Advanced Synthesis and Commercial Scalability of Benzothiazole-Based Tie2 Inhibitor Intermediates
Advanced Synthesis and Commercial Scalability of Benzothiazole-Based Tie2 Inhibitor Intermediates
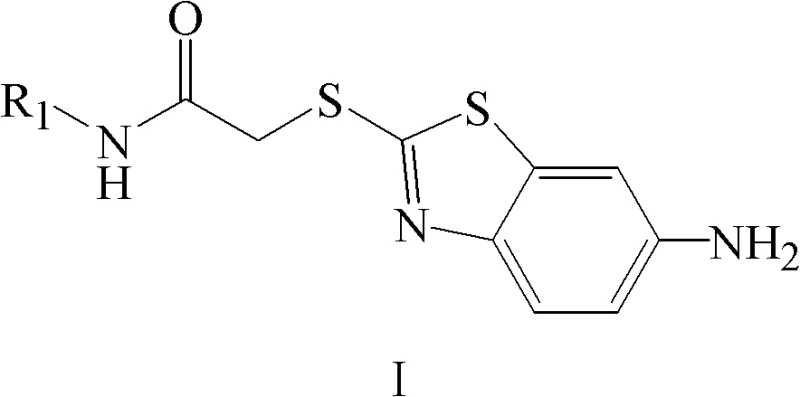
The development of efficient and safe antitumor drugs remains a paramount challenge in modern medicinal chemistry, particularly in the realm of targeted therapy. Patent CN102127067B introduces a significant advancement in this field by disclosing a novel class of 2-(6-aminobenzothiazole-2-mercapto)-acetamide derivatives. These compounds function as potent inhibitors of the Tie2 receptor tyrosine kinase, a critical target in disrupting tumor angiogenesis. For pharmaceutical developers and procurement specialists, understanding the synthetic accessibility and structural versatility of these intermediates is crucial for accelerating oncology drug pipelines. This report provides a deep technical analysis of the patented methodology, highlighting its potential for cost reduction in API manufacturing and its reliability as a source for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of complex heterocyclic systems containing both benzothiazole and amide functionalities often involves multi-step sequences that suffer from low overall yields and harsh reaction conditions. Conventional routes may require the use of expensive transition metal catalysts for C-S or C-N bond formation, which introduces significant challenges in downstream processing, specifically regarding the removal of heavy metal residues to meet stringent regulatory standards for pharmaceutical ingredients. Furthermore, older methodologies frequently rely on protecting group strategies that add unnecessary steps, increase solvent consumption, and extend production lead times. These inefficiencies not only inflate the cost of goods but also complicate the supply chain continuity for clinical trial materials, making the search for more direct and atom-economical pathways a priority for R&D directors.
The Novel Approach
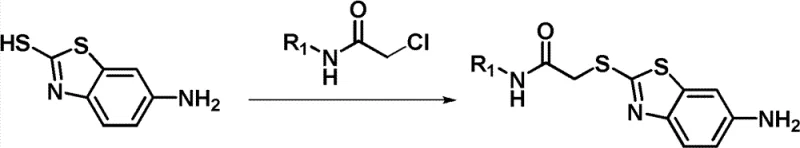
The methodology described in CN102127067B offers a streamlined alternative by utilizing a direct nucleophilic substitution strategy. The core innovation lies in the alkylation of 6-amino-2-mercaptobenzothiazole with pre-formed chloroacetamide derivatives. This approach bypasses the need for complex catalytic cycles and allows for the rapid assembly of the pharmacophore under relatively mild conditions. By employing readily available starting materials such as substituted amines and chloroacetyl chloride, the process significantly simplifies the synthetic tree. The reaction proceeds efficiently in common organic solvents like acetone or dichloromethane, utilizing inexpensive inorganic or organic bases. This shift towards a more convergent synthesis strategy not only enhances the overall yield but also improves the impurity profile, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Nucleophilic Substitution and Amide Formation
The chemical transformation at the heart of this patent involves a classic SN2 nucleophilic substitution mechanism, optimized for heterocyclic substrates. The sulfur atom of the 6-amino-2-mercaptobenzothiazole acts as a potent nucleophile, attacking the electrophilic methylene carbon of the chloroacetamide moiety. The presence of the electron-withdrawing carbonyl group adjacent to the leaving chloride enhances the electrophilicity of the methylene carbon, facilitating the displacement reaction. The use of bases such as potassium carbonate or triethylamine serves to deprotonate the thiol group, generating the more reactive thiolate anion in situ. This mechanistic pathway is highly robust and tolerant of various substituents on the amine nitrogen (R1), allowing for the introduction of diverse heterocyclic systems like pyridines, pyrimidines, and thiazoles without compromising reaction efficiency.
