable of handling corrosive molten salts, capital expenditure for new production lines is significantly lowered. Moreover, the avoidance of cryogenic cooling (-50°C) removes a major energy drain from the utility budget. The simplified workup procedure, which relies on phase separation and filtration rather than complex extractions or chromatography, reduces labor hours and solvent consumption, leading to drastic improvements in overall process economics without compromising yield.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the complexity of multi-step syntheses or the scarcity of exotic reagents. This two-step route utilizes dihydrocoumarin, a bulk chemical available from multiple global sources, decoupling production from single-source bottlenecks. The robustness of the reaction conditions means that batch-to-batch variability is minimized, allowing for predictable production scheduling. The ability to scale from 100g to 100kg without changing the fundamental reaction parameters ensures that supply can be ramped up quickly to meet sudden spikes in market demand, providing a competitive edge in reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: Modern manufacturing mandates strict adherence to environmental regulations, and this process excels in sustainability metrics. The replacement of stoichiometric amounts of aluminum salts with catalytic amounts of resin and PPA drastically reduces the generation of heavy metal-containing solid waste and acidic wastewater. The closed-loop potential for toluene recovery minimizes VOC emissions. These factors simplify the permitting process for new facilities and reduce the costs associated with waste disposal and environmental remediation, making the commercial scale-up of complex pharmaceutical intermediates both economically and ecologically viable.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this production method is vital for stakeholders evaluating its adoption. The following questions address common concerns regarding process safety, catalyst lifecycle, and product quality assurance. The answers are derived directly from the experimental data and mechanistic studies presented in the patent literature, providing a transparent view of the technology's capabilities and limitations in an industrial setting.
Q: Why is the new resin-catalyzed route superior to the traditional aluminum chloride melt method?
A: The traditional method requires temperatures of 180-220°C and creates a solid melt that is impossible to stir at scale, leading to carbonization and safety hazards. The new route operates in a liquid toluene phase at moderate temperatures (115°C), ensuring excellent heat transfer and mixing.
Q: What is the specific role of the strong acid resin in the cyclization step?
A: The macroporous strong acid styrene cation exchange resin acts as a heterogeneous catalyst that activates the phenolic ring for intramolecular attack while facilitating the dehydration process alongside polyphosphoric acid, preventing the formation of side products.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process has been validated from gram scale up to 100L pilot reactors with consistent yields of 86-88%. The use of recoverable solvents and heterogeneous catalysts makes it highly adaptable for metric-ton manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Hydroxy-1-indanone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent to production requires more than just chemical knowledge; it demands engineering excellence and unwavering commitment to quality. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the CN113248356A process are fully realized in our manufacturing suites. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of detecting trace impurities, guaranteeing that every batch of 4-hydroxy-1-indanone meets the exacting standards required for API synthesis. We understand that in the fast-paced pharmaceutical industry, time is money, and our optimized workflows are designed to accelerate your project timelines.
We invite global partners to leverage our technical expertise to secure their supply chains. By collaborating with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this innovative route can improve your bottom line. We encourage interested parties to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us handle the complexities of synthesis so you can focus on delivering life-saving therapies to the market, confident in the knowledge that your critical intermediates are sourced from a partner dedicated to innovation and reliability.
