Advanced Solvent-Free Synthesis of 4,4'-Bis(Alpha,Alpha-Dimethylbenzyl)Diphenylamine for Global Rubber Industries
Advanced Solvent-Free Synthesis of 4,4'-Bis(Alpha,Alpha-Dimethylbenzyl)Diphenylamine for Global Rubber Industries
The global demand for high-performance polymer additives continues to drive innovation in synthetic methodologies, particularly for antioxidants that ensure the longevity of rubber and plastic materials. Patent CN101538208A introduces a transformative approach to manufacturing a critical mixture of 4-(alpha, alpha-dimethylbenzyl) diphenylamine and 4,4-bis (alpha, alpha-dimethylbenzyl) diphenylamine. This technology represents a significant departure from legacy processes by utilizing activated clay as a heterogeneous catalyst under solvent-free conditions. For R&D directors and technical leaders, this patent offers a compelling blueprint for achieving superior product purity while simultaneously addressing stringent environmental regulations. The process operates effectively within a temperature range of 120-175°C, specifically optimized around 125-155°C, ensuring thermal stability during the alkylation reaction. By eliminating the need for corrosive liquid acids and organic solvents, this method not only simplifies the downstream processing but also enhances the overall safety profile of the manufacturing facility.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alkylated diphenylamine antioxidants has relied heavily on homogeneous acid catalysis, often involving strong mineral acids like hydrochloric acid or sulfuric acid. As referenced in prior art such as CN1195661A, these traditional routes necessitate a complex post-reaction workup that includes acidification, followed by multiple stages of alkaline neutralization and extensive water washing. This multi-step purification protocol imposes a severe burden on production efficiency, leading to high labor intensity and significant equipment corrosion due to the aggressive chemical environment. Furthermore, the generation of large volumes of acidic and alkaline wastewater creates a substantial environmental liability, requiring costly treatment infrastructure to meet modern discharge standards. The use of organic solvents in these conventional methods further complicates the process, introducing risks associated with solvent recovery, flammability, and residual solvent contamination in the final API or additive product.
The Novel Approach
In stark contrast, the methodology disclosed in CN101538208A leverages the unique properties of activated clay (attapulgite) to drive the alkylation of diphenylamine with alpha-methyl styrene. This solid acid catalyst enables a clean, solvent-free reaction environment that fundamentally alters the economic and operational dynamics of production. The novel approach allows for the direct formation of the crude product mixture, which primarily consists of the desired di-alkylated species along with a minor fraction of the mono-alkylated intermediate. Crucially, the post-reaction processing is drastically simplified to merely filtering off the solid catalyst and subjecting the filtrate to vacuum distillation. This elimination of aqueous washing steps not only prevents the formation of emulsions and reduces water consumption to near zero but also ensures that the final product possesses exceptional thermal and oxidative stability. The ability to operate without solvents means that reactor volume is utilized much more efficiently, directly translating to higher throughput per batch.
Mechanistic Insights into Solid Acid Catalyzed Alkylation
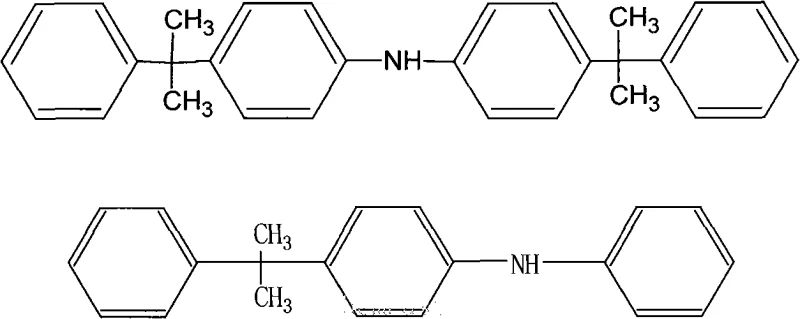
The core of this technological advancement lies in the Friedel-Crafts alkylation mechanism facilitated by the Lewis and Brønsted acid sites present on the surface of the activated clay. When diphenylamine and alpha-methyl styrene are heated together in the presence of this catalyst, the double bond of the styrene derivative is activated, forming a carbocation intermediate that attacks the para-position of the diphenylamine ring. The selectivity of this reaction is finely tuned by the molar ratio of the reactants; the patent specifies a ratio of diphenylamine to alpha-methyl styrene between 1:1.5 and 1:2.6, with a preference for 1:2.0 to 2.6. This excess of the olefin drives the equilibrium towards the formation of the di-alkylated product, which constitutes 90-95% of the final mixture by weight. The remaining 5-10% is the mono-alkylated species, which acts synergistically to provide comprehensive antioxidant protection in rubber matrices. The heterogeneous nature of the catalyst ensures that the active sites are accessible while allowing for easy physical separation post-reaction.
From an impurity control perspective, the absence of liquid acid catalysts prevents the formation of tar-like polymeric byproducts that are common in homogeneous acid systems. The mild reaction conditions (125-155°C) minimize the risk of thermal degradation or cracking of the alpha-methyl styrene, which can occur at higher temperatures. Furthermore, the vacuum distillation step serves a dual purpose: it removes any unreacted low-boiling alpha-methyl styrene and isolates the high-boiling antioxidant mixture with high precision. This results in a product with a melting point above 85°C and extremely low volatility, characteristics that are essential for preventing blooming in finished rubber goods. The structural integrity of the cumyl groups attached to the nitrogen center provides steric hindrance that protects the amine functionality from rapid oxidation, thereby extending the service life of the protected polymer.
