Revolutionizing Fragrance Synthesis with Novel Dioxotungsten(VI) Catalytic Systems
Revolutionizing Fragrance Synthesis with Novel Dioxotungsten(VI) Catalytic Systems
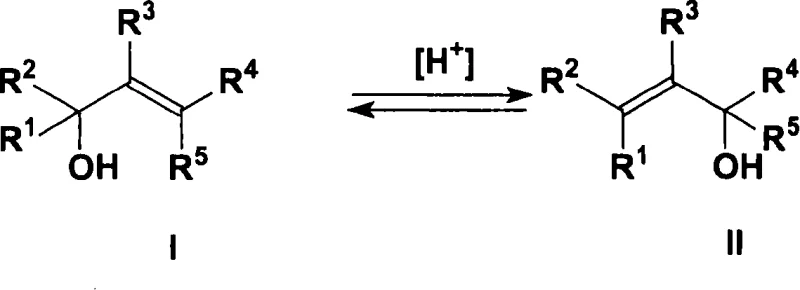
The landscape of fine chemical synthesis, particularly within the flavor and fragrance sector, is constantly evolving to meet stricter purity standards and efficiency demands. A pivotal advancement in this domain is documented in patent CN1304108C, which introduces a sophisticated method for the isomerization of allyl alcohols. This technology specifically targets the equilibrium transformation between primary or secondary allyl alcohols, such as geraniol and nerol, and their tertiary counterparts like linalool. The core innovation lies in the utilization of novel dioxotungsten(VI) complexes that operate effectively without the detrimental presence of halogens. For R&D directors and process engineers, this represents a significant leap forward, offering a pathway to high-purity intermediates essential for vitamin synthesis and premium perfume formulations. The ability to drive this equilibrium with enhanced selectivity and activity addresses long-standing challenges in terpene chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the isomerization of allyl alcohols has been fraught with chemical inefficiencies that compromise both yield and product quality. Traditional acid-catalyzed methods, while conceptually simple, are notoriously non-selective. These acidic conditions often trigger severe side reactions, including dehydration and cyclization, which generate complex impurity profiles that are difficult and costly to remove downstream. Furthermore, earlier attempts to utilize transition metal catalysts, such as molybdenum or vanadium compounds, presented their own set of operational hazards. Specifically, prior art tungsten catalysts often relied on tungsten oxytetrachloride as a precursor. This reliance introduced chlorine into the reaction system, leading to corrosive damage in stainless steel reactors and, more critically, leaving trace chloride residues in the final product. For applications in the fragrance and vitamin industries, where organoleptic properties and safety are paramount, such halogen contamination is unacceptable and necessitates expensive purification steps.
The Novel Approach
The methodology outlined in CN1304108C fundamentally reengineers the catalytic system to overcome these historical bottlenecks. By employing dioxotungsten(VI) complexes coordinated with aminoalcohols or aminophenols, the process achieves a halogen-free environment from the very inception of catalyst synthesis. This shift eliminates the risk of equipment corrosion and product contamination associated with chloride-based precursors. The new catalysts demonstrate superior activity, allowing the isomerization equilibrium to be reached more rapidly and at potentially optimized conditions compared to older tungsten alkoxide systems. Moreover, the use of ligands like 8-hydroxyquinoline stabilizes the tungsten center, enhancing the selectivity for the desired 1,3-hydroxyl migration while suppressing degradation pathways. This results in a cleaner reaction profile, significantly reducing the burden on downstream purification units and aligning perfectly with the needs of a reliable agrochemical intermediate supplier or fragrance manufacturer seeking cost reduction in manufacturing.
Mechanistic Insights into Dioxotungsten(VI) Catalyzed Isomerization
The mechanistic elegance of this transformation relies on the specific coordination chemistry of the tungsten(VI) center. The reaction corresponds to a 1,3-migration of the hydroxyl group coupled with an internal migration of the double bond. In the presence of the dioxotungsten(VI) complex, the metal center acts as a Lewis acid, coordinating with the allylic alcohol substrate to facilitate this rearrangement. The presence of bidentate ligands, such as aminoalcohols or aminophenols, creates a specific steric and electronic environment around the tungsten atom. This environment is crucial for stabilizing the transition state of the isomerization while preventing the catalyst from decomposing into inactive tungsten oxides, a common failure mode in previous generations of catalysts. The homogeneous nature of the catalyst ensures uniform interaction with the substrate, whether the reaction is conducted in bulk allyl alcohol or in organic solvents like toluene or xylene.
Impurity control is intrinsically linked to the halogen-free nature of the catalyst preparation. In conventional routes using tungsten oxytetrachloride, incomplete removal of chloride leads to the formation of chlorinated byproducts and promotes acid-catalyzed side reactions even after the metal is removed. By synthesizing the catalyst from tungstic acid and hydrogen peroxide, followed by ligand exchange, the new method ensures that no halogen species are introduced. This purity is critical when producing high-purity OLED material precursors or pharmaceutical intermediates where trace metals and halogens are strictly regulated. Additionally, the stability of the complex reduces the precipitation of tungsten oxide during the reaction, which not only maintains catalytic activity over longer periods but also prevents the physical fouling of reactor vessels and distillation columns, thereby enhancing overall process reliability.

