Revolutionizing THC Precursor Synthesis: A Catalyst-Free Approach For Commercial Scale-Up Of Complex Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic pathways that balance high yield with operational simplicity, particularly for complex chiral intermediates used in cannabinoid synthesis. Patent CN1511130A introduces a groundbreaking methodology for the addition reaction of (+)-2-carene epoxide without the necessity of acid or base catalysts, marking a significant departure from traditional rearrangement protocols. This innovation addresses the longstanding challenge of product mixture formation inherent in acid-catalyzed processes, offering a route that is not only chemically elegant but also commercially viable for large-scale manufacturing. By coupling (+)-2-carene epoxide with compounds possessing both nucleophilic and electrophilic moieties, such as water or olivetol, this method achieves complete reaction conversion while preserving the critical stereochemical integrity required for downstream bioactivity. For R&D directors and procurement specialists, this represents a pivotal shift towards more sustainable and cost-effective production of high-purity pharmaceutical intermediates, specifically targeting the synthesis of tetrahydrocannabinol (THC) precursors with enhanced reliability.
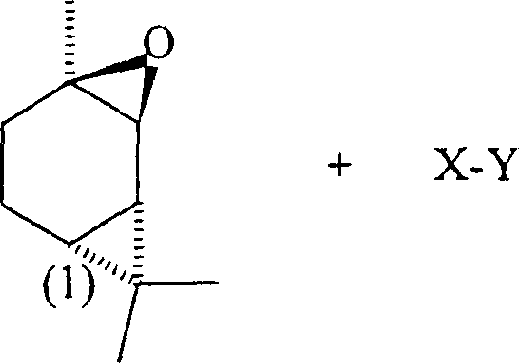
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the transformation of (+)-2-carene epoxide into valuable intermediates like (+)-p-menthadienol or diols has relied heavily on acid-catalyzed rearrangement reactions, which are fraught with significant chemical and operational inefficiencies. The fundamental issue lies in the high acid instability of the adjacent cyclopropyl-epoxy moiety, which readily releases strain energy from the two three-membered rings to form a very stable cyclopropylmethyl cation. This cationic intermediate is highly reactive and non-selective, leading to a complex mixture of rearrangement products rather than a single target compound, thereby drastically reducing the yield of the desired material. Literature precedents, such as those by Bledsoe and Bulliard, demonstrate that even under optimized conditions using metatitanic acid or pyridinium p-toluenesulfonate, yields often struggle to exceed moderate levels, with significant amounts of byproducts requiring costly and time-consuming purification steps. Furthermore, the use of strong acids necessitates specialized corrosion-resistant reactor equipment and generates hazardous waste streams that complicate environmental compliance and increase the overall carbon footprint of the manufacturing process.
The Novel Approach
In stark contrast, the method disclosed in CN1511130A eliminates the need for any acid or base catalyst, thereby preventing the formation of the problematic cyclopropylmethyl cation and steering the reaction towards a direct addition mechanism. This novel approach allows the (+)-2-carene epoxide to react thoroughly with reagents like water, alcohols, or phenols under neutral or mildly buffered conditions, resulting in a much cleaner reaction profile with significantly improved yields. The absence of harsh catalytic conditions means that the stereochemistry of the two chiral centers is maintained as the reaction progresses, which is critical for the biological activity of the final cannabinoid products. By avoiding the chaotic rearrangement pathways typical of acid catalysis, this method simplifies the downstream purification process, as the reaction mixture primarily consists of the target compound and unreacted starting materials that can be easily separated. This technological leap not only enhances the chemical efficiency but also aligns perfectly with modern green chemistry principles, offering a sustainable alternative for the production of high-purity pharmaceutical intermediates.
