Advanced Synthesis of Diphenylphosphoryl Mesityl Methanone for Commercial UV Curing Applications
Introduction to Next-Generation Photoinitiator Synthesis
The rapid expansion of the UV curing industry, particularly in coatings, inks, and 3D printing, demands photoinitiators that offer superior reactivity while adhering to increasingly stringent environmental regulations. Patent CN102875598A introduces a transformative methodology for synthesizing critical Type I photoinitiators, specifically (diphenylphosphoryl)(mesityl)methanone and its bis-analog (phenylphosphoryl)bis(mesitylmethanone). These compounds are pivotal for initiating free-radical polymerization in unsaturated systems, yet their traditional manufacturing routes have long been plagued by toxicity and waste issues. The disclosed innovation shifts the paradigm by utilizing 1,3,5-trimethyl-2-(trichloromethyl)benzene as a versatile key building block, effectively bypassing the need for hazardous acyl chlorides or toxic heavy metal oxidants. This strategic pivot not only enhances the chemical efficiency of the synthesis but also aligns with global green chemistry initiatives, offering a sustainable pathway for the mass production of high-performance optical materials.
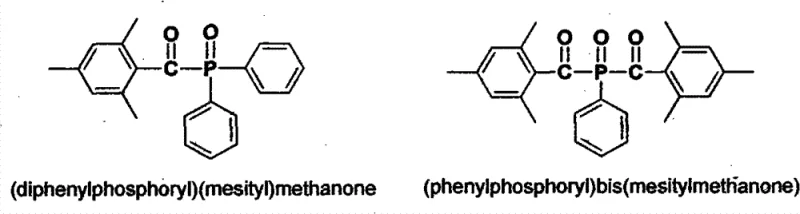
For R&D directors and procurement specialists seeking a reliable photoinitiator supplier, understanding the structural integrity and synthetic accessibility of these molecules is paramount. The patent details a robust framework where the core aromatic ketone structure is assembled through a novel condensation mechanism rather than traditional acylation. This approach minimizes the formation of difficult-to-remove side products, thereby streamlining the downstream purification processes which are often the most cost-intensive phase of fine chemical manufacturing. By securing a supply chain based on this advanced intellectual property, manufacturers can mitigate regulatory risks associated with heavy metal residues and chlorinated solvent emissions, ensuring a cleaner final product profile that meets the exacting standards of the electronics and healthcare sectors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of acyl phosphine oxides has relied heavily on two distinct but problematic pathways, both of which impose significant operational and environmental burdens. The first conventional route involves the Arbuzov reaction between mesityl benzoyl chloride and dialkyl phenylphosphonites; however, the synthesis of the requisite acyl chloride generates substantial quantities of corrosive hydrochloric acid and sulfur dioxide gas, creating severe corrosion issues for reactor vessels and necessitating expensive scrubbing systems. Furthermore, the subsequent Arbuzov step often employs volatile alkyl halides like methyl chloride or monochloroethane, which are potent ozone-depleting substances, forcing manufacturers to invest heavily in containment and recovery infrastructure to meet environmental compliance standards. The second traditional pathway utilizes mesityl benzaldehyde followed by addition with diphenylphosphine oxide and subsequent oxidation; critically, this oxidation step typically requires stoichiometric amounts of heavy metal catalysts such as molybdenum or tungsten salts, introducing the risk of toxic metal contamination in the final API or polymer additive, which is unacceptable for high-purity applications.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN102875598A leverages the unique reactivity of the trichloromethyl group to forge the carbon-phosphorus bond directly, eliminating the need for pre-formed acyl chlorides or toxic metal oxidants. By reacting 1,3,5-trimethyl-2-(trichloromethyl)benzene with organic phosphine precursors (such as diphenylphosphine salts), the process achieves a highly efficient condensation that proceeds under relatively mild conditions. This novel approach fundamentally alters the impurity profile of the reaction, as the byproducts are primarily inorganic salts and water-soluble species that can be easily removed via aqueous workup, rather than complex organic tars or heavy metal complexes. For a cost reduction in UV curing material manufacturing, this translates to a drastic simplification of the purification train, reducing solvent consumption and energy usage while simultaneously improving the overall atom economy of the synthesis.
