Scaling Advanced 1,2-Azaborine Heterocycles for Next-Generation Pharmaceutical and Material Applications
The landscape of modern medicinal chemistry and materials science is undergoing a paradigm shift with the emergence of boron-nitrogen (BN) isosteres as viable alternatives to traditional carbon-carbon (CC) aromatic systems. Patent CN102482293A details a groundbreaking methodology for the preparation of substituted 1,2-azaborine heterocycles, a class of compounds that uniquely combines aromaticity with the distinct electronic characteristics of the inorganic B-N unit. For R&D directors and procurement strategists in the pharmaceutical and fine chemical sectors, this technology represents a critical opportunity to access novel chemical space that was previously difficult or impossible to explore. The patent outlines robust synthetic pathways that allow for the selective functionalization of the azaborine ring at positions 1, 2, 3, 5, and 6, thereby creating a versatile library of intermediates suitable for high-value applications ranging from next-generation API development to advanced electronic materials.
As a reliable pharmaceutical intermediate supplier, understanding the nuances of these synthetic routes is essential for integrating them into existing supply chains. The disclosed methods leverage well-established organic transformations, such as electrophilic aromatic substitution (EAS) and transition-metal catalyzed cross-couplings, ensuring that the transition from bench-scale discovery to commercial manufacturing is seamless. By replacing specific C=C bonds with isoelectronic B-N units, chemists can modulate the electronic density, metabolic stability, and binding affinity of drug candidates without significantly altering their steric profile. This capability is particularly relevant for developing safer analogs of existing drugs, such as avoiding the toxic quinone metabolites associated with acetaminophen, thus addressing a major pain point in drug safety and regulatory approval processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of BN-containing heterocycles has been plagued by significant challenges that hindered their widespread adoption in industrial settings. Traditional methods often suffered from poor regioselectivity, making it difficult to introduce specific substituents at desired positions on the ring without generating complex mixtures of isomers that are costly and time-consuming to separate. Furthermore, many prior art routes relied on harsh reaction conditions or unstable precursors that decomposed before the final product could be isolated, leading to low overall yields and inconsistent batch quality. The lack of a general, modular synthetic strategy meant that each new derivative required a bespoke and often inefficient synthetic campaign, drastically increasing the cost of goods sold (COGS) and extending lead times for high-purity pharmaceutical intermediates. Additionally, the sensitivity of the B-N bond to hydrolysis and oxidation in earlier generations of compounds posed significant storage and handling risks, complicating logistics for global supply chain heads who require stable, long-shelf-life inventory.
The Novel Approach
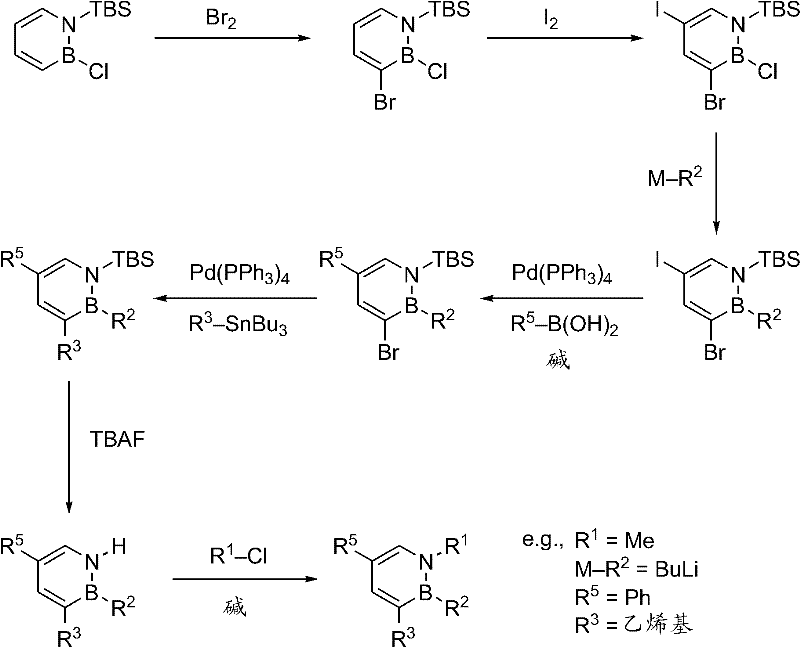
The methodology presented in CN102482293A overcomes these historical barriers through a strategic combination of protecting group chemistry and sequential functionalization. The core innovation lies in the use of N-protected 1,2-azaborine precursors that serve as stable platforms for further derivatization. By employing electrophilic aromatic substitution, specifically bromination and iodination, the process allows for the precise installation of leaving groups at the C(3) and C(5) positions. This creates a 'versatile intermediate' capable of undergoing diverse downstream transformations. Unlike conventional approaches that might struggle with the electron-deficient nature of the boron-containing ring, this novel route exploits the unique reactivity of the system to achieve high selectivity. The ability to subsequently displace these halogen handles via Suzuki, Stille, or nucleophilic substitution reactions provides a modular toolkit for constructing complex molecular architectures. This flexibility ensures cost reduction in pharmaceutical intermediate manufacturing by minimizing waste and maximizing the utility of a single common precursor for multiple end-products.
