Advanced Synthesis of Alkoxyethyl-2-Acetamido-2-Deoxy-α-D-Glucopyranosides for Commercial Scale-Up
The chemical industry is currently witnessing a significant paradigm shift towards bio-based amphiphilic molecules that combine high performance with environmental sustainability. A pivotal development in this sector is detailed in Chinese Patent CN107011395B, which discloses a novel class of N-acetylglucosamine compounds characterized by the general structure shown in Formula (I). These alkoxyethyl-2-acetamido-2-deoxy-α-D-glucopyranosides represent a strategic evolution in surfactant design, specifically engineered to overcome the inherent solubility limitations of traditional alkyl-N-acetylglucosamines. By incorporating an alkoxyethyl linker, the invention successfully modulates the hydrophilic-lipophilic balance (HLB), enabling the creation of surfactants that maintain excellent water solubility even with longer hydrophobic chains. This structural innovation is not merely academic; it opens up vast commercial opportunities for reliable surfactant supplier networks seeking to offer high-purity specialty chemicals for pharmaceutical, cosmetic, and agrochemical applications. The ability to tune the alkyl chain length (where n ranges from 0 to 11) provides a versatile platform for cost reduction in fine chemical manufacturing, allowing formulators to select the precise surface activity required for diverse end-use products without compromising on solubility or stability.
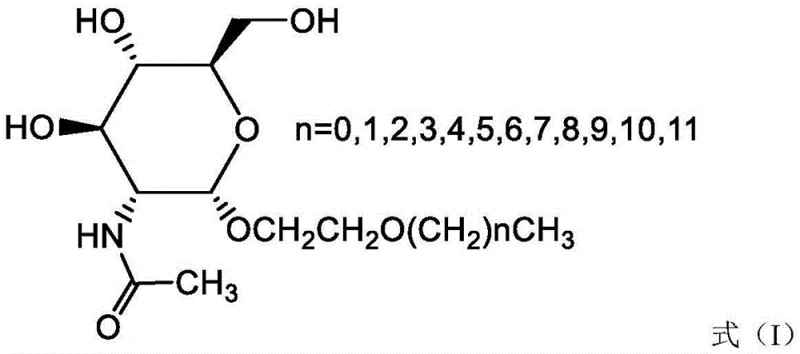
The development of these compounds addresses a critical bottleneck in the commercialization of sugar-based surfactants. Conventional alkyl-N-acetyl-D-glucosides, while biologically active, suffer from a severe drawback: their water solubility drops precipitously when the alkyl chain carbon number reaches or exceeds eight. This physical limitation restricts their utility in aqueous formulations, particularly in industries requiring high concentrations of active ingredients. The traditional approach often necessitates complex solubilization strategies or the use of co-solvents, which can introduce toxicity concerns or increase formulation costs. In contrast, the novel approach described in the patent utilizes an alkoxyethyl group (ROCH2CH2-) to replace the direct alkyl attachment. This ether linkage acts as a hydrophilic spacer, effectively decoupling the hydrophobic tail from the polar headgroup. Consequently, the resulting molecules exhibit significantly improved water solubility and superior surface-active properties, including reduced surface tension and enhanced foaming stability. This structural modification transforms a niche biochemical reagent into a robust industrial surfactant capable of functioning as a reliable agrochemical intermediate or a key component in personal care formulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of glycosylated surfactants has been plagued by challenges related to stereocontrol and solubility management. Traditional Fischer glycosylation methods often yield anomeric mixtures that are difficult to separate, leading to products with inconsistent physical properties. Furthermore, when attempting to synthesize alkyl-N-acetylglucosamines with longer hydrophobic tails to enhance emulsification power, chemists encounter the "solubility cliff." As the hydrophobic character increases, the molecule loses its ability to interact favorably with water, rendering it useless for many aqueous applications. This limitation forces manufacturers to rely on petrochemical-derived surfactants or less efficient sugar derivatives that require harsh processing conditions. The reliance on non-renewable feedstocks and the generation of difficult-to-treat wastewater streams further exacerbate the environmental footprint of conventional surfactant manufacturing. Additionally, the purification of these conventional glycosides often requires energy-intensive processes, driving up the operational expenditure and limiting the scalability of the production process for high-purity pharmaceutical intermediates.