Mechanistic Insights into Trichloromethyl-Mediated Condensation
The core chemical innovation lies in the nucleophilic substitution capability of the trichloromethyl moiety attached to the electron-rich mesitylene ring. In this mechanism, the phosphine precursor, activated as a nucleophilic salt (e.g., using Lithium, Sodium, or Potassium bases), attacks the electrophilic carbon of the trichloromethyl group. This step is kinetically favorable due to the strong electron-withdrawing nature of the three chlorine atoms, which stabilizes the transition state and facilitates the displacement of chloride ions. Unlike the sterically hindered addition to benzaldehyde derivatives, this substitution pathway is less sensitive to steric bulk, allowing for high conversion rates even with bulky diphenylphosphine groups. The resulting intermediate is a chloro-phosphine species, which retains the structural integrity of the mesityl ketone framework without requiring harsh acidic conditions that could lead to demethylation or ring degradation.
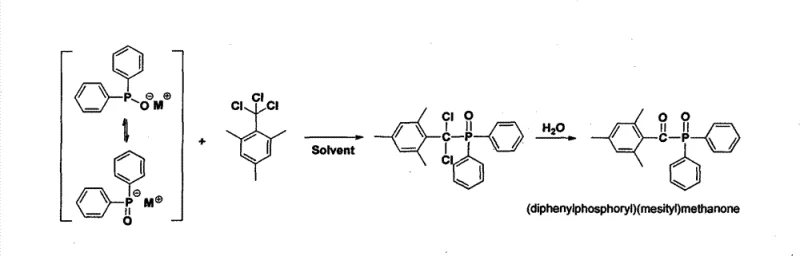
Following the condensation, the protocol employs a mild oxidative hydrolysis step to convert the trivalent phosphorus intermediate into the final pentavalent phosphine oxide. The patent specifies the use of benign oxidants such as hydrogen peroxide, air, or oxygen, which stand in sharp contrast to the hazardous chromates or permanganates used in older methodologies. This oxidation is chemoselective, targeting the phosphorus center exclusively while leaving the sensitive aromatic ketone and methyl substituents untouched. From an impurity control perspective, this selectivity is crucial; it prevents the formation of over-oxidized byproducts or ring-hydroxylated impurities that are notoriously difficult to separate via crystallization. The ability to perform this oxidation in situ, potentially without isolating the moisture-sensitive chloro-phosphine intermediate, further enhances the process safety and throughput, making it an ideal candidate for continuous flow manufacturing or large-batch processing.
How to Synthesize Diphenylphosphoryl Mesityl Methanone Efficiently
The practical execution of this synthesis involves a sequential workflow designed to maximize yield while minimizing exposure to hazardous reagents. The process begins with the preparation of the key electrophile, 1,3,5-trimethyl-2-(trichloromethyl)benzene, typically derived from the Friedel-Crafts alkylation of mesitylene with tetrachloroethylene, a reaction that itself is well-established and scalable. Subsequently, the phosphine precursor is generated in situ or added as a pre-formed salt into an anhydrous solvent system such as tetrahydrofuran (THF) or toluene. The condensation is allowed to proceed to completion, monitored via TLC or HPLC, before the introduction of the oxidant. The detailed standardized synthesis steps, including specific molar ratios, temperature profiles, and workup procedures validated by the patent examples, are outlined below to guide process engineers in replicating this high-efficiency route.
- Preparation of the key electrophile, 1,3,5-trimethyl-2-(trichloromethyl)benzene, via Friedel-Crafts alkylation of mesitylene.
- Condensation reaction between the trichloromethyl intermediate and a phosphine precursor salt (e.g., diphenylphosphine lithium salt) in anhydrous solvent.
