Scalable Metal-Free Synthesis of Functionalized Aryl Sulfone Derivatives for Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking greener, more efficient pathways to construct complex molecular scaffolds, particularly those containing sulfone motifs which are prevalent in bioactive molecules. Patent CN112457263A introduces a groundbreaking methodology for the synthesis of aryl sulfone derivatives that fundamentally shifts the paradigm from traditional metal-catalyzed processes to a sustainable, metal-free radical bifunctionalization strategy. This innovation leverages water as the exclusive reaction medium, utilizing potassium persulfate as a benign oxidant to drive the coupling of quinoxaline-2(1H)-one compounds with olefins and aryl sulfinates. By operating at a moderate temperature of 70°C and eliminating the need for precious metal catalysts, this process addresses critical pain points regarding environmental compliance, operational safety, and downstream purification costs. For R&D directors and procurement specialists, this technology represents a viable route to high-purity intermediates with a significantly reduced ecological footprint, aligning perfectly with modern green chemistry principles and the increasing regulatory pressure to minimize heavy metal residues in active pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of aryl sulfone derivatives via olefin bifunctionalization has relied heavily on transition metal catalysis and the use of hazardous organic solvents. Traditional protocols often necessitate expensive palladium, copper, or iron catalysts, which not only inflate raw material costs but also introduce severe challenges in post-reaction processing. The removal of trace metal contaminants to meet pharmaceutical grade specifications requires additional purification steps, such as scavenging resins or extensive recrystallization, which inevitably lower overall yields and extend production timelines. Furthermore, the reliance on volatile organic compounds (VOCs) like dichloromethane or acetonitrile as solvents poses significant safety hazards related to flammability and toxicity, while generating substantial volumes of chemical waste that require costly disposal. These conventional methods often suffer from limited substrate scope, struggling to tolerate sensitive functional groups, and frequently require harsh reaction conditions that compromise selectivity and generate complex impurity profiles, thereby hindering their scalability for commercial manufacturing.
The Novel Approach
In stark contrast, the novel method disclosed in the patent utilizes a radical-mediated mechanism that operates efficiently in an aqueous environment without any metal catalyst participation. This approach employs readily available aryl sulfinates and olefins as starting materials, activated by potassium persulfate to generate sulfonyl radicals that add across the double bond of the olefin. The resulting alkyl radical intermediate then undergoes oxidative coupling with the quinoxaline-2(1H)-one core, constructing the highly functionalized sulfone architecture in a single synthetic step. 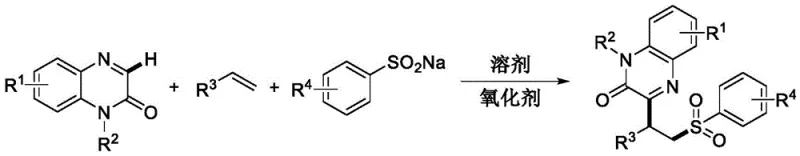 This metal-free protocol not only simplifies the reaction setup but also drastically reduces the environmental burden by replacing toxic organic solvents with water. The mild reaction conditions (70°C) ensure excellent functional group compatibility, allowing for the incorporation of diverse substituents such as halogens, ethers, and trifluoromethyl groups without degradation. This streamlined process enhances step economy and offers a robust platform for the rapid generation of structural diversity, making it an ideal candidate for the cost-effective manufacturing of complex pharmaceutical intermediates.
This metal-free protocol not only simplifies the reaction setup but also drastically reduces the environmental burden by replacing toxic organic solvents with water. The mild reaction conditions (70°C) ensure excellent functional group compatibility, allowing for the incorporation of diverse substituents such as halogens, ethers, and trifluoromethyl groups without degradation. This streamlined process enhances step economy and offers a robust platform for the rapid generation of structural diversity, making it an ideal candidate for the cost-effective manufacturing of complex pharmaceutical intermediates.