How to Synthesize 4,4'-Bis(Alpha,Alpha-Dimethylbenzyl)Diphenylamine Efficiently
To implement this synthesis route effectively, manufacturers must adhere to precise thermal profiles and mixing protocols to maximize yield and catalyst life. The process begins with the dehydration of the activated clay catalyst at elevated temperatures to remove adsorbed moisture, which is critical for maintaining catalytic activity. Following this preparation, the diphenylamine is melted and mixed with the catalyst under a nitrogen atmosphere to prevent premature oxidation. The alpha-methyl styrene is then introduced gradually to manage the exothermic nature of the alkylation reaction. Detailed standardized synthesis steps see the guide below.
- Charge diphenylamine and activated clay catalyst into a reactor equipped with agitation and nitrogen protection, then heat to melt the amine.
- Slowly add excess alpha-methyl styrene while maintaining temperature between 125°C and 155°C to facilitate controlled alkylation.
- Perform hot filtration to recover the solid catalyst, followed by vacuum distillation of the filtrate to isolate the final antioxidant mixture.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the technology described in CN101538208A offers a strategic pathway to optimize operational expenditures and mitigate supply risks. The transition from a multi-step aqueous workup to a simple filtration and distillation sequence fundamentally reduces the complexity of the manufacturing workflow. This simplification directly correlates to a reduction in utility consumption, as there is no longer a need for vast quantities of process water or the energy-intensive heating and cooling cycles associated with washing and phase separation. Additionally, the elimination of corrosive acids extends the lifespan of reactor vessels and piping, deferring capital expenditure on equipment replacement. The robustness of this solvent-free process ensures consistent batch-to-batch quality, reducing the likelihood of costly rejections or re-processing.
- Cost Reduction in Manufacturing: The most significant financial benefit arises from the complete removal of solvent purchase, recovery, and disposal costs. In traditional fine chemical manufacturing, solvent management can account for a substantial portion of variable costs; by operating solvent-free, this margin is entirely recovered. Furthermore, the ability to separate and potentially recycle the activated clay catalyst reduces the recurring cost of catalytic materials. The streamlined process also lowers labor costs, as the operator time required for monitoring complex washing and neutralization steps is eliminated. These factors combine to create a leaner cost structure that provides a competitive edge in the pricing of high-purity rubber additives.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, diphenylamine and alpha-methyl styrene, are commodity chemicals with well-established global supply chains, reducing the risk of raw material shortages. The simplicity of the process equipment means that production can be scaled up or shifted between facilities with minimal technical barriers, ensuring continuity of supply even during regional disruptions. The high yield range of 85-92% reported in the patent examples indicates a highly efficient conversion of raw materials, minimizing waste and maximizing the output per unit of input. This efficiency buffers the supply chain against fluctuations in raw material pricing, allowing for more stable long-term contracting.
- Scalability and Environmental Compliance: Scaling this process from pilot to commercial production is straightforward because the reaction does not rely on mass transfer limitations often seen in biphasic liquid-liquid systems. The absence of wastewater generation simplifies environmental permitting and compliance, removing a major bottleneck for capacity expansion in regions with strict ecological regulations. The solid waste generated (spent catalyst) is non-hazardous compared to acidic sludge, making disposal or regeneration easier and cheaper. This environmental compatibility future-proofs the manufacturing asset against tightening global sustainability mandates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced alkylation technology. These insights are derived directly from the experimental data and claims within the patent documentation to provide clarity for potential partners. Understanding these nuances is critical for evaluating the feasibility of integrating this process into existing production lines.
Q: How does the activated clay catalyst improve the environmental profile compared to traditional methods?
A: Unlike traditional liquid acid catalysts that require extensive aqueous washing and neutralization steps generating hazardous wastewater, the activated clay catalyst is a solid that can be removed via simple hot filtration. This eliminates the need for acid/base washing, drastically reducing three-waste emissions and equipment corrosion risks.
Q: What is the typical composition ratio of the mono-alkylated to di-alkylated products in this process?
A: By controlling the molar ratio of diphenylamine to alpha-methyl styrene (typically 1:1.5 to 1:2.6), the process yields a mixture containing approximately 5-10% of the mono-alkylated derivative and 90-95% of the desired 4,4'-bis(alpha,alpha-dimethylbenzyl)diphenylamine, optimizing performance for rubber stabilization.
Q: Can the catalyst be reused to further lower production costs?
A: Yes, the patent explicitly states that the activated clay catalyst can be separated from the reaction mixture by filtration or centrifugation and potentially reused for subsequent batches without significant loss of activity, contributing to substantial raw material cost savings.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,4'-Bis(Alpha,Alpha-Dimethylbenzyl)Diphenylamine Supplier
As the global market for high-performance rubber antioxidants expands, the ability to deliver consistent, high-purity materials becomes a key differentiator. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging deep expertise in solid-acid catalyzed reactions to manufacture complex amine derivatives. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory successes are seamlessly translated into industrial reality. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 4,4'-bis(alpha,alpha-dimethylbenzyl)diphenylamine meets the exacting standards required by top-tier tire and polymer manufacturers.
We invite industry leaders to collaborate with us to unlock the full potential of this efficient synthetic route. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is optimized for both performance and profitability in the coming fiscal year.
