Revolutionizing Triazine UV Absorbent Production: A Green Ionic Liquid Catalysis Strategy
Revolutionizing Triazine UV Absorbent Production: A Green Ionic Liquid Catalysis Strategy
The global demand for high-performance ultraviolet (UV) stabilizers in polymers and coatings continues to surge, driving the need for more efficient and environmentally compliant synthesis routes for key intermediates. Patent CN110437168B introduces a groundbreaking methodology for preparing 2-(2,4-dihydroxyphenyl)-4,6-bisaryl-1,3,5-triazine, a critical precursor for advanced UV absorbers like UV-1577 and UV-405. This technology fundamentally shifts the paradigm from traditional Lewis acid catalysis to a sustainable tetrafluoroborate ionic liquid system. By replacing corrosive and waste-generating aluminum chloride with recyclable ionic liquids, this process addresses the twin challenges of environmental compliance and cost-efficiency in fine chemical manufacturing. The structural integrity of the target molecule, essential for its light-stabilizing properties, is maintained through mild reaction conditions that minimize degradation.
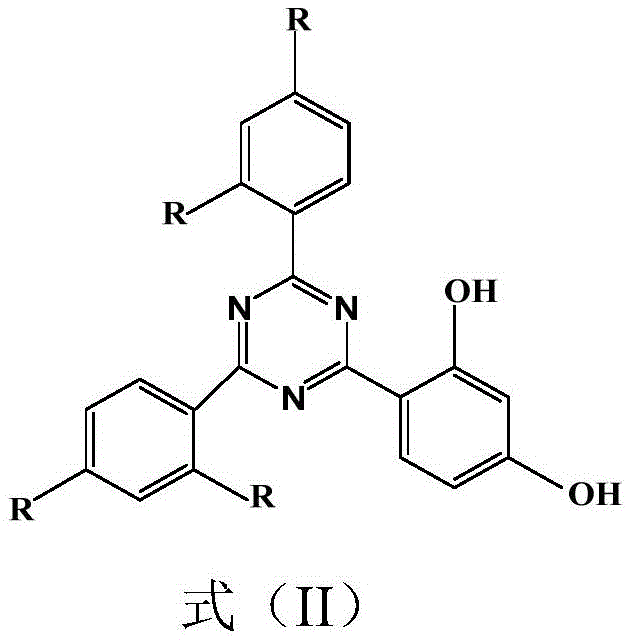
For R&D directors evaluating new supply chains, the purity profile offered by this ionic liquid route is particularly compelling. Traditional methods often struggle with metal contamination and complex purification steps required to remove aluminum salts. In contrast, the novel approach utilizes HCl gas as a precise initiator within an ionic liquid matrix, facilitating a cleaner Friedel-Crafts alkylation. This results in intermediates with purity levels exceeding 99%, significantly reducing the burden on downstream purification processes. The ability to tune the ionic liquid composition, such as using 1-butyl-3-methylimidazolium tetrafluoroborate, allows for optimization of solubility and reaction kinetics, ensuring consistent batch-to-batch quality that meets the rigorous standards of the polymer additive industry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of triazine-based UV absorber intermediates has relied heavily on aluminum chloride (AlCl3) as a catalyst for Friedel-Crafts reactions. As illustrated in the traditional reaction pathways, this approach necessitates stoichiometric or excess amounts of AlCl3, which reacts vigorously to form complexes that are notoriously difficult to decompose. The hydrolysis of these aluminum complexes generates vast quantities of acidic wastewater and solid sludge, creating a severe environmental liability and increasing waste disposal costs exponentially. Furthermore, the recovery of unreacted raw materials from the aluminum sludge is technically challenging and economically unviable, leading to lower overall atom economy. The harsh conditions required often promote side reactions, resulting in impurity profiles that complicate the final crystallization and reduce the yield of the high-value UV absorber precursor.
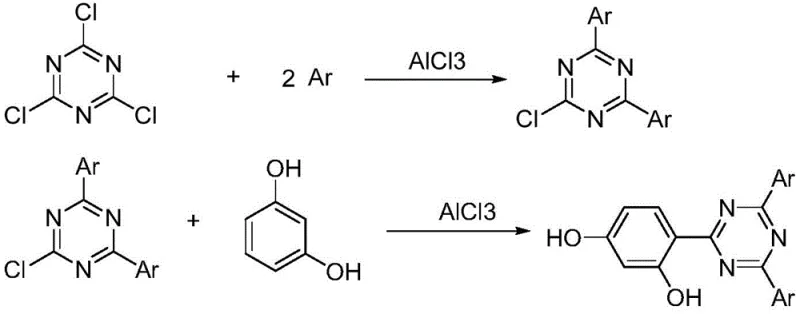
The Novel Approach
The innovative method disclosed in the patent circumvents these issues by employing tetrafluoroborate ionic liquids as a dual-function medium acting as both the solvent and the catalyst. This system operates under much milder conditions, typically between 0°C and 15°C for the initial alkylation, which suppresses unwanted side reactions and preserves the integrity of the triazine ring. The introduction of HCl gas serves as a highly effective initiator, triggering the reaction without the need for massive quantities of solid Lewis acids. Crucially, the ionic liquid phase remains stable throughout the process and can be separated from the product via simple filtration. This separation capability is the key differentiator, allowing the catalyst system to be recycled and reused multiple times—experimental data indicates successful reuse for at least five cycles with minimal loss in catalytic activity. This closed-loop system dramatically reduces the consumption of fresh reagents and eliminates the generation of hazardous aluminum waste.