How to Synthesize Allyl Alcohol Efficiently
The synthesis protocol described in the patent offers a robust framework for producing valuable tertiary allyl alcohols like linalool from readily available geraniol or nerol. The process begins with the in situ or ex situ preparation of the active dioxotungsten(VI) species, typically involving the reaction of tungstic acid with aqueous hydrogen peroxide to form an oxyperoxo intermediate, which is then complexed with 8-hydroxyquinoline. This catalyst solution is then introduced to the reactant alcohol stream. The reaction is typically conducted at elevated temperatures ranging from 150°C to 250°C, conditions that are manageable in standard industrial reactors. To drive the equilibrium towards the desired product, the process can be integrated with continuous distillation to remove the lower-boiling product or by using water scavengers to eliminate water formed by minor dehydration side reactions. Detailed standardized synthetic steps for implementing this technology follow below.
- Prepare the dioxotungsten(VI) complex by reacting tungstic acid with hydrogen peroxide and adding 8-hydroxyquinoline ligands.
- Mix the catalyst solution with the reactant allyl alcohol (e.g., geraniol) in a reactor equipped for heating up to 250°C.
- Heat the mixture to 150-250°C under inert atmosphere and monitor conversion, optionally removing water to shift equilibrium.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic technology translates into tangible operational efficiencies and risk mitigation. The elimination of halogenated precursors removes the need for specialized corrosion-resistant alloys in reactors and piping, allowing for the use of standard stainless steel equipment which significantly lowers capital expenditure. Furthermore, the absence of chloride contamination simplifies the quality control workflow, as extensive testing for halogen residues becomes less critical, and the risk of batch rejection due to off-spec impurity profiles is drastically reduced. This streamlined process flow enhances the overall throughput of the manufacturing facility, ensuring a more consistent supply of critical intermediates for the global market.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the catalyst synthesis and the reduction in downstream processing costs. By avoiding the multi-step, hazardous synthesis of tungsten oxytetrachloride, the raw material costs for catalyst preparation are significantly lowered. Additionally, the high selectivity of the new catalyst minimizes the formation of heavy ends and polymeric byproducts, which increases the overall yield of the valuable fragrance intermediate. This improvement in mass balance means that less raw material is wasted, and the energy consumption per kilogram of product is reduced due to simpler distillation requirements. The qualitative shift towards a cleaner reaction profile directly supports cost reduction in fine chemical manufacturing by lowering waste disposal fees and extending the lifecycle of processing equipment.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the availability of specialized, hazardous reagents. The new method utilizes widely available and stable starting materials such as tungstic acid, hydrogen peroxide, and 8-hydroxyquinoline. This reliance on commodity chemicals reduces the risk of supply disruptions associated with niche halogenated precursors. Moreover, the robustness of the catalyst system allows for longer campaign runs without frequent catalyst replacement or reactor cleaning, ensuring a steady output of product. This reliability is crucial for maintaining the production schedules of downstream customers in the vitamin and fragrance industries, who depend on just-in-time delivery of high-quality intermediates to meet their own market demands.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the halogen-free nature of the process offers substantial advantages. The absence of chlorine eliminates the generation of corrosive acidic waste streams and halogenated organic waste, simplifying effluent treatment and ensuring compliance with increasingly stringent environmental regulations. The process is amenable to both batch and continuous operation, providing the flexibility needed to scale from pilot plant trials to multi-ton commercial production seamlessly. The ability to operate without exotic solvents, often using the reactant alcohol itself as the solvent, further reduces the environmental footprint and VOC emissions, aligning with modern green chemistry principles and corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this isomerization technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this catalytic system into existing production lines.
Q: What are the advantages of the new tungsten catalyst over traditional acid catalysts?
A: Unlike traditional acid catalysts which promote unwanted side reactions like dehydration and cyclization, the novel dioxotungsten(VI) complexes offer high selectivity for the 1,3-hydroxyl migration without degrading the terpene structure.
Q: How does this method address chlorine contamination issues?
A: The patent describes a halogen-free preparation route using tungstic acid and peroxide, eliminating the need for tungsten oxytetrachloride and preventing corrosive chloride residues in the final fragrance product.
Q: Can this process be scaled for industrial production of linalool?
A: Yes, the process operates at standard industrial temperatures (150-250°C) and allows for continuous operation or batch processing with simple distillation workups, making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dioxotungsten(VI) Complex Supplier
The technological potential of halogen-free dioxotungsten(VI) catalysis represents a significant opportunity for manufacturers of high-value fragrance and vitamin intermediates. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are successfully translated into robust industrial processes. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch meets the exacting standards required by the global pharmaceutical and flavor industries. We understand the critical nature of catalyst performance in determining the overall economics of your synthesis.
We invite you to collaborate with us to optimize your production of allyl alcohol derivatives. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific facility and volume requirements. Please contact our technical procurement team to request specific COA data for our catalyst grades and to discuss route feasibility assessments for your target molecules. Together, we can enhance your supply chain resilience and drive down manufacturing costs through advanced catalytic solutions.