Mechanistic Insights into Catalyst-Free Nucleophilic Addition
The mechanistic underpinning of this catalyst-free addition reaction is rooted in the direct nucleophilic attack on the epoxide ring without prior protonation, which fundamentally alters the reaction trajectory compared to traditional acid-catalyzed routes. In the absence of a strong acid catalyst, the reaction is believed to follow a pathway more consistent with the Razdan mechanism, where the nucleophilic portion of the reagent X-Y attacks the electrophilic center of the epoxide directly. This avoids the rapid formation of the cyclopropylmethyl cation that typically leads to indiscriminate rearrangement, allowing for a controlled ring-opening that preserves the structural integrity of the carene skeleton. The reaction mixture, composed of the epoxide, the X-Y compound, and optionally an inert solvent or pH buffer, creates an environment where the nucleophile can effectively compete with potential rearrangement pathways, ensuring that the addition product is formed preferentially. This level of control is essential for maintaining the specific stereochemical configuration required for the subsequent synthesis of (-)-Δ9-tetrahydrocannabinol, as any loss of chirality at this stage would render the final product biologically inactive or less potent.
Furthermore, the impurity control mechanism inherent in this catalyst-free process is superior because it minimizes the generation of isomeric byproducts that are difficult to separate from the target molecule. In acid-catalyzed systems, the formation of various dienes and alcohols creates an impurity profile that often requires multiple chromatography steps or recrystallizations to resolve, driving up production costs and extending lead times. By contrast, the neutral conditions of this new method ensure that the primary impurity is often just the unreacted starting material or simple addition byproducts that do not share the same structural complexity as rearrangement isomers. This simplified impurity谱 allows for more straightforward analytical monitoring using standard techniques like HPLC or TLC, enabling tighter quality control during the manufacturing process. For R&D teams, this means that method validation and scale-up activities can proceed more rapidly, as the robustness of the reaction reduces the risk of batch-to-batch variability caused by subtle changes in acid concentration or temperature.
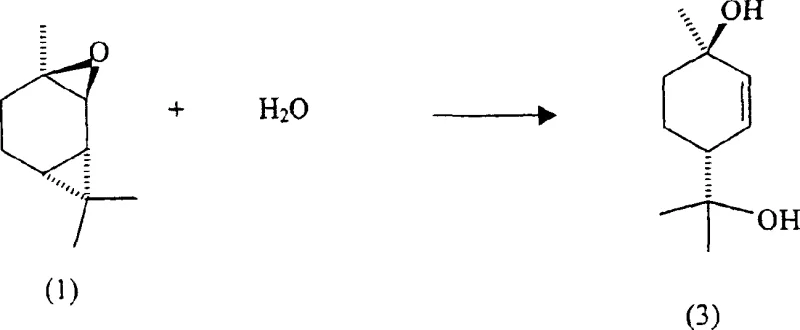
How to Synthesize (+)-p-menth-2-ene-1,8-diol Efficiently
The synthesis of (+)-p-menth-2-ene-1,8-diol using this patented catalyst-free method involves a streamlined procedure that begins with the preparation of the reaction mixture using either crude or distilled (+)-2-carene epoxide. The process is designed to be flexible, allowing for the use of crude epoxidized products directly from turpentine sources without the need for extensive pre-purification, which significantly reduces raw material costs and processing time. The reaction is typically carried out in an aqueous pH buffer solution maintained within a specific range, such as pH 5.7 to 5.9, to ensure optimal reaction kinetics while preventing any unintended acid-catalyzed side reactions. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by suspending crude or distilled (+)-2-carene epoxide in an aqueous pH buffer solution or inert solvent depending on the nucleophile used.
- Maintain the reaction temperature between 10°C to 150°C, preferably around 40°C for aqueous reactions, ensuring vigorous stirring to facilitate the coupling without acid catalysts.