Mechanistic Insights into Electrophilic and Nucleophilic Substitution on BN-Rings
A deep dive into the reaction mechanisms reveals why this platform is so powerful for R&D teams aiming to optimize lead compounds. The presence of the boron atom fundamentally alters the electron distribution of the six-membered ring compared to benzene. The patent highlights that the boron center, when protected or substituted appropriately, can activate adjacent positions for electrophilic attack, a behavior that is counter-intuitive for typical electron-deficient heterocycles. Specifically, the sequential EAS reactions described allow for the orthogonal installation of halogens. Following this, the boron-chlorine bond itself becomes a site for nucleophilic attack. When treated with strong nucleophiles such as organolithium or Grignard reagents, the B-Cl bond undergoes displacement to form a new B-C or B-heteroatom bond. This dual reactivity—electrophilic substitution on the carbon framework and nucleophilic substitution at the boron center—creates a three-dimensional vector for diversification that is unavailable in all-carbon systems.
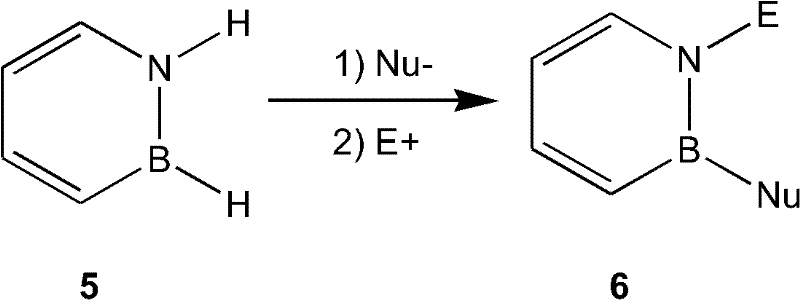
Furthermore, the patent elucidates a unique aromatic nucleophilic substitution pathway for the parent 1,2-dihydro-1,2-azaborine. In this mechanism, the ring reacts with a nucleophile (Nu-) to form an intermediate anionic species, which is then quenched with an electrophile (E+) to yield the substituted product. This one-pot transformation is particularly valuable because it allows for the simultaneous introduction of two different substituents on the nitrogen and boron atoms, respectively. The reaction tolerates a wide range of nucleophiles, including oxygen-based species like tert-butoxide and various carbon-based nucleophiles (sp3, sp2, and sp hybridized). This broad substrate scope implies that impurity profiles can be tightly controlled by selecting reagents that minimize side reactions, ensuring the high purity required for clinical-grade materials. The mechanistic understanding provided here empowers process chemists to predict reactivity trends and troubleshoot potential bottlenecks during scale-up.
How to Synthesize Substituted 1,2-Azaborine Efficiently
The synthesis of these advanced heterocycles is designed to be operationally simple while maintaining high chemical fidelity. The process begins with the preparation of the N-protected azaborine core, which can be achieved through ring-closing metathesis (RCM) of diene precursors or hydroboration strategies. Once the core is established, the functionalization sequence proceeds through the halogenation steps mentioned previously. It is crucial for laboratory personnel to maintain anhydrous conditions during the nucleophilic displacement steps to prevent hydrolysis of the reactive boron intermediates. The detailed standardized synthesis steps below outline the precise stoichiometry and conditions required to replicate the high yields reported in the patent literature, ensuring reproducibility across different manufacturing sites.
- Initiate the synthesis by treating N-protected 1,2-azaborine starting materials with bromine (Br2) to achieve selective C(3) bromination via electrophilic aromatic substitution.
- Perform subsequent iodination using I2 to generate a C(5) iodinated versatile intermediate, allowing for diverse functionalization at multiple ring positions.
