Advanced Zero-Wastewater Synthesis of N,N-Dibenzylhydroxylamine Antioxidants for Industrial Scale-Up
The global demand for high-performance polymer additives continues to drive innovation in synthetic methodologies, particularly for antioxidants that ensure the longevity and stability of plastic materials. Patent CN102391149B introduces a groundbreaking approach to the manufacture of N,N-dibenzylhydroxylamine derivatives, addressing critical environmental and efficiency bottlenecks inherent in legacy production techniques. This technology leverages a strictly anhydrous reaction system utilizing organic amines as acid-binding agents, fundamentally altering the waste profile of the synthesis. By shifting away from traditional inorganic bases and aqueous workups, this method effectively controls pollutant generation at the source, offering a compelling solution for manufacturers seeking to align with stringent environmental regulations while maintaining high throughput. The significance of this patent lies not only in its ecological benefits but also in its ability to enhance the economic viability of producing these specialized carbon radical scavengers through simplified downstream processing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of N,N-dibenzylhydroxylamine antioxidants has relied heavily on processes involving inorganic neutralizing agents such as anhydrous sodium carbonate, often conducted in solvents that tolerate or contain moisture. A major drawback of these conventional routes is the inevitable generation of water during the neutralization of hydrochloric acid, which creates a conducive environment for the hydrolysis of the alkyl benzyl halide starting materials. This hydrolysis side reaction produces alkyl benzyl alcohol impurities, which are difficult to separate and directly diminish the yield of the target antioxidant. Furthermore, the resulting reaction mixture contains inorganic salts like sodium chloride, necessitating extensive aqueous washing steps to purify the product. Data from prior art indicates that producing a single ton of product via these traditional methods can generate between 10 to 15 tons of industrial wastewater containing residual solvents like dimethylformamide, posing severe environmental disposal challenges and inflating operational costs related to effluent treatment.
The Novel Approach
In stark contrast, the methodology disclosed in CN102391149B utilizes a completely anhydrous organic system where organic amines serve as the acid-binding agent instead of inorganic bases. This strategic substitution prevents the formation of water within the reaction matrix, thereby suppressing the hydrolysis of the alkyl benzyl halide and preserving the integrity of the starting material for the desired nucleophilic substitution. The by-product formed is an organic amine hydrochloride salt, which exhibits distinct solubility characteristics allowing for removal via hot filtration rather than aqueous extraction. This innovation effectively eliminates the generation of wastewater at the production source, solving the pollution issues associated with traditional discharge. Additionally, the process facilitates the recovery and recycling of the organic amine, creating a closed-loop system that enhances atom economy. The structural versatility of this approach allows for the synthesis of various derivatives where R can be hydrogen or C1-C10 alkyl groups, as illustrated in the general formula below.
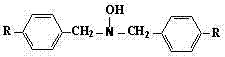
Mechanistic Insights into Anhydrous Nucleophilic Substitution
The core chemical transformation in this process is a nucleophilic substitution where hydroxylamine hydrochloride reacts with alkyl benzyl halides. In the absence of water, the organic amine efficiently scavenges the proton released during the formation of the N-C bond, driving the equilibrium forward without introducing competing nucleophiles like hydroxide ions that would lead to alcohol formation. The patent specifies a two-stage temperature profile to optimize this mechanism: an initial lower temperature phase (10-80°C) for the controlled addition of the halide to manage exothermicity and minimize side reactions, followed by a higher temperature phase (60-130°C) to ensure complete conversion of the starting materials. This thermal management is crucial for maximizing the yield of the N,N-dibenzylhydroxylamine derivative while maintaining the stability of the hydroxylamine moiety, which can be sensitive to harsh oxidative or thermal conditions if not properly managed within the solvent system.
Impurity control is intrinsically linked to the choice of reagents and the anhydrous environment. By preventing the formation of alkyl benzyl alcohol, the crude product profile is significantly cleaner, reducing the burden on final crystallization steps. The reaction mechanism relies on the lone pair electrons of the nitrogen in the hydroxylamine attacking the benzylic carbon of the halide. The presence of the organic amine ensures that the hydrochloric acid by-product is immediately neutralized to form a precipitable salt, shifting the reaction equilibrium towards the product side according to Le Chatelier's principle. The starting materials, specifically the alkyl benzyl halides where X is Cl or Br, are shown in the structural representation below, highlighting the leaving group essential for this substitution pathway.
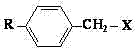
How to Synthesize N,N-Dibenzylhydroxylamine Efficiently
The synthesis protocol outlined in the patent provides a robust framework for laboratory and pilot-scale production, emphasizing precise thermal control and phase separation techniques. The process begins with the preparation of an anhydrous reaction mixture containing hydroxylamine hydrochloride and a tertiary amine such as triethylamine in a suitable organic solvent like toluene or xylene. Following the two-stage heating and addition cycle, the reaction mixture undergoes a critical hot filtration step to remove the amine salt, followed by solvent recovery and crystallization. This sequence ensures that the final antioxidant product meets high purity specifications required for polymer applications. For detailed operational parameters including specific molar ratios, solvent volumes, and temperature ramping rates, please refer to the standardized synthesis guide provided below.
