Advanced Photocatalytic Synthesis of Deuterated Amino Acid Esters for Commercial Scale-up
The pharmaceutical industry is constantly seeking innovative synthetic routes to enhance the metabolic stability and efficacy of drug candidates, and the technology disclosed in patent CN114751801B represents a significant breakthrough in this domain. This patent introduces a novel method for preparing deuterated amino acid esters through photocatalysis, utilizing deuterium water as a cost-effective deuterium source under mild reaction conditions. Deuterated compounds are increasingly critical in modern drug development due to the kinetic isotope effect, where the stronger carbon-deuterium bond slows down metabolic clearance, thereby prolonging the half-life of the drug and reducing the frequency of administration. The traditional synthesis of such compounds often involves complex multi-step procedures with expensive reagents, but this new photocatalytic approach streamlines the process by directly deuterating amino acid esters using a sophisticated composite catalyst system. This innovation not only simplifies the synthetic pathway but also opens new avenues for the efficient production of high-value deuterated intermediates essential for clinical research and commercial drug manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of deuterated amino acid derivatives has been plagued by significant technical and economic challenges that hinder widespread adoption in large-scale manufacturing. Conventional methods typically require the pre-synthesis of specific precursors such as imidates or dehydroamino acid esters, which adds unnecessary steps to the production line and increases the overall cost of goods. Furthermore, these traditional routes often rely on expensive reducing agents and harsh reaction conditions that can compromise the integrity of sensitive functional groups within the molecule. The need for specialized reagents and the generation of complex waste streams make these processes less attractive from both an environmental and a supply chain perspective. Additionally, controlling the site-selectivity of deuteration in conventional chemistry is notoriously difficult, often leading to mixtures of isotopologues that require rigorous and costly purification to meet the stringent purity standards required for pharmaceutical applications.
The Novel Approach
In stark contrast to the limitations of the past, the photocatalytic method described in the patent offers a streamlined and economically viable solution for producing deuterated amino acid esters. By employing a composite catalyst system consisting of a Lewis acid, tetraphenylphosphonium chloride, tetraphenylboron salt, borane derivatives, and aluminum oxide, the reaction proceeds efficiently at room temperature under ultraviolet light. This approach eliminates the need for pre-functionalized precursors, allowing for the direct use of readily available amino acid esters as starting materials. The mild conditions ensure that sensitive protecting groups and stereocenters remain intact, thereby preserving the optical purity of the final product. The use of deuterium water, which is significantly cheaper and more accessible than other deuterium sources, drastically reduces the raw material costs, making the commercial production of these high-value intermediates much more feasible for supply chain managers looking to optimize budgets without compromising quality.
Mechanistic Insights into Photocatalytic Deuteration
The core of this technological advancement lies in the synergistic action of the composite catalyst system which facilitates the activation of the carbon-hydrogen bond and its subsequent replacement with deuterium. The Lewis acid component plays a crucial role in coordinating with the substrate, increasing its electrophilicity and making the alpha-proton more susceptible to exchange. Simultaneously, the tetraphenylboron salts and borane derivatives act as electron donors and mediators in the photocatalytic cycle, absorbing UV energy to generate reactive species that drive the deuteration process. The aluminum oxide serves as a support and potentially as a co-catalyst, stabilizing the reaction intermediates and ensuring a smooth progression of the catalytic cycle. This multi-component catalyst design is engineered to maximize the efficiency of deuterium transfer from the heavy water solvent to the specific alpha-position of the amino acid ester, achieving high isotopic enrichment without the need for stoichiometric amounts of expensive deuterated reagents.
From an impurity control perspective, this mechanism offers distinct advantages over thermal or metal-catalyzed hydrogenation methods. The mild photocatalytic conditions minimize the formation of thermal degradation byproducts and prevent racemization, which is a common concern in amino acid chemistry. The selectivity of the reaction is inherently high due to the specific activation mode of the catalyst system, which targets the alpha-carbon adjacent to the ester group while leaving other potential reaction sites untouched. This high level of chemoselectivity simplifies the downstream purification process, as fewer side products are generated that would otherwise require complex chromatographic separation. For R&D directors, this means a cleaner reaction profile and a more robust process that can be reliably transferred from the laboratory to the pilot plant with minimal risk of unexpected impurity profiles emerging during scale-up.
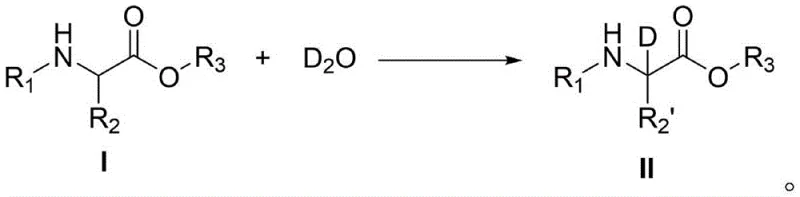
The versatility of this synthetic route is further demonstrated by its applicability to a wide range of amino acid ester substrates, including those with bulky side chains and various protecting groups. The patent data illustrates successful deuteration across different structural classes, confirming the robustness of the catalyst system against steric hindrance and electronic variations. This broad substrate scope is critical for pharmaceutical manufacturers who often need to produce libraries of deuterated analogs for structure-activity relationship studies. The ability to handle diverse substrates under the same general conditions simplifies process development and reduces the time required to optimize conditions for new molecules. Furthermore, the reaction can be tuned by adjusting the UV wavelength and power, providing an additional layer of control over the reaction kinetics and allowing for fine-tuning of the deuterium incorporation levels to meet specific regulatory or research requirements.
