Advanced Synthesis of 3-Carboxybenzaldehyde: A Breakthrough in Commercial Manufacturing and Purity Control
The global demand for high-purity aromatic aldehydes continues to surge, driven by their critical role as building blocks in the synthesis of complex pharmaceutical agents and agrochemicals. Among these, 3-carboxybenzaldehyde (CAS 619-21-6) stands out as a particularly valuable yet historically difficult-to-source intermediate. Recent intellectual property disclosures, specifically patent CN110981719A, have illuminated a robust and economically viable pathway for its production. This technical insight report analyzes the novel methodology disclosed in the patent, which leverages m-methylbenzonitrile as a strategic starting material. By circumventing the regio-selectivity challenges associated with traditional xylene oxidation, this new route offers a compelling value proposition for R&D directors seeking reliable supply chains and procurement managers aiming for significant cost optimization in fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of meta-substituted benzaldehydes has been plagued by inherent chemical challenges that hinder efficient commercial scale-up. Traditional routes often rely on the oxidation of dimethylbenzenes (xylenes), a process that suffers from poor regio-selectivity, yielding complex mixtures of ortho, meta, and para isomers that are energetically expensive to separate. Furthermore, direct oxidation methods frequently require harsh conditions involving heavy metal catalysts or aggressive oxidants, which can lead to over-oxidation of the aldehyde group to the corresponding carboxylic acid, thereby drastically reducing yield. These inefficiencies translate directly into supply chain volatility and inflated costs, creating a bottleneck for downstream manufacturers who require consistent, high-purity inputs for sensitive drug synthesis applications.
The Novel Approach
The methodology outlined in patent CN110981719A represents a paradigm shift by utilizing m-methylbenzonitrile as the foundational feedstock. This strategic choice exploits the directing effects of the nitrile group to ensure perfect meta-substitution from the outset, effectively eliminating the isomer separation nightmare. The process flows through a logical sequence of hydrolysis, photo-chlorination, and a Sommelet-type oxidation, each step optimized for maximum conversion and minimal byproduct formation. By decoupling the introduction of the carboxyl and aldehyde functionalities through distinct, controlled reaction environments, this approach achieves superior purity profiles. The use of m-tolunitrile, a low-cost and widely available commodity, further anchors the economic feasibility of this route, making it an ideal candidate for reliable pharmaceutical intermediate supplier networks.
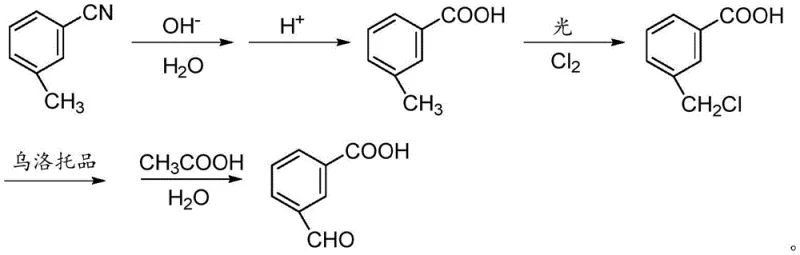
Mechanistic Insights into the Multi-Step Synthesis Strategy
The core of this synthetic innovation lies in its meticulous control over reaction mechanisms to preserve functional group integrity. The initial step involves the alkaline hydrolysis of the nitrile group to a carboxylic acid, typically conducted at elevated temperatures between 100°C and 110°C. This transformation is quantitative and robust, converting the lipophilic nitrile into the more polar m-methylbenzoic acid, which is easily isolated via acidification. Subsequently, the methyl group undergoes free-radical chlorination under light irradiation in a solvent like carbon tetrachloride. This photochemical activation allows for the selective substitution of benzylic hydrogens with chlorine atoms without affecting the aromatic ring or the carboxyl group, generating the crucial 3-carboxyl benzyl chloride intermediate with high fidelity.
The final transformation utilizes the classic Sommelet reaction mechanism, where the benzyl chloride intermediate reacts with urotropine (hexamethylenetetramine) to form a quaternary ammonium salt. This salt is then subjected to hydrolysis using a mixture of glacial acetic acid and water. The choice of glacial acetic acid is particularly astute; compared to strong mineral acids, it provides a milder acidic environment that facilitates the decomposition of the quaternary salt into the desired aldehyde while suppressing side reactions such as polymerization or further oxidation. This gentle hydrolysis step is critical for achieving the reported purity levels of over 99%, ensuring the final product meets the stringent specifications required for high-purity OLED material or API intermediate applications.