- Execute nucleophilic displacement on the boron-chlorine bond using organolithium or Grignard reagents, followed by palladium-catalyzed cross-coupling to introduce desired aryl or alkyl substituents.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this 1,2-azaborine technology offers tangible strategic benefits beyond mere chemical novelty. The primary advantage lies in the modularity of the synthesis, which directly translates to supply chain resilience. Because a single versatile intermediate can be diverted into multiple distinct final products through late-stage functionalization, manufacturers can hold stock of the common precursor rather than maintaining large inventories of numerous finished SKUs. This consolidation significantly reduces warehousing costs and minimizes the risk of obsolescence. Furthermore, the reliance on commodity reagents such as bromine, iodine, and standard palladium catalysts ensures that raw material sourcing remains stable and unaffected by the supply volatility often associated with exotic specialty chemicals. This stability is critical for maintaining continuous production schedules and meeting the rigorous delivery timelines demanded by global pharmaceutical clients.
- Cost Reduction in Manufacturing: The streamlined synthetic route eliminates the need for multi-step protection and deprotection sequences that are common in traditional heterocycle synthesis. By utilizing direct electrophilic substitution and efficient cross-coupling reactions, the overall step count is reduced, which inherently lowers labor, energy, and solvent consumption. Additionally, the high selectivity of the reactions minimizes the formation of difficult-to-remove impurities, reducing the burden on downstream purification processes such as chromatography or recrystallization. This efficiency drives down the cost per kilogram of the final active pharmaceutical ingredient (API) intermediate, providing a competitive edge in pricing negotiations without compromising on quality standards.
- Enhanced Supply Chain Reliability: The robustness of the described chemistry means that the process is less susceptible to minor fluctuations in reaction parameters, leading to consistent batch-to-batch quality. This reliability is paramount for supply chain heads who must guarantee the continuity of supply for critical drug substances. The ability to scale these reactions from gram to kilogram scales using standard glass-lined or stainless steel reactors ensures that the technology is ready for immediate commercial deployment. Moreover, the stability of the N-protected intermediates allows for safer transportation and longer storage periods, mitigating the risks associated with the degradation of sensitive chemical assets during transit.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, the process offers significant advantages. The avoidance of highly toxic reagents and the potential for solvent recycling align with modern green chemistry principles. The high atom economy of the coupling reactions ensures that waste generation is minimized, simplifying waste treatment protocols and reducing disposal costs. As regulatory bodies worldwide tighten restrictions on chemical manufacturing emissions, adopting a cleaner, more efficient synthetic route future-proofs the supply chain against evolving compliance requirements. This proactive approach to environmental stewardship enhances the corporate reputation of the manufacturer and facilitates smoother regulatory audits.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of 1,2-azaborine chemistry in industrial settings. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for decision-makers evaluating this technology for their portfolios.
Q: What are the primary advantages of 1,2-azaborine isosteres over traditional carbon-based aromatics in drug design?
A: 1,2-Azaborine isosteres offer unique electronic properties and metabolic stability compared to their all-carbon benzene analogs. Specifically, the boron-nitrogen unit can prevent the formation of toxic quinone metabolites, as seen in acetaminophen analogs, while maintaining similar steric profiles for receptor binding.
Q: Is the synthesis of these BN-heterocycles scalable for commercial production?
A: Yes, the disclosed synthetic routes utilize standard industrial reagents such as bromine, iodine, and common organometallic species (BuLi, Grignard). The processes avoid exotic catalysts where possible and rely on robust reactions like electrophilic aromatic substitution and Suzuki/Stille couplings, facilitating scale-up from kilogram to metric ton quantities.
Q: How does the BN-substitution affect the optical properties of conjugated materials?
A: Incorporating the BN unit into conjugated systems, such as polyphenyls or polypyridines, typically results in a significant red-shift in absorption spectra and increased molar absorptivity. This makes them highly valuable for optoelectronic applications like OLEDs and solar energy conversion materials.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2-Azaborine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of BN-isosteres in the development of next-generation therapeutics and advanced materials. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from discovery to market is seamless. We are committed to delivering high-purity 1,2-azaborine intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom synthesis of novel analogs or large-scale supply of established intermediates, our infrastructure is designed to support your most demanding projects with speed and precision.
We invite you to collaborate with us to unlock the full potential of boron-nitrogen chemistry for your applications. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can drive value and innovation in your supply chain.