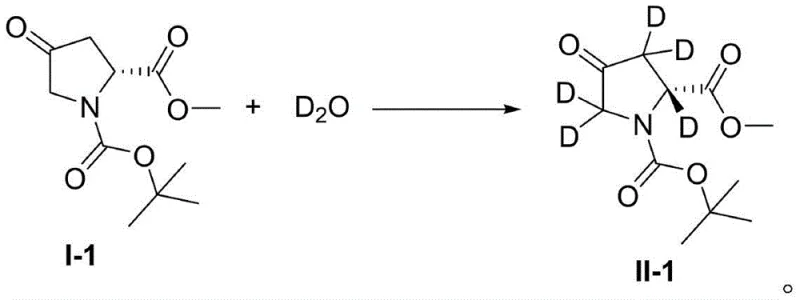
How to Synthesize Deuterated Amino Acid Esters Efficiently
The operational procedure for this synthesis is designed to be straightforward and compatible with standard chemical manufacturing equipment, facilitating easy adoption by production teams. The process begins with the dissolution of the amino acid ester compound in deuterium water, followed by the addition of the precise molar ratios of the composite catalyst components under a nitrogen atmosphere to prevent oxidation. The mixture is then subjected to UV irradiation at room temperature for a duration ranging from 0.5 to 24 hours, depending on the specific substrate and desired conversion level. Detailed standardized synthesis steps see the guide below.
- Dissolve the amino acid ester compound in deuterium water and add the composite catalyst system under nitrogen protection.
- Irradiate the mixture with UV light (265nm-400nm) at room temperature for 0.5 to 24 hours to facilitate deuteration.
- Filter the catalyst, extract the organic phase, and purify via column chromatography to obtain the high-purity deuterated product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photocatalytic technology translates into tangible strategic advantages that go beyond mere technical performance. The primary benefit lies in the substantial cost reduction achieved by replacing expensive deuterium sources and complex precursors with cheap deuterium water and simple amino acid esters. This shift in raw material strategy significantly lowers the entry barrier for producing deuterated intermediates, allowing companies to budget more effectively for their drug development pipelines. Moreover, the simplification of the synthetic route reduces the number of unit operations required, which in turn lowers labor costs, energy consumption, and waste disposal fees. These cumulative savings contribute to a more competitive cost structure, enabling companies to offer high-quality deuterated materials at more attractive price points in the global market.
- Cost Reduction in Manufacturing: The elimination of expensive reducing agents and the use of deuterium water as the primary deuterium source lead to a drastic reduction in raw material expenses. By avoiding the synthesis of specialized precursors like imidates, the process removes entire stages of production, thereby saving on reagents, solvents, and processing time. This streamlined approach ensures that the cost of goods sold is minimized, providing a significant margin improvement for commercial manufacturing operations. The efficiency of the catalyst system also means that lower catalyst loadings can be used while maintaining high yields, further contributing to the overall economic viability of the process.
- Enhanced Supply Chain Reliability: The reliance on readily available and commodity-grade chemicals such as amino acid esters and deuterium water ensures a stable and secure supply chain. Unlike specialized reagents that may have long lead times or single-source risks, the raw materials for this process are widely accessible from multiple global suppliers. This diversity in sourcing options mitigates the risk of supply disruptions and allows for more flexible inventory management. Additionally, the mild reaction conditions reduce the need for specialized high-pressure or high-temperature equipment, making it easier to find contract manufacturing organizations capable of executing the synthesis without significant capital investment.
- Scalability and Environmental Compliance: The room temperature operation and the use of UV light make this process inherently safer and easier to scale up compared to exothermic thermal reactions. The reduced energy footprint aligns with modern green chemistry principles, helping companies meet increasingly stringent environmental regulations and sustainability goals. The simplified workup procedure, involving basic filtration and extraction, minimizes the generation of hazardous waste and reduces the burden on waste treatment facilities. This environmental compatibility not only lowers compliance costs but also enhances the corporate social responsibility profile of the manufacturing entity, which is increasingly important for partnerships with major pharmaceutical clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic deuteration technology. These answers are derived directly from the patent specifications and are intended to provide clarity on the process capabilities and limitations. Understanding these details is crucial for making informed decisions about integrating this method into your existing production workflows or R&D programs.
Q: What are the advantages of this photocatalytic deuteration method over traditional methods?
A: This method utilizes cheap deuterium water as the deuterium source and operates under mild room temperature conditions, avoiding the need for expensive precursors like imidates or harsh reducing agents required in conventional synthesis.
Q: What is the deuterium incorporation efficiency of this process?
A: The patent data indicates a high deuterium incorporation rate, often exceeding 93% to 99% depending on the specific substrate, ensuring high isotopic purity for metabolic studies.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process uses readily available raw materials, operates at room temperature without high pressure, and involves simple workup procedures, making it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Deuterated Amino Acid Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this photocatalytic technology and are fully equipped to leverage it for your commercial needs. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to market supply is seamless. Our facilities are designed to handle complex photochemical reactions with precision, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest industry standards. We understand the critical nature of deuterated intermediates in drug development and are committed to delivering materials that support your regulatory filings and clinical trials without delay.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs through this advanced synthetic route. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating how this technology can improve your bottom line. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to evaluate the technical and commercial benefits firsthand. By partnering with us, you gain access to a reliable supply of high-purity deuterated amino acid esters that will accelerate your drug development timeline.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
