Advanced Metal-Free Synthesis of Beta-Diaryl-Difluoroketones for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to incorporate fluorine atoms into complex organic scaffolds, driven by the unique metabolic stability and lipophilicity that fluorine imparts to drug candidates. Patent CN115028505B introduces a groundbreaking preparation method for β,β-di(hetero)aryl-α,α-difluoroketones, a class of compounds serving as critical building blocks in modern medicinal chemistry. This innovation addresses the longstanding challenge of constructing gem-difluoroketone motifs without relying on harsh conditions or scarce transition metal catalysts. By utilizing di(hetero)arylmethyltriphenylphosphonium salts and difluoroenol silyl ethers, the disclosed technology enables a direct fluoroalkylation reaction that proceeds under remarkably mild conditions. For R&D directors and procurement specialists alike, this represents a significant shift towards more sustainable and economically viable manufacturing processes for high-value fluorinated intermediates.
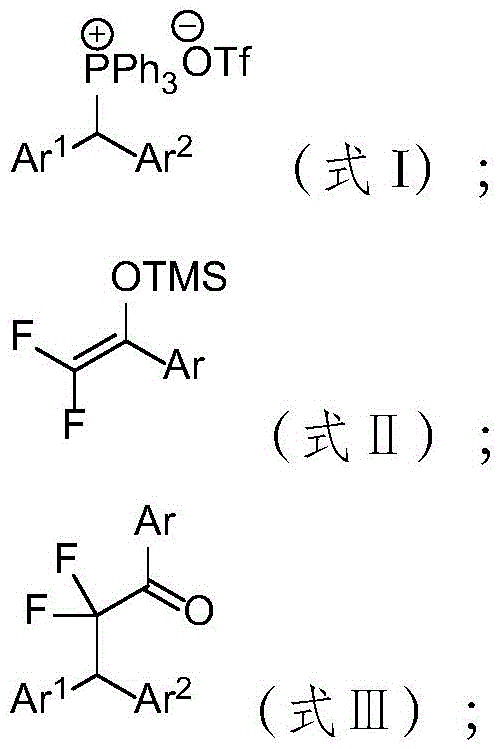
Traditionally, the synthesis of α,α-difluoroketones has often involved multi-step sequences requiring stoichiometric amounts of oxidants or precious metal catalysts, which introduce significant cost and purity burdens. Conventional methods frequently struggle with functional group compatibility, particularly when sensitive heterocycles are present in the molecular architecture. The limitations of these older technologies manifest as lower overall yields, complex waste streams containing heavy metals, and stringent safety requirements for handling reactive reagents. In contrast, the novel approach detailed in the patent leverages the inherent reactivity of phosphonium salts to drive the formation of the carbon-carbon bond directly. This eliminates the need for external catalysts or additives, thereby streamlining the workflow and reducing the potential for metal contamination in the final active pharmaceutical ingredient (API) intermediate.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, accessing gem-difluoroketone structures has required chemists to navigate a landscape of restrictive reaction parameters. Many established protocols necessitate the use of strong bases or cryogenic temperatures to control selectivity, which escalates energy consumption and operational complexity on a manufacturing scale. Furthermore, the reliance on transition metal catalysis, while effective for certain transformations, creates a bottleneck in the supply chain due to the volatility of metal prices and the rigorous regulatory scrutiny regarding residual metal levels in pharmaceutical products. The purification steps associated with removing these metal residues often involve specialized scavengers or chromatography, adding time and cost to the production timeline. Additionally, the substrate scope in traditional methods is often narrow, failing to accommodate the diverse array of electron-rich or electron-deficient aryl groups required for next-generation drug discovery programs.
The Novel Approach
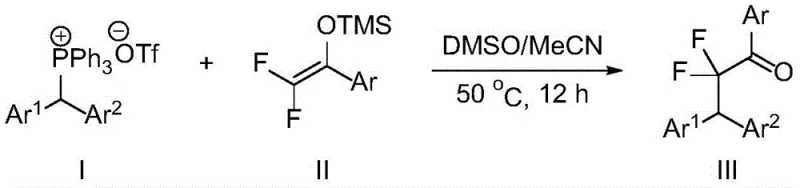
The methodology presented in CN115028505B circumvents these obstacles by employing a catalyst-free system that operates efficiently at moderate temperatures. The core of this innovation lies in the strategic pairing of di(hetero)arylmethyltriphenylphosphonium salts with difluoroenol silyl ethers (DFESEs). This combination facilitates a smooth fluoroalkylation process that tolerates a wide range of functional groups, including methoxy, halogen, and alkyl substituents, without compromising yield or selectivity. The reaction proceeds in a mixed solvent system, specifically optimized to balance solubility and reactivity, ensuring that even sterically hindered substrates can be converted effectively. This approach not only simplifies the synthetic route but also aligns with green chemistry principles by minimizing waste generation and avoiding the use of toxic heavy metals, making it an ideal candidate for reliable pharmaceutical intermediate supplier networks aiming for sustainability.
Mechanistic Insights into Metal-Free Fluoroalkylation
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams looking to adapt this chemistry for their specific pipelines. The reaction likely proceeds through a nucleophilic attack where the difluoroenol silyl ether acts as a nucleophile towards the electrophilic center of the phosphonium salt. The absence of a metal catalyst suggests that the activation energy is sufficiently lowered by the electronic properties of the reactants and the stabilizing effect of the solvent medium. The use of dimethyl sulfoxide (DMSO) and acetonitrile (MeCN) plays a pivotal role in stabilizing the transition state and facilitating the departure of the triphenylphosphine oxide byproduct. This mechanism ensures that the gem-difluoro motif is installed with high fidelity, preserving the integrity of the adjacent stereocenters and functional groups. Such mechanistic clarity provides confidence in the reproducibility of the process across different batches and scales.