Impurity control is inherently managed through the stoichiometry and reaction conditions specified in the patent. By carefully controlling the molar ratios of the chloroacetamide precursor and the benzothiazole core, side reactions such as over-alkylation or hydrolysis of the chloroacetyl group are minimized. The subsequent purification steps, which often involve aqueous extraction to remove salt byproducts followed by silica gel chromatography or recrystallization, effectively separate the desired product from unreacted starting materials and minor side products. The patent data indicates that this rigorous control leads to products with purity levels exceeding 95%, as confirmed by HPLC analysis, ensuring that the material is suitable for subsequent biological evaluation and potential therapeutic application.
How to Synthesize 2-(6-Aminobenzothiazole-2-mercapto)-acetamide Efficiently
The synthesis protocol outlined in the patent provides a reproducible framework for generating these valuable intermediates. The process begins with the preparation of the chloroacetamide building block, followed by the coupling with the benzothiazole core. This modular approach allows for the easy variation of the R1 group to explore structure-activity relationships. The detailed standardized synthesis steps see the guide below ensure consistent quality and yield across different batches.
- Prepare the chloroacetamide precursor by reacting a substituted amine with chloroacetyl chloride in dichloromethane under basic conditions.
- Perform the key alkylation by refluxing 6-amino-2-mercaptobenzothiazole with the prepared chloroacetamide in acetone using a base like potassium carbonate.
- For further functionalization, react the resulting amino-intermediate with an acid chloride in dichloromethane to yield the final di-substituted derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the synthetic route described in CN102127067B presents substantial advantages over traditional methods. The reliance on commodity chemicals such as chloroacetyl chloride, substituted amines, and common bases eliminates the dependency on scarce or geopolitically sensitive reagents. This abundance of raw materials ensures a stable supply chain and mitigates the risk of production delays caused by material shortages. Furthermore, the avoidance of precious metal catalysts removes the need for specialized scavenging resins and extensive analytical testing for metal residues, leading to significant cost savings in the manufacturing process.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and complex protecting group manipulations drastically reduces the raw material costs associated with production. The use of simple solvents like acetone and dichloromethane, which are easily recovered and recycled, further lowers the operational expenditure. Additionally, the high yields reported in the examples, often exceeding 90%, minimize waste generation and maximize the output per batch, contributing to a more economical production model for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The synthetic pathway utilizes widely available starting materials that are produced by multiple global suppliers, reducing the risk of single-source dependency. The robustness of the reaction conditions, which do not require extreme temperatures or pressures, allows for manufacturing in a broader range of facilities, enhancing supply chain flexibility. This reliability is critical for maintaining continuous production schedules and meeting the demanding timelines of drug development projects.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the successful execution of reactions on gram scales in the patent examples without loss of efficiency. The straightforward workup procedures, involving filtration and crystallization, are easily adaptable to large-scale industrial equipment. Moreover, the reduced use of hazardous reagents and the potential for solvent recycling align with green chemistry principles, facilitating compliance with increasingly stringent environmental regulations and reducing the burden of waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these benzothiazole derivatives. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for industry professionals.
Q: What is the primary biological target of these benzothiazole derivatives?
A: These compounds are designed as small molecule antagonists targeting the Tie2 receptor tyrosine kinase, which plays a critical role in tumor angiogenesis and endothelial cell migration.
Q: Are the synthesis conditions suitable for large-scale manufacturing?
A: Yes, the process utilizes common solvents like acetone and dichloromethane with standard bases such as triethylamine or potassium carbonate, avoiding expensive transition metal catalysts and facilitating easy scale-up.
Q: How is product purity ensured in this synthetic route?
A: The protocol includes robust purification steps such as aqueous workups, silica gel column chromatography, and recrystallization, consistently achieving purity levels above 95% as verified by HPLC and NMR analysis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(6-Aminobenzothiazole-2-mercapto)-acetamide Derivatives Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis and manufacturing for the pharmaceutical industry, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of benzothiazole chemistry, ensuring stringent purity specifications and rigorous QC labs validate every batch against the highest international standards. We understand the critical nature of oncology research and are committed to providing reliable support for your drug development programs.
We invite you to contact our technical procurement team to discuss your specific requirements for these antitumor intermediates. By partnering with us, you can request a Customized Cost-Saving Analysis tailored to your project's scale and timeline. Reach out today to obtain specific COA data and route feasibility assessments that will empower your R&D and supply chain strategies.