Mechanistic Insights into Metal-Free Radical Bifunctionalization
The core of this synthetic breakthrough lies in the elegant radical cascade initiated by the oxidant. Potassium persulfate serves as the radical initiator, oxidizing the aryl sulfinate salt to generate a reactive sulfonyl radical species. This electrophilic radical selectively attacks the electron-rich double bond of the olefin substrate, forming a new carbon-sulfur bond and generating a stabilized benzylic or alkyl radical intermediate. This intermediate subsequently attacks the C3 position of the quinoxaline-2(1H)-one ring system, forging the critical carbon-carbon bond that links the sulfone moiety to the heterocyclic core. Finally, an oxidative dehydrogenation step restores aromaticity or stabilizes the final product structure, completing the bifunctionalization sequence. The absence of metal coordination complexes means the reaction trajectory is governed purely by electronic and steric factors of the radical species, leading to high regioselectivity and minimizing side reactions associated with metal-ligand interactions. This mechanistic clarity allows chemists to predictably tune reaction outcomes by modifying the electronic nature of the olefin or sulfinate components.
From an impurity control perspective, the metal-free nature of this reaction is transformative. In traditional metal-catalyzed couplings, side products often arise from homocoupling of the metal species or incomplete catalyst turnover, leading to difficult-to-remove organometallic impurities. Here, the primary byproducts are inorganic salts derived from the oxidant and the sulfinate counterion, which are easily removed during the aqueous workup and extraction phases. The use of water as a solvent further aids in impurity management; as the organic product typically has low solubility in cold water, it precipitates or separates cleanly upon cooling, leaving inorganic salts in the aqueous phase. This inherent phase separation simplifies the isolation process and ensures a cleaner crude product profile before chromatography. 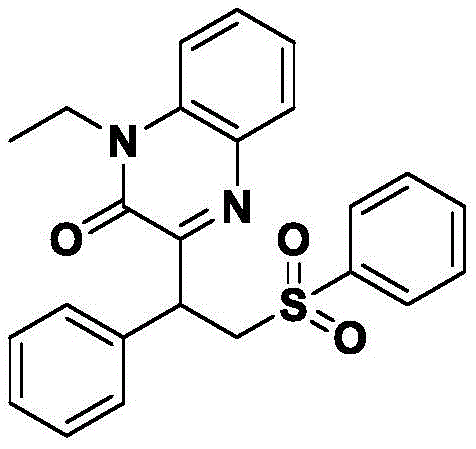 The structural integrity of the products, such as the phenyl-substituted derivative shown in Example 1, confirms that the radical conditions are mild enough to preserve sensitive functionalities while effectively building molecular complexity. This level of purity control is essential for downstream applications where trace impurities can affect biological activity or formulation stability.
The structural integrity of the products, such as the phenyl-substituted derivative shown in Example 1, confirms that the radical conditions are mild enough to preserve sensitive functionalities while effectively building molecular complexity. This level of purity control is essential for downstream applications where trace impurities can affect biological activity or formulation stability.
How to Synthesize 1-Ethyl-3-(1-phenyl-2-(phenylsulfonyl)ethyl)quinoxalin-2(1H)-one Efficiently
The synthesis of this specific aryl sulfone derivative exemplifies the operational simplicity and efficiency of the patented method. By reacting 1-ethylquinoxaline-2(1H)-one with styrene and sodium phenyl sulfinate in the presence of potassium persulfate, manufacturers can achieve high yields under environmentally benign conditions. The process avoids the need for inert atmosphere techniques or anhydrous solvents, further lowering the barrier to entry for scale-up. Detailed standard operating procedures for this transformation, including precise stoichiometric ratios and purification parameters, are outlined in the technical guidelines below to ensure reproducibility and quality consistency.
- Charge a reaction vessel with quinoxaline-2(1H)-one, olefin, aryl sulfinate, and potassium persulfate oxidant in specific molar ratios.
- Add water as the sole solvent and heat the mixture to 70°C under oil bath conditions for approximately 24 hours.