The Novel Approach
The methodology outlined in the patent introduces a sophisticated three-step synthetic route that elegantly bypasses these historical constraints. By employing a Lewis acid-catalyzed glycosylation followed by a selective protection-deprotection strategy, the process ensures high stereochemical purity, specifically favoring the alpha-anomer which is often more stable and biologically relevant. The introduction of the alkoxyethyl moiety is the cornerstone of this innovation, providing a tunable hydrophilic domain that compensates for the hydrophobicity of the alkyl chain. This allows for the synthesis of a homologous series of compounds (from methoxy to dodecyloxy derivatives) that retain excellent solubility profiles. The process utilizes readily available starting materials, such as N-acetyl-D-glucosamine derived from chitin, aligning with green chemistry principles. This approach not only enhances the functional performance of the surfactant—evidenced by lower critical micelle concentrations (CMC) and superior emulsification of oils like rapeseed oil—but also streamlines the supply chain by utilizing biomass-derived feedstocks. The result is a commercially viable pathway for producing advanced surfactants that meet the rigorous demands of modern industrial applications.
Mechanistic Insights into BF3-Catalyzed Glycosylation and Protection
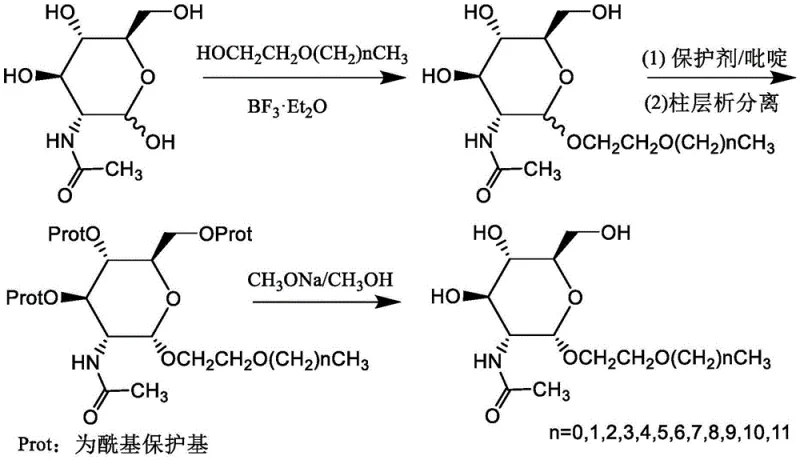
The core of this synthesis lies in the precise manipulation of carbohydrate chemistry to achieve the desired 1,2-cis glycosidic linkage. The first step involves the reaction of N-acetyl-D-glucosamine with various alkoxyethanols in the presence of boron trifluoride diethyl etherate (BF3·Et2O). This Lewis acid catalyst activates the anomeric center of the sugar, facilitating the nucleophilic attack by the alcohol. The reaction is typically conducted in polar aprotic solvents like acetonitrile or nitromethane at elevated temperatures ranging from 50°C to 150°C. This thermal energy is crucial for overcoming the activation barrier and ensuring complete conversion, although it initially yields a mixture of alpha and beta anomers. The mechanistic pathway likely proceeds through an oxocarbenium ion intermediate, where the neighboring group participation of the C2-acetamido group plays a subtle role in directing stereoselectivity, although the initial kinetic product is often a mixture. The subsequent steps are designed to refine this mixture into a single, high-purity isomer.
To isolate the desired alpha-anomer, the crude mixture undergoes a protection step using acetic anhydride in pyridine. This acetylation masks the free hydroxyl groups at positions 3, 4, and 6, increasing the lipophilicity of the molecule and facilitating separation via column chromatography. The difference in polarity between the alpha and beta acetylated intermediates allows for effective purification. Once the pure alpha-protected intermediate is secured, the final step involves a Zemplén deacetylation. Treatment with sodium methoxide in methanol selectively cleaves the ester bonds at the 3, 4, and 6 positions without affecting the stable glycosidic bond or the N-acetyl group. This mild basic condition ensures that the delicate sugar backbone remains intact while restoring the hydrophilic hydroxyl groups necessary for surfactant activity. The entire sequence demonstrates a high degree of chemical control, minimizing side reactions and ensuring that the final product meets stringent purity specifications required for sensitive applications like membrane protein extraction.
How to Synthesize Alkoxyethyl N-Acetylglucosamine Efficiently
The synthesis of these high-value surfactants requires strict adherence to the optimized reaction conditions to ensure reproducibility and yield. The process begins with the careful selection of the alkoxyethanol chain length to match the desired HLB value, followed by the catalytic glycosylation under anhydrous conditions to prevent hydrolysis. The subsequent protection and deprotection steps must be monitored closely using TLC or HPLC to prevent over-reaction or degradation of the sugar moiety. While laboratory-scale examples utilize column chromatography for purification, commercial scale-up would likely transition to crystallization or continuous chromatography techniques to improve throughput. The detailed standardized synthesis steps, including specific molar ratios, solvent choices, and temperature profiles, are critical for achieving the high purity levels demanded by the pharmaceutical and cosmetic industries.
