Advanced Solid Base Catalysis for High-Purity Antioxidant 1076 Production and Commercial Scale-Up
Advanced Solid Base Catalysis for High-Purity Antioxidant 1076 Production and Commercial Scale-Up
The global demand for high-performance polymer additives continues to surge, driven by the need for durable materials in automotive, packaging, and construction sectors. At the forefront of this industry is Antioxidant 1076, a critical phenolic antioxidant known for its exceptional compatibility and thermal stability. A pivotal advancement in the manufacturing of this essential chemical is detailed in patent CN102020562B, which discloses a novel synthesis method utilizing a solid base catalyst. This technology represents a significant leap forward from traditional homogeneous catalytic processes, addressing long-standing challenges regarding catalyst recovery, product purity, and environmental sustainability. By leveraging a proprietary solid base catalyst derived from porous hydroxyapatite, manufacturers can now achieve yields exceeding 85% with purity levels greater than 97.5%. This report provides a deep technical analysis of this breakthrough, offering strategic insights for R&D directors seeking process optimization and procurement leaders focused on cost-effective supply chains.
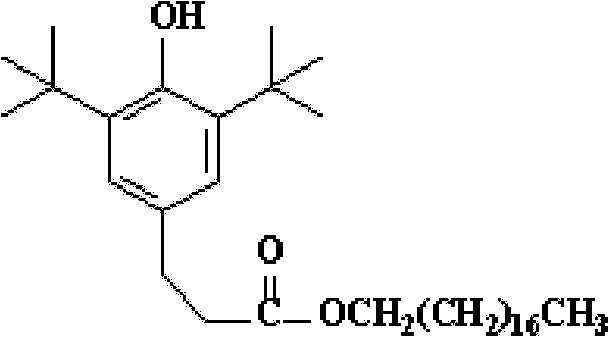
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Antioxidant 1076 has relied heavily on transesterification reactions catalyzed by homogeneous systems. Common catalysts cited in prior art include calcium oxide (CaO), organic tin compounds, tetrabutyl titanate, aluminum triisopropoxide, and lithium acetate. While these catalysts can facilitate the reaction between methyl 3-(3,5-di-tert-butyl-4-hydroxyphenyl)propionate and octadecanol, they suffer from severe operational drawbacks. The primary issue is the difficulty in separating the catalyst from the final product mixture, often necessitating complex neutralization, washing, and purification steps that generate substantial wastewater. Furthermore, the catalytic efficiency of some traditional bases like CaO is often insufficient, leading to inconsistent yields and variable product quality. More effective homogeneous catalysts, such as organic tin or titanium derivatives, are frequently expensive and cannot be recovered, resulting in a linear increase in raw material costs and a heavier environmental burden due to heavy metal residues.
The Novel Approach
In stark contrast to these legacy methods, the technology outlined in patent CN102020562B introduces a heterogeneous solid base catalyst system that fundamentally transforms the reaction landscape. This innovative approach utilizes a self-prepared solid base catalyst where porous hydroxyapatite serves as a carrier, impregnated with alkaline solutions such as strontium nitrate, tetramethylammonium hydroxide, or potassium hydroxide. The reaction proceeds smoothly under moderate thermal conditions, typically between 100°C and 130°C, ensuring energy efficiency while maintaining high reaction rates. The most transformative aspect of this novel approach is the physical state of the catalyst; being a solid, it can be effortlessly separated from the liquid reaction mixture via simple filtration. This not only streamlines the post-reaction workup but also enables the catalyst to be recycled and reused multiple times without significant loss of activity. As illustrated in the reaction scheme below, this clean transesterification pathway directly converts the methyl ester and octadecanol into the target antioxidant with minimal byproduct formation.