- Combine hydroxylamine hydrochloride, organic amine acid-binding agent, and organic solvent in a reactor. Heat to 10-80°C and dropwise add alkyl benzyl halide, maintaining temperature for 30-150 minutes.
- Raise the reaction temperature to 60-130°C and continue heating for 180-240 minutes to complete the formation of N,N-dibenzylhydroxylamine derivatives and organic amine hydrochloride by-products.
- Add additional organic solvent, heat to dissolve the product, and perform hot filtration to separate the solid organic amine hydrochloride salt from the product solution.
- Concentrate the filtrate, cool to crystallize the product, filter, wash with ethanol, and dry to obtain high-purity N,N-dibenzylhydroxylamine antioxidant crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this zero-wastewater synthesis route offers transformative economic and logistical benefits. The elimination of aqueous washing steps drastically reduces the volume of effluent requiring treatment, leading to substantial cost savings in environmental compliance and waste disposal fees. Moreover, the ability to recover and recycle the organic amine acid scavenger reduces the recurring cost of raw materials, enhancing the overall margin profile of the manufacturing process. The simplified purification workflow, which relies on filtration and crystallization rather than complex extraction and drying of aqueous phases, shortens the production cycle time and increases the throughput capacity of existing reactor infrastructure without the need for significant capital expenditure on new wastewater treatment facilities.
- Cost Reduction in Manufacturing: The transition to an anhydrous system removes the necessity for expensive wastewater treatment infrastructure and the associated energy costs for evaporating large volumes of water during product isolation. By avoiding the formation of inorganic salt sludge, the process minimizes solid waste disposal costs. Furthermore, the high selectivity of the reaction reduces the loss of valuable starting materials to hydrolysis by-products, ensuring that a greater proportion of input costs are converted into saleable high-purity antioxidant product, thereby optimizing the cost of goods sold.
- Enhanced Supply Chain Reliability: The raw materials required for this process, including alkyl benzyl halides and common organic amines like triethylamine, are widely available commodity chemicals with stable supply chains. The robustness of the anhydrous process against variations in raw material quality, specifically regarding moisture content which is actively managed, ensures consistent batch-to-batch quality. This reliability reduces the risk of production delays caused by off-spec batches or the need for re-processing, allowing for more predictable delivery schedules to downstream polymer manufacturers who depend on just-in-time inventory models.
- Scalability and Environmental Compliance: The unit operations involved—stirred tank reaction, hot filtration, and distillation—are standard in the fine chemical industry and scale linearly from pilot to commercial production. The absence of wastewater discharge simplifies the permitting process for new production lines in regions with strict environmental regulations. This scalability ensures that suppliers can rapidly ramp up production to meet surging demand for polymer additives without facing regulatory bottlenecks, securing long-term supply continuity for global customers seeking sustainable sourcing options.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this green synthesis technology. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing clarity on how this method outperforms traditional aqueous-based routes. Understanding these distinctions is vital for technical teams evaluating process feasibility and for commercial teams assessing the total cost of ownership for this antioxidant intermediate.
Q: How does this synthesis method eliminate wastewater pollution compared to traditional processes?
A: Traditional methods utilize inorganic bases like sodium carbonate in aqueous or semi-aqueous systems, generating significant saline wastewater during the washing steps to remove salts. This patented process employs an anhydrous organic system with organic amines as acid scavengers. The by-product, organic amine hydrochloride, is removed via hot filtration as a solid, completely bypassing the need for aqueous washing and thus achieving zero wastewater discharge.
Q: What represents the primary advantage regarding product yield and impurity control?
A: The strict anhydrous conditions prevent the hydrolysis of the alkyl benzyl halide starting material, which typically converts to alkyl benzyl alcohol impurities in the presence of water and base. By eliminating this side reaction pathway, the process significantly improves the conversion rate to the desired N,N-dibenzylhydroxylamine derivative, resulting in higher overall yields and superior product purity without complex purification steps.
Q: Can the organic amine acid scavenger be recovered and reused in this process?
A: Yes, the process is designed for circular efficiency. The solid by-product obtained from hot filtration is the organic amine hydrochloride salt. This salt can be further treated with alkali (such as sodium hydroxide) to liberate the free organic amine, which can then be distilled and recycled back into the synthesis reaction, substantially reducing raw material consumption and operational costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N,N-Dibenzylhydroxylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance antioxidants play in the durability and safety of modern polymer products. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the innovative zero-wastewater process described in CN102391149B can be seamlessly translated into reliable industrial supply. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of N,N-dibenzylhydroxylamine meets the exacting standards required for advanced polymer stabilization applications, delivering consistency that R&D teams can trust for their formulation development.
We invite you to collaborate with us to leverage this advanced synthesis technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can reduce your total landed cost while supporting your sustainability goals through greener chemical production methods.