How to Synthesize 3-Carboxybenzaldehyde Efficiently
Implementing this synthesis route requires precise adherence to the reaction parameters defined in the patent to maximize yield and safety. The process is designed to be operationally simple, avoiding the need for exotic reagents or cryogenic conditions, which facilitates easier technology transfer from lab to plant. Operators must carefully control the stoichiometry of the chlorination step to prevent poly-chlorination and manage the exothermic nature of the hydrolysis reactions. For a detailed breakdown of the specific mass ratios, temperature profiles, and workup procedures necessary to replicate these results, please refer to the standardized protocol below.
- Hydrolyze m-methylbenzonitrile with aqueous alkali at 100-110°C, followed by acidification to isolate m-methylbenzoic acid.
- Perform radical chlorination on m-methylbenzoic acid using chlorine gas under light irradiation in carbon tetrachloride to form 3-carboxyl benzyl chloride.
- React 3-carboxyl benzyl chloride with urotropine, followed by hydrolysis with glacial acetic acid and water to yield the final aldehyde product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process addresses several critical pain points that typically burden the procurement of specialized aromatic intermediates. The shift to m-methylbenzonitrile as a starting material immediately lowers the baseline cost of goods sold, as it is a bulk chemical with a stable market price compared to specialized oxidized derivatives. Furthermore, the simplicity of the unit operations—hydrolysis, chlorination, and filtration—reduces the capital expenditure required for reactor setups and minimizes the operational complexity for manufacturing teams. This streamlined workflow not only accelerates production cycles but also enhances the overall reliability of the supply chain by reducing the number of potential failure points during batch processing.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of recyclable solvents like carbon tetrachloride and chloroform significantly lower the variable costs associated with production. By avoiding complex purification steps such as column chromatography and relying instead on crystallization and filtration, the process drastically simplifies the downstream processing requirements. This efficiency translates into substantial cost savings that can be passed down the value chain, offering a competitive pricing structure for cost reduction in fine chemical manufacturing without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reliance on commoditized raw materials ensures that production is not held hostage by the availability of niche reagents. Since m-methylbenzonitrile and urotropine are produced at massive scales globally, the risk of supply disruption is minimized. Additionally, the robustness of the reaction conditions means that batches are less likely to fail due to minor fluctuations in temperature or pressure, leading to more predictable lead times. This stability is crucial for reducing lead time for high-purity pharmaceutical intermediates, allowing downstream partners to maintain leaner inventory levels with confidence.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up of complex aromatic aldehydes, with specific provisions for tail gas treatment during the chlorination step to meet environmental regulations. The ability to recover and reuse solvents further aligns the process with green chemistry principles, reducing the volume of hazardous waste generated. This environmental compliance not only mitigates regulatory risk but also future-proofs the manufacturing site against tightening emission standards, ensuring long-term operational continuity.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis method is essential for stakeholders evaluating its adoption for commercial production. The following questions address common inquiries regarding the scalability, purity, and economic viability of the process described in patent CN110981719A. These answers are derived directly from the experimental data and beneficial effects reported in the documentation, providing a clear picture of what partners can expect when integrating this technology into their supply chains.
Q: What is the primary advantage of using m-tolunitrile as a starting material?
A: Using m-tolunitrile allows for precise meta-substitution control and utilizes a low-cost, readily available commodity chemical, significantly reducing raw material expenses compared to traditional xylenes oxidation routes.
Q: How does this method ensure high product purity?
A: The process employs mild hydrolysis conditions using glacial acetic acid in the final step, which minimizes byproduct formation and ensures the generation of white needle-like crystals with purity exceeding 99%.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the patent explicitly states the method features simple operation, safe reaction conditions, and the ability to recycle solvents, making it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Carboxybenzaldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of securing a stable supply of high-value intermediates like 3-carboxybenzaldehyde. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full manufacturing is seamless and efficient. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch meets the exacting standards required for pharmaceutical and electronic material applications. Our infrastructure is designed to handle the specific safety and environmental requirements of chlorination and oxidation chemistries safely and responsibly.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific project needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, ensuring that our capabilities align perfectly with your development timelines and quality expectations.