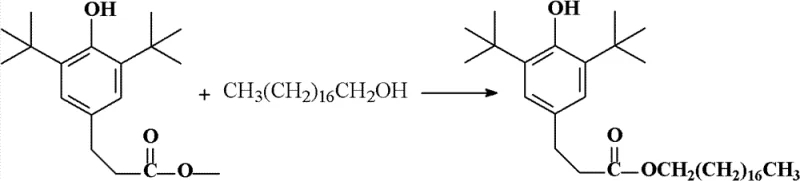
Mechanistic Insights into Solid Base Catalyzed Transesterification
The efficacy of this synthesis route lies in the unique physicochemical properties of the modified hydroxyapatite catalyst. Hydroxyapatite possesses a high surface area and specific pore structure that, when impregnated with strong alkaline species, creates highly active basic sites on the solid surface. During the transesterification process, these basic sites activate the carbonyl group of the methyl 3-(3,5-di-tert-butyl-4-hydroxyphenyl)propionate, facilitating a nucleophilic attack by the hydroxyl group of the octadecanol. The porous nature of the support ensures excellent dispersion of the active alkaline components, maximizing the contact area between the reactants and the catalyst. This heterogeneity prevents the leaching of active species into the bulk solution, which is a common failure mode in other solid catalysts. Consequently, the reaction kinetics are significantly improved compared to weaker solid bases like unmodified CaO, allowing the process to reach completion within a relatively short timeframe of 2 to 5 hours. The robustness of the catalyst structure also withstands the thermal stress of the reaction environment, maintaining its integrity over multiple cycles.
From an impurity control perspective, this mechanistic advantage translates directly into superior product quality. In homogeneous catalysis, residual metal ions often remain trapped in the product matrix, requiring aggressive washing that can lead to product loss or hydrolysis. With the solid base system, the catalyst is physically removed before the crystallization step. The process involves cooling the reaction mixture to room temperature, adding ethanol as a solvent to induce precipitation, and filtering off the solid catalyst. The filtrate is then cooled further to 10-15°C to crystallize the pure Antioxidant 1076. This sequence effectively isolates the product from the catalytic residue, ensuring that the final material meets stringent purity specifications of ≥97.5% without the need for extensive downstream purification. The absence of soluble metal contaminants is particularly critical for applications in food packaging and medical-grade polymers, where regulatory compliance regarding heavy metals is non-negotiable.
How to Synthesize Antioxidant 1076 Efficiently
Implementing this advanced synthesis route requires precise control over catalyst preparation and reaction parameters to maximize yield and purity. The process begins with the meticulous preparation of the solid base catalyst, involving the impregnation of porous hydroxyapatite followed by controlled calcination. Once the catalyst is ready, the transesterification is conducted in a reactor equipped with heating, stirring, and reflux capabilities. The molar ratio of the methyl ester substrate to octadecanol is carefully adjusted, typically ranging from 1:1 to 1:1.2, to drive the equilibrium towards the product side. The reaction temperature is maintained between 100°C and 130°C, a range that balances reaction speed with energy consumption. Following the reaction period of 2 to 5 hours, the workup procedure leverages the heterogeneous nature of the catalyst for easy separation. For a comprehensive, step-by-step technical guide on executing this synthesis, please refer to the standardized protocol below.
- Prepare the solid base catalyst by impregnating porous hydroxyapatite in an alkaline solution such as Sr(NO3)2, followed by drying and calcination at 400-500°C.
- Conduct the transesterification reaction by mixing methyl 3-(3,5-di-tert-butyl-4-hydroxyphenyl)propionate and octadecanol with the catalyst at 100-130°C for 2-5 hours.
- Cool the reaction mixture, add ethanol to precipitate the product, filter to recover the reusable catalyst, and crystallize the filtrate to obtain pure Antioxidant 1076.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift from homogeneous to solid base catalysis offers profound economic and logistical benefits that extend far beyond simple yield improvements. The adoption of this technology addresses several critical pain points in the manufacturing of polymer additives, specifically regarding operational expenditure and supply continuity. By eliminating the reliance on expensive, single-use homogeneous catalysts like organic tin or titanium esters, manufacturers can achieve substantial cost reductions in raw material procurement. Furthermore, the simplified workup process reduces the consumption of solvents and water required for washing, lowering utility costs and waste disposal fees. These efficiencies collectively contribute to a more competitive pricing structure for the final antioxidant product, providing a distinct market advantage.