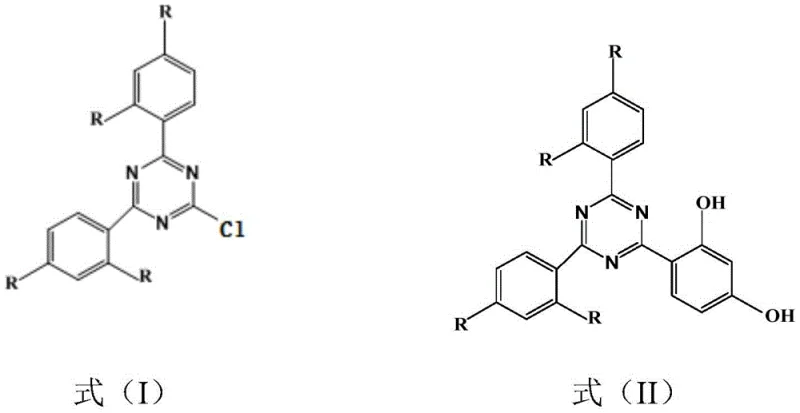
Mechanistic Insights into Ionic Liquid Catalyzed Friedel-Crafts Alkylation
The mechanistic advantage of this process lies in the unique physicochemical properties of the tetrafluoroborate ionic liquids, such as alkyl imidazole tetrafluoroborate and alkyl pyridine tetrafluoroborate. These ionic species possess excellent solvating power for organic reactants like cyanuric chloride and aromatic hydrocarbons, creating a homogeneous reaction environment that enhances mass transfer rates. The cationic nature of the ionic liquid likely stabilizes the carbocation intermediates formed during the electrophilic aromatic substitution, lowering the activation energy required for the alkylation of the triazine ring. Unlike traditional solvents, the ionic liquid does not volatilize, ensuring that the reaction concentration remains constant and preventing the loss of volatile aromatic feedstocks. The specific structure of the ionic liquid, defined by the alkyl chain length on the nitrogen heterocycle, can be tuned to optimize viscosity and polarity, further refining the reaction selectivity towards the desired 2,4,6-substituted product.
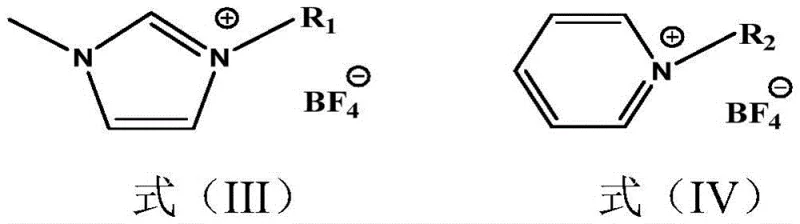
Impurity control is inherently built into this mechanism through the precise regulation of the HCl gas flow. By introducing HCl concurrently with the dropwise addition of the aromatic hydrocarbon, the reaction initiation is synchronized with the availability of the substrate. This prevents the accumulation of reactive intermediates that could otherwise lead to poly-alkylation or ring degradation. The mild temperature range of 5°C to 15°C during the first stage ensures that the exothermic nature of the Friedel-Crafts reaction is managed effectively, preventing thermal runaways that often cause charring or decomposition in batch reactors. Subsequent reaction with resorcinol at slightly elevated temperatures (60-100°C) proceeds smoothly in the same ionic medium, leveraging the existing catalytic environment to drive the substitution of the remaining chlorine atom. This seamless transition between reaction stages without solvent exchange simplifies the operational workflow and minimizes material handling risks.
How to Synthesize 2-(2,4-dihydroxyphenyl)-4,6-bisaryl-1,3,5-triazine Efficiently
The synthesis protocol outlined in the patent offers a streamlined pathway for producing high-purity triazine intermediates suitable for commercial UV stabilizer manufacturing. The process begins with the dissolution of cyanuric chloride in the selected tetrafluoroborate ionic liquid, followed by the controlled addition of benzene or m-xylene under an HCl atmosphere. Detailed operational parameters, including specific molar ratios and temperature ramps, are critical for maximizing yield and minimizing byproduct formation. The following guide summarizes the standardized steps derived from the patent examples to ensure reproducible results in a pilot or production setting.
- React cyanuric chloride with aromatic hydrocarbons (benzene or m-xylene) in the presence of tetrafluoroborate ionic liquid and HCl gas initiator at 0-15°C to form the chloro-intermediate.