- Oxidative hydrolysis of the resulting chloro-phosphine intermediate using hydrogen peroxide or air to yield the final phosphine oxide ketone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this trichloromethyl-based synthesis route offers profound strategic advantages that extend beyond simple chemical yield. By fundamentally redesigning the synthetic pathway to avoid regulated ozone-depleting substances and restricted heavy metals, manufacturers can significantly reduce the overhead costs associated with environmental compliance, waste disposal, and safety monitoring. The elimination of complex distillation steps required to separate volatile chlorinated byproducts means that capital expenditure on specialized corrosion-resistant equipment can be minimized, allowing for the use of standard glass-lined or stainless steel reactors. This simplification of the physical plant requirements directly contributes to a lower barrier to entry for production and enhances the flexibility of the supply chain to respond to market fluctuations.
- Cost Reduction in Manufacturing: The economic impact of removing heavy metal catalysts from the process cannot be overstated, as it eliminates the need for expensive metal scavenging resins and the associated loss of product yield during purification. Furthermore, the use of commodity-grade raw materials like mesitylene and tetrachloroethylene ensures that the input costs remain stable and predictable, shielding the production budget from the volatility often seen with specialized fine chemical reagents. The simplified workup procedure, which relies on basic aqueous extractions rather than complex chromatographic separations, drastically reduces solvent consumption and energy demand for solvent recovery, leading to substantial operational expenditure savings over the lifecycle of the product.
- Enhanced Supply Chain Reliability: Sourcing raw materials for this novel route is inherently more secure because the key starting materials are bulk petrochemical derivatives with established global supply networks, unlike specialized acyl chlorides which may have limited suppliers. The robustness of the reaction conditions, which tolerate a broader range of temperatures and do not require strictly anhydrous environments for every step, reduces the risk of batch failures due to minor deviations in operating parameters. This operational resilience ensures consistent delivery schedules and reduces the likelihood of supply disruptions caused by equipment maintenance or regulatory shutdowns associated with more hazardous legacy processes.
- Scalability and Environmental Compliance: The scalability of this process is supported by its exothermic profile, which is manageable in large-scale reactors without the need for cryogenic cooling, thereby facilitating a smooth transition from pilot plant to commercial tonnage production. From an environmental standpoint, the absence of sulfur dioxide and hydrochloric acid gas emissions during the key bond-forming step simplifies the permitting process for new manufacturing facilities and reduces the liability associated with fugitive emissions. The generation of benign inorganic salts as the primary waste stream allows for easier treatment and disposal, aligning the manufacturing footprint with the sustainability goals of downstream customers in the automotive and electronics industries.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aiming to clarify the feasibility and benefits for potential licensees or manufacturing partners. Understanding these nuances is essential for evaluating the technology's fit within existing production portfolios and for assessing the long-term value proposition of switching to this greener synthetic platform.
Q: What are the primary environmental advantages of this new synthesis route?
A: Unlike traditional methods that rely on heavy metal oxidants (like Molybdenum or Tungsten salts) or generate significant hydrochloric acid and sulfur dioxide gas, this novel route utilizes mild oxidation conditions and avoids ozone-depleting chlorinated byproducts, significantly reducing environmental compliance burdens.
Q: How does the use of 1,3,5-trimethyl-2-(trichloromethyl)benzene improve product purity?
A: The trichloromethyl group serves as a highly reactive electrophile that allows for direct condensation without the steric hindrance issues often encountered in long-chain alcohol Arbuzov reactions. This leads to more complete conversion and simplifies the removal of impurities, resulting in a higher purity profile suitable for sensitive optical applications.
Q: Is this process scalable for industrial production of photoinitiators?
A: Yes, the process relies on commodity raw materials like mesitylene and tetrachloroethylene, and the reaction conditions (ambient to moderate temperatures, standard solvents like THF or Toluene) are easily adaptable to large-scale reactors, ensuring robust supply chain continuity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diphenylphosphoryl Mesityl Methanone Supplier
As the global demand for high-performance photoinitiators continues to surge, NINGBO INNO PHARMCHEM stands ready to leverage this cutting-edge synthesis technology to deliver superior products to the market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volume requirements of major multinational corporations without compromising on quality. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of (diphenylphosphoryl)(mesityl)methanone meets the exacting standards required for optical radiation curing applications.
We invite R&D directors and procurement leaders to contact our technical procurement team to discuss how this innovative route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential economic benefits specific to your operation. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate how our commitment to green chemistry and process excellence can drive value for your organization.