- Cost Reduction in Manufacturing: The implementation of a recyclable solid base catalyst fundamentally alters the cost structure of Antioxidant 1076 production. Unlike traditional catalysts that are consumed in the process or require complex neutralization, this solid catalyst can be filtered and reused multiple times, drastically reducing the per-batch catalyst cost. Additionally, the elimination of heavy metal removal steps simplifies the purification process, saving on labor, time, and auxiliary chemicals. This streamlined workflow results in significant operational savings, allowing for more aggressive pricing strategies in the competitive polymer additive market without compromising margins.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness and simplicity of this synthesis method. The reaction conditions are mild and safe, operating at atmospheric pressure and moderate temperatures, which minimizes the risk of process upsets or safety incidents that could disrupt production schedules. The high yield consistency, reported at ≥85% with purity ≥97.5%, ensures predictable output volumes, enabling manufacturers to meet delivery commitments reliably. Moreover, the raw materials—methyl 3-(3,5-di-tert-butyl-4-hydroxyphenyl)propionate and octadecanol—are commodity chemicals with stable supply lines, further securing the production pipeline against raw material volatility.
- Scalability and Environmental Compliance: Scaling this process from pilot to commercial production is straightforward due to the absence of complex separation units typically required for homogeneous catalysts. The solid-liquid separation via filtration is a unit operation that scales linearly and efficiently. From an environmental standpoint, the process is inherently greener; the recyclable catalyst reduces solid waste generation, and the lack of toxic metal residues simplifies wastewater treatment. This alignment with green chemistry principles facilitates easier regulatory compliance and enhances the sustainability profile of the supply chain, a key factor for modern corporate procurement policies focused on ESG criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the solid base catalytic synthesis of Antioxidant 1076. These answers are derived directly from the experimental data and technical specifications provided in patent CN102020562B, ensuring accuracy and relevance for industry professionals evaluating this technology for potential adoption or sourcing.
Q: What are the advantages of using a solid base catalyst over traditional homogeneous catalysts for Antioxidant 1076?
A: Traditional methods often utilize homogeneous catalysts like organic tin or titanium esters which are difficult to separate and generate significant waste. The solid base catalyst described in patent CN102020562B is heterogeneous, allowing for easy filtration and recycling, which drastically reduces production costs and environmental impact while maintaining high catalytic activity.
Q: What purity and yield levels can be achieved with this novel synthesis method?
A: According to the experimental data provided in the patent, this method consistently achieves a product purity of greater than or equal to 97.5% and a yield exceeding 85%. Specific embodiments demonstrated purities up to 99.0% and yields reaching 94.71%, indicating a highly efficient and robust process suitable for high-specification applications.
Q: Is the catalyst reusable, and how does this impact the manufacturing process?
A: Yes, the solid base catalyst is fully recyclable. After the reaction, the catalyst is recovered via simple filtration and can be reused in subsequent batches. This reusability eliminates the need for complex neutralization and washing steps required for homogeneous catalysts, simplifying the workflow and enhancing the overall economic viability of large-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Antioxidant 1076 Supplier
The technological advancements detailed in patent CN102020562B underscore the immense potential for optimizing the production of high-value polymer additives like Antioxidant 1076. At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating such innovative laboratory methodologies into robust, industrial-scale operations. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of solid base catalysis are fully realized in mass manufacturing. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Antioxidant 1076 meets the exacting standards required by the global polymer industry.
We invite procurement leaders and technical directors to collaborate with us to leverage these process efficiencies for your supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring a seamless transition to a more sustainable and cost-effective source of high-purity Antioxidant 1076.