- Isolate the product by extraction with organic solvents such as ethyl acetate or heptane, followed by purification via recrystallization or chromatography to achieve high purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this catalyst-free synthesis route offers profound advantages for procurement managers and supply chain heads who are tasked with optimizing costs and ensuring material availability. The elimination of acid catalysts removes the need for expensive neutralization reagents and the associated waste treatment processes, leading to substantial cost savings in terms of both raw material consumption and environmental compliance fees. Additionally, the ability to use crude reaction mixtures and simpler workup procedures means that the overall manufacturing cycle time is drastically simplified, allowing for faster turnover and improved responsiveness to market demand fluctuations. This efficiency gain is particularly valuable in the volatile cannabinoid market, where speed to market can be a decisive competitive advantage for suppliers aiming to capture market share.
- Cost Reduction in Manufacturing: The removal of acid catalysts from the process equation eliminates the need for specialized corrosion-resistant equipment and the costly disposal of hazardous acidic waste, resulting in a leaner and more economical production model. By avoiding the complex purification steps required to remove catalyst residues and rearrangement byproducts, manufacturers can achieve significant reductions in solvent usage and energy consumption, further driving down the cost of goods sold. This qualitative improvement in process efficiency translates directly to better margin protection and the ability to offer more competitive pricing to downstream pharmaceutical clients without compromising on quality standards.
- Enhanced Supply Chain Reliability: The use of readily available starting materials like turpentine derivatives and common solvents ensures a robust supply chain that is less susceptible to disruptions caused by the scarcity of specialized reagents. The mild reaction conditions, operating at atmospheric pressure and moderate temperatures, reduce the risk of equipment failure and safety incidents, thereby ensuring consistent production uptime and reliable delivery schedules. This stability is crucial for maintaining long-term contracts with major pharmaceutical partners who require guaranteed supply continuity for their critical drug development pipelines.
- Scalability and Environmental Compliance: The simplicity of the catalyst-free protocol makes it highly scalable from laboratory benchtop to industrial tonnage production without the engineering bottlenecks associated with handling large volumes of strong acids. The reduced generation of hazardous waste aligns with increasingly stringent global environmental regulations, minimizing the regulatory burden and potential liabilities associated with chemical manufacturing. This forward-looking approach to process design ensures that the production facility remains compliant with future sustainability mandates, safeguarding the long-term viability of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions and answers are derived directly from the technical specifics of patent CN1511130A, addressing common concerns regarding the feasibility and advantages of this catalyst-free synthesis method. These insights are intended to provide clarity for technical decision-makers evaluating the potential integration of this route into their existing manufacturing frameworks. The responses highlight the key differentiators of this technology, focusing on yield improvement, stereochemical retention, and operational simplicity.
Q: Why is the catalyst-free method superior to acid-catalyzed rearrangement for carene epoxide?
A: Acid-catalyzed methods typically generate unstable cyclopropylmethyl cations leading to complex product mixtures and low yields, whereas the catalyst-free approach maintains stereochemistry and produces a single dominant product with significantly higher purity.
Q: Can this process be scaled for industrial production of THC intermediates?
A: Yes, the process utilizes mild conditions, atmospheric pressure, and readily available solvents, making it highly suitable for commercial scale-up without the need for specialized corrosion-resistant equipment required for strong acid catalysis.
Q: What are the cost implications of eliminating the acid catalyst?
A: Eliminating the acid catalyst removes the need for expensive neutralization steps, heavy metal removal processes, and complex waste treatment, resulting in substantial cost savings and a streamlined manufacturing workflow.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (+)-2-Carene Epoxide Supplier
As a leading CDMO expert, NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent concept to industrial reality is seamless and efficient. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of (+)-2-carene epoxide and its derivatives meets the exacting standards required for pharmaceutical applications. We understand the critical nature of chiral intermediates in drug synthesis and have optimized our processes to deliver the high-purity pharmaceutical intermediates necessary for your success.
We invite you to engage with our technical procurement team to request specific COA data and route feasibility assessments tailored to your project needs. By leveraging our expertise, you can secure a Customized Cost-Saving Analysis that demonstrates how integrating this catalyst-free technology can optimize your supply chain and reduce overall manufacturing expenses. Let us partner with you to engineer the next generation of cannabinoid therapeutics with precision and reliability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