- Glycosylation of N-acetyl-D-glucosamine with alkoxyethanol using BF3·Et2O catalyst at elevated temperatures to form an alpha/beta mixture.
- Selective acetylation of the hydroxyl groups using acetic anhydride and pyridine, followed by chromatographic separation to isolate the alpha-anomer.
- Deprotection of the acetyl groups using sodium methoxide in methanol to yield the final high-purity alkoxyethyl glucoside surfactant.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this technology offers substantial strategic benefits beyond mere technical performance. The primary advantage lies in the raw material sourcing; N-acetyl-D-glucosamine is derived from chitin, which is the second most abundant biopolymer on Earth, sourced from seafood processing waste like shrimp and crab shells. This abundance translates into a highly stable and cost-effective supply chain, insulating manufacturers from the volatility associated with petrochemical feedstocks. Furthermore, the synthetic route avoids the use of expensive transition metal catalysts, relying instead on common Lewis acids and base reagents that are inexpensive and easy to handle. This simplification of the catalyst system eliminates the need for costly heavy metal removal steps, which are often a regulatory and financial burden in API manufacturing. The elimination of these complex purification stages results in significant cost savings and a streamlined production workflow.
- Cost Reduction in Manufacturing: The process leverages cheap, renewable biomass feedstocks and avoids precious metal catalysts, leading to a drastically simplified bill of materials. By removing the need for expensive transition metals and the associated downstream purification infrastructure, the overall production cost is substantially lowered. The high atom economy of the glycosylation step further contributes to waste reduction, minimizing disposal costs and enhancing the overall economic efficiency of the manufacturing process.
- Enhanced Supply Chain Reliability: Sourcing raw materials from the global seafood industry provides a diversified and resilient supply base that is less susceptible to geopolitical disruptions than oil-dependent supply chains. The chemical stability of the intermediates allows for flexible production scheduling and inventory management, ensuring consistent availability of the final surfactant products. This reliability is crucial for long-term contracts in the personal care and pharmaceutical sectors where supply continuity is paramount.
- Scalability and Environmental Compliance: The reaction conditions utilize standard organic solvents and reagents that are compatible with existing large-scale reactor infrastructure, facilitating easy commercial scale-up of complex surfactants. Moreover, the bio-based nature of the final product aligns with increasingly stringent environmental regulations and consumer demand for green chemistry solutions. The biodegradability of the sugar-based surfactant reduces the environmental impact of wastewater effluent, simplifying compliance with environmental discharge standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these novel glycosides. Understanding these aspects is essential for R&D teams evaluating the feasibility of integrating these surfactants into new formulations. The answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industrial decision-makers.
Q: How does the alkoxyethyl modification improve solubility compared to traditional alkyl glucosides?
A: The introduction of the hydrophilic oxyethyl fragment (-OCH2CH2-) between the hydrophobic alkyl chain and the sugar headgroup significantly enhances water solubility, overcoming the limitations of traditional alkyl-N-acetylglucosamines which become insoluble when the alkyl chain exceeds 8 carbons.
Q: What are the primary industrial applications for these novel surfactants?
A: These compounds serve as mild non-ionic surfactants for membrane protein extraction, emulsifiers in cosmetics and pharmaceuticals, and green additives in food processing, leveraging their biocompatibility and excellent foaming properties.
Q: Is the raw material N-acetyl-D-glucosamine sustainable for large-scale production?
A: Yes, N-acetyl-D-glucosamine is derived from chitin, the second most abundant natural polysaccharide found in crustacean shells and fungal cell walls, ensuring a renewable and cost-effective supply chain for manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alkoxyethyl N-Acetylglucosamine Supplier
As the demand for bio-based, high-performance surfactants continues to grow, partnering with an experienced CDMO is essential for successful commercialization. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory bench to industrial plant is seamless and efficient. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of alkoxyethyl-2-acetamido-2-deoxy-α-D-glucopyranoside meets the highest quality standards required for pharmaceutical and cosmetic applications. We understand the critical importance of consistency and reliability in the supply of fine chemical intermediates.
We invite you to collaborate with our technical team to explore how this patented technology can optimize your product portfolio. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to support your R&D and procurement strategies, ensuring you stay ahead in the competitive landscape of specialty surfactants.