- Introduce resorcinol to the reaction mixture and heat to 60-100°C to facilitate the substitution reaction, yielding the final dihydroxyphenyl triazine product.
- Filter the crude product, recycle the ionic liquid filtrate for subsequent batches (up to 5 cycles), and purify the solid using methanol recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the transition to this ionic liquid-based synthesis represents a strategic opportunity to de-risk the supply of critical UV absorber intermediates. The elimination of aluminum chloride removes a major bottleneck associated with hazardous waste disposal and regulatory compliance, which often leads to supply disruptions in traditional manufacturing hubs. By adopting a cleaner technology, suppliers can ensure more consistent production schedules不受 environmental inspections or waste treatment capacity limits. The ability to recycle the expensive ionic liquid catalyst multiple times translates directly into reduced raw material costs over the long term, providing a buffer against volatility in chemical feedstock prices. This process stability enhances the reliability of the supply chain, ensuring that downstream polymer manufacturers receive their UV stabilizer ingredients on time and with consistent quality specifications.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the drastic reduction in waste treatment expenses. Traditional aluminum chloride processes generate significant volumes of acidic wastewater that require neutralization and specialized disposal, incurring high operational costs. By replacing this with a recyclable ionic liquid system, the facility eliminates these waste streams entirely. Furthermore, the catalyst reuse capability means that the effective cost per kilogram of catalyst consumed is a fraction of that in single-use systems. Although the initial investment in ionic liquids may be higher than bulk aluminum chloride, the lifecycle cost is significantly lower due to the extended utility of the catalytic medium and the simplified workup procedure which reduces labor and energy consumption.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the logistical challenges of handling corrosive and moisture-sensitive reagents like aluminum chloride and Grignard reagents. The ionic liquid method utilizes stable, non-volatile liquids that are easier to store and transport, reducing the risk of supply interruptions due to hazardous material shipping restrictions. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, allowing for greater flexibility in sourcing aromatic hydrocarbons. This resilience ensures that production targets can be met even when facing minor supply chain fluctuations, providing a competitive advantage in meeting Just-In-Time delivery commitments for global polymer clients.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies environmental risks, but this green chemistry approach scales linearly without increasing the toxic load. The absence of heavy metal contaminants in the final product simplifies the regulatory approval process for UV absorbers used in food-contact materials or medical devices. The closed-loop nature of the ionic liquid system aligns perfectly with modern sustainability goals and carbon footprint reduction initiatives. Facilities implementing this technology can market their products as 'green' or 'sustainably produced,' adding value for end-users who are increasingly pressured to demonstrate environmental stewardship in their own supply chains. This alignment with ESG (Environmental, Social, and Governance) criteria future-proofs the manufacturing asset against tightening environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ionic liquid catalysis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the feasibility of adoption. Understanding these nuances is essential for technical teams assessing the integration of this method into existing production lines.
Q: How does the ionic liquid method improve upon traditional aluminum chloride catalysis?
A: Traditional methods generate massive amounts of acidic wastewater and aluminum sludge that are difficult to treat. The ionic liquid method acts as both solvent and catalyst, allowing for simple filtration recovery and reuse up to 5 times without significant loss of activity, drastically reducing waste treatment costs.
Q: What represents the critical quality control parameter in this synthesis?
A: The purity of the intermediate 2-chloro-4,6-bisaryl-1,3,5-triazine is critical. The patent demonstrates that controlling the HCl gas initiation rate and temperature (0-15°C) minimizes side reactions, ensuring the final UV absorber intermediate achieves >99% purity.
Q: Is this process scalable for industrial production of UV stabilizers?
A: Yes, the process eliminates the handling hazards associated with large quantities of aluminum chloride and Grignard reagents. The ability to recycle the ionic liquid solvent system makes it economically viable for large-scale manufacturing of UV-1577 and UV-405 precursors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(2,4-dihydroxyphenyl)-4,6-bisaryl-1,3,5-triazine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the shift towards greener, more efficient synthesis routes is not just a regulatory requirement but a strategic imperative for the fine chemical industry. Our expertise in scaling diverse pathways from 100 kgs to 100 MT/annual commercial production positions us as an ideal partner for bringing this advanced ionic liquid technology to the market. We possess the rigorous QC labs and stringent purity specifications necessary to validate the high-quality standards demanded by the UV stabilizer sector. Our team is dedicated to optimizing these novel catalytic systems to ensure that every batch of triazine intermediate meets the exacting requirements for light stability and color performance in final polymer applications.
We invite you to collaborate with us to leverage this cutting-edge synthesis method for your specific UV absorber needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this waste-free process for your operations. We encourage you to reach out for specific COA data and route feasibility assessments tailored to your volume requirements. Let us help you secure a sustainable, high-purity supply of 2-(2,4-dihydroxyphenyl)-4,6-bisaryl-1,3,5-triazine that drives value and innovation in your product portfolio.