- Upon completion, cool the reaction, extract with ethyl acetate, and purify the crude product via column chromatography to isolate the target aryl sulfone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this aqueous, metal-free synthesis route offers compelling economic and logistical benefits that extend beyond simple yield metrics. The elimination of expensive transition metal catalysts directly reduces the bill of materials, while the removal of metal scavenging steps shortens the overall production cycle time. Furthermore, the use of water as a solvent mitigates the risks and costs associated with the storage, handling, and disposal of large volumes of flammable organic solvents, enhancing facility safety and reducing insurance premiums. This process aligns with sustainability goals, potentially qualifying for green manufacturing incentives and improving the corporate environmental profile, which is increasingly important for securing contracts with major pharmaceutical partners who prioritize responsible sourcing.
- Cost Reduction in Manufacturing: The most significant cost driver in traditional sulfone synthesis is often the catalyst system and the subsequent purification required to remove metal residues. By utilizing potassium persulfate, a commodity chemical, instead of precious metals, the raw material costs are drastically lowered. Additionally, the simplified workup procedure, which relies on phase separation rather than complex chromatography or scavenging, reduces labor hours and solvent consumption. This cumulative effect results in a substantially lower cost of goods sold (COGS), allowing for more competitive pricing in the global market for pharmaceutical intermediates without compromising on quality or purity specifications.
- Enhanced Supply Chain Reliability: The reagents required for this synthesis—quinoxalinones, styrenes, aryl sulfinates, and persulfates—are widely available commodity chemicals with stable supply chains, unlike specialized ligands or catalysts that may face geopolitical or manufacturing bottlenecks. The robustness of the reaction conditions (tolerance to air and moisture due to the aqueous medium) reduces the risk of batch failures due to environmental fluctuations, ensuring consistent output. This reliability translates to shorter lead times and more predictable delivery schedules, enabling downstream customers to maintain leaner inventories and respond more agilely to market demands for API precursors.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental challenges, particularly when dealing with exothermic reactions in organic solvents. The use of water as a heat sink in this reaction provides excellent thermal control, minimizing the risk of thermal runaway during scale-up from laboratory to pilot and commercial scales. Moreover, the aqueous waste stream is significantly easier and cheaper to treat compared to mixed organic waste, facilitating compliance with increasingly stringent environmental regulations. This ease of scale-up ensures that the technology can seamlessly transition from gram-scale R&D to multi-ton commercial production, securing long-term supply continuity for high-volume applications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this green synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a transparent view of the method's capabilities and limitations for potential adopters and partners.
Q: What are the primary advantages of using water as a solvent in this sulfone synthesis?
A: Using water eliminates the need for flammable and volatile organic solvents, significantly reducing environmental impact and safety risks. Additionally, water facilitates easier product separation upon cooling due to low solubility of organic products, simplifying downstream processing and reducing waste treatment costs.
Q: Does this metal-free method meet pharmaceutical purity standards?
A: Yes, the absence of transition metal catalysts removes the critical bottleneck of heavy metal residue removal, which is a stringent requirement for API intermediates. This inherently improves the impurity profile and reduces the complexity of purification steps required to meet regulatory specifications.
Q: What is the substrate scope for the olefin and sulfinate components?
A: The method demonstrates excellent functional group tolerance, accommodating various substituted styrenes (including chloro, methoxy, and trifluoromethyl groups) and diverse aryl sulfinates (such as naphthyl and thiophene derivatives), allowing for the construction of a wide library of complex sulfone architectures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Sulfone Derivative Supplier
The technological advancements detailed in CN112457263A underscore the immense potential of metal-free, aqueous-phase chemistry in modern pharmaceutical manufacturing. At NINGBO INNO PHARMCHEM, we recognize the strategic value of such green methodologies and have integrated similar innovative approaches into our CDMO service portfolio. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries are translated into robust, cost-effective industrial processes. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of aryl sulfone derivative meets the exacting standards required for drug development and commercialization.
We invite forward-thinking R&D and procurement leaders to collaborate with us to leverage this cutting-edge synthesis route for your specific project needs. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your target molecule, evaluating the economic feasibility of switching to this greener protocol. We encourage you to contact us today to request specific COA data for our catalog intermediates or to discuss route feasibility assessments for your proprietary compounds, ensuring a secure and sustainable supply chain for your critical pharmaceutical ingredients.