Impurity control is another critical aspect where this novel method excels, offering distinct advantages over radical-based or metal-catalyzed alternatives. In many traditional fluorination reactions, side reactions such as defluorination or over-oxidation can lead to complex impurity profiles that are difficult to separate. However, the ionic nature of this fluoroalkylation pathway minimizes the formation of radical byproducts, resulting in a cleaner crude reaction mixture. The patent data indicates that simple workup procedures, involving extraction and standard silica gel chromatography, are sufficient to achieve purities exceeding 98%. This high level of chemical purity is essential for meeting the stringent specifications required by global regulatory bodies for clinical trial materials. By reducing the burden on downstream purification, manufacturers can significantly shorten the lead time for high-purity pharmaceutical intermediates, accelerating the path from benchtop discovery to commercial production.
How to Synthesize Beta-Diaryl-Difluoroketones Efficiently
To implement this synthesis effectively, operators must adhere to the specific solvent ratios and temperature profiles outlined in the patent embodiments. The process begins with the precise weighing of the phosphonium salt and the difluoroenol silyl ether, typically in a molar ratio of 1:2 to 1:3 to drive the reaction to completion. The choice of solvent is paramount; a mixture of DMSO and acetonitrile in a 3:1 volume ratio has been identified as optimal for maximizing yield while maintaining manageable viscosity. The reaction mixture is then heated to 50°C and stirred for approximately 12 hours under an air atmosphere, eliminating the need for inert gas setups which further reduces operational costs. Detailed standardized synthesis steps follow below to ensure consistent results.
- Prepare the reaction mixture by combining di(hetero)arylmethyltriphenylphosphonium salt and difluoroenol silyl ether in a solvent system.
- Utilize a mixed solvent of dimethyl sulfoxide and acetonitrile (3: 1 ratio) to optimize solubility and reaction kinetics.
- Maintain the reaction temperature at 50°C for 12 hours under air atmosphere to achieve high conversion without metal catalysts.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology translates into tangible strategic benefits beyond mere chemical efficiency. The elimination of transition metal catalysts removes a significant variable from the cost structure, shielding the project from the price volatility associated with palladium, platinum, or rhodium. Moreover, the mild reaction conditions reduce the energy load required for heating or cooling, contributing to substantial cost savings in utility expenses over the lifecycle of the product. The simplicity of the workup procedure means that manufacturing facilities can process larger batches with existing equipment, enhancing throughput without the need for capital-intensive infrastructure upgrades. This operational flexibility is key to ensuring supply continuity in a market where demand for fluorinated building blocks is rapidly increasing.
- Cost Reduction in Manufacturing: The catalyst-free nature of this process fundamentally alters the cost equation by removing the expense of precious metals and the specialized ligands often required to stabilize them. Without the need for metal scavenging resins or extensive washing protocols to meet residual metal limits, the consumption of auxiliary materials is drastically simplified. This reduction in material inputs directly lowers the cost of goods sold (COGS), allowing for more competitive pricing strategies in the global marketplace. Furthermore, the high yields reported in the patent embodiments suggest that raw material utilization is maximized, minimizing waste disposal costs and improving the overall atom economy of the synthesis.
- Enhanced Supply Chain Reliability: Relying on readily available starting materials such as phosphonium salts and silyl ethers mitigates the risk of supply disruptions that often plague specialty reagent markets. Since the reaction does not depend on air-sensitive catalysts, storage and handling requirements are less stringent, reducing the logistical complexity of transporting hazardous materials. This robustness ensures that production schedules can be maintained consistently, even in the face of external supply chain pressures. For a reliable pharmaceutical intermediate supplier, this means the ability to commit to longer-term contracts with greater confidence in delivery performance.
- Scalability and Environmental Compliance: The use of common industrial solvents like DMSO and acetonitrile facilitates easy scale-up from laboratory to pilot and commercial plants without the need for specialized reactor linings or containment systems. The absence of heavy metals simplifies wastewater treatment processes, ensuring compliance with increasingly strict environmental regulations regarding effluent discharge. This environmental compatibility not only reduces regulatory risk but also enhances the corporate social responsibility profile of the manufacturing operation. The process is designed to be green and low-pollution, aligning with the sustainability goals of major multinational corporations seeking responsible partners for their chemical sourcing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this fluorination technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the method's capabilities and limitations. Understanding these details helps stakeholders make informed decisions about integrating this route into their existing manufacturing portfolios.
Q: What are the primary advantages of this metal-free fluoroalkylation method?
A: The primary advantages include the elimination of expensive transition metal catalysts, mild reaction conditions (50°C), and excellent functional group tolerance, which simplifies downstream purification and reduces environmental impact.
Q: Can this synthesis method be scaled for industrial production?
A: Yes, the use of common solvents like DMSO and acetonitrile, combined with the absence of sensitive catalysts, makes this process highly amenable to commercial scale-up and robust supply chain integration.
Q: What types of substituents are tolerated in this reaction?
A: The method demonstrates broad substrate scope, tolerating various groups such as methoxy, halogens, methyl, and heteroaryl moieties like indole and thiophene, ensuring versatility for diverse pharmaceutical intermediate synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Diaryl-Difluoroketones Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the chemistry described in CN115028505B for the production of advanced fluorinated intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from development to market. Our facility is equipped with stringent purity specifications and rigorous QC labs capable of verifying the absence of metal contaminants and confirming the structural integrity of complex fluorinated scaffolds. We are committed to delivering high-purity pharmaceutical intermediates that meet the exacting standards of the global healthcare industry.
We invite you to collaborate with our technical team to explore how this metal-free synthesis can optimize your specific supply chain requirements. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how we can support your long-term strategic goals with reliable, high-quality chemical solutions.
