Revolutionizing Agrochemical Intermediates: A Green Solvent-Free Synthesis for Beta-Chloro Dicarbonyls
Revolutionizing Agrochemical Intermediates: A Green Solvent-Free Synthesis for Beta-Chloro Dicarbonyls
The chemical industry is currently witnessing a pivotal shift towards greener, more efficient synthetic methodologies, particularly in the realm of agrochemical intermediates where cost and environmental compliance are paramount. Patent CN112574141B introduces a groundbreaking solvent-free synthesis method for beta-chloro alpha,gamma-dicarbonyl compounds, utilizing chlorine gas and anhydrous aluminum chloride as a catalyst system. This innovation addresses critical pain points in traditional chlorination processes, such as the generation of hazardous sulfur dioxide byproducts and the reliance on expensive chlorinating reagents like sulfuryl chloride. By operating under mild conditions between 20°C and 50°C without the need for organic solvents, this technology offers a robust pathway for producing high-purity intermediates essential for the next generation of organochlorine pesticides. For R&D directors and supply chain managers, this represents a significant opportunity to optimize manufacturing protocols while adhering to stricter environmental regulations.
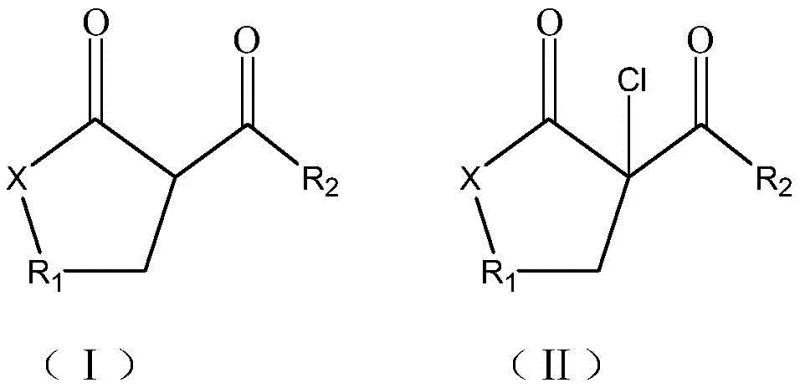
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of beta-chloro alpha,gamma-dicarbonyl compounds has relied heavily on chlorinating agents such as sulfuryl chloride or solid phosgene, which present substantial logistical and environmental challenges. These traditional reagents are not only costly to procure and handle due to their toxicity and instability but also generate stoichiometric amounts of sulfur dioxide gas as a byproduct, necessitating complex scrubbing systems to meet emission standards. Furthermore, conventional methods often suffer from poor selectivity, leading to multi-substitution products at unintended positions on the carbon skeleton, which drastically reduces the overall yield and complicates downstream purification. The requirement for organic solvents in these legacy processes further inflates production costs through solvent purchase, recovery, and disposal, creating a bottleneck for large-scale commercialization of organochlorine pesticide precursors.
The Novel Approach
In stark contrast, the novel approach detailed in the patent leverages elemental chlorine gas activated by anhydrous aluminum chloride under strictly solvent-free conditions to achieve superior reaction control. This methodology fundamentally alters the reaction landscape by eliminating the solvent medium, thereby increasing the effective concentration of reactants and enhancing the reaction rate without the need for excessive heating. The use of chlorine gas, a widely available commodity chemical, significantly lowers raw material costs compared to specialized chlorinating agents, while the absence of sulfur-containing reagents ensures that no sulfur dioxide is produced, aligning perfectly with modern green chemistry principles. The result is a streamlined process where the target beta-chloro product can be isolated with purity exceeding 90% simply through water washing and drying, bypassing the energy-intensive distillation or crystallization steps typically required in older synthetic routes.
Mechanistic Insights into Lewis Acid Catalyzed Chlorination
The core of this technological advancement lies in the precise mechanistic interaction between the anhydrous aluminum chloride catalyst and the chlorine gas, which creates a highly selective electrophilic chlorination environment. Anhydrous aluminum chloride acts as a potent Lewis acid, coordinating with the chlorine molecule to polarize the Cl-Cl bond, thereby generating a more reactive chloronium-like species that selectively targets the active beta-hydrogen of the alpha,gamma-dicarbonyl substrate. This activation mechanism is crucial because the beta-hydrogen in these dicarbonyl systems is inherently acidic and prone to substitution, but without the catalyst, the reaction with molecular chlorine can be uncontrollable, leading to radical pathways and polychlorinated impurities. By fine-tuning the molar ratio of the catalyst to the substrate between 0.03:1 and 0.2:1, the process ensures that the chlorination proceeds via a controlled ionic mechanism, effectively suppressing side reactions and maximizing the conversion to the mono-chlorinated target.
Furthermore, the substrate scope of this reaction is remarkably broad, accommodating various cyclic and acyclic alpha,gamma-dicarbonyl structures as demonstrated by the diverse range of successful examples in the patent data. As illustrated in the structural variations below, the method is effective for cyclopentanone derivatives, butyrolactones, and even cyclohexanone analogues, indicating a high degree of functional group tolerance. The ability to successfully chlorinate substrates with different R groups (such as alkyl, alkoxy, or haloalkyl chains) without compromising yield suggests that the steric and electronic demands of the transition state are well-managed by the catalyst system. This versatility is vital for agrochemical manufacturers who often require a library of structurally related intermediates to develop new pesticide variants, as it allows for a unified synthetic platform rather than requiring bespoke conditions for each new molecule.
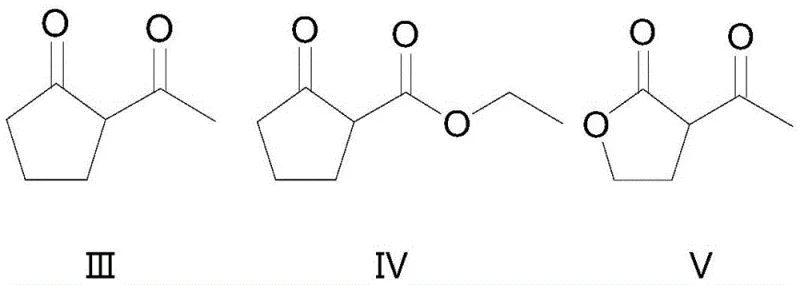
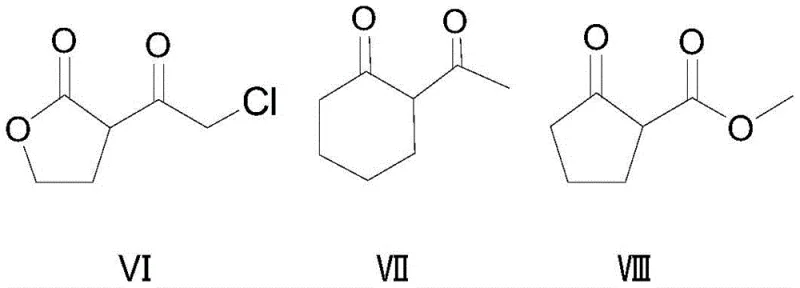
How to Synthesize Beta-Chloro Alpha,Gamma-Dicarbonyl Compounds Efficiently
To implement this synthesis effectively, operators must adhere to strict control parameters regarding temperature and gas flow rates to maintain the delicate balance between reaction speed and selectivity. The patent outlines a procedure where chlorine gas is introduced at a specific rate relative to the catalyst mass (1-3 g/h per gram of catalyst) while maintaining the reaction mixture between 20°C and 50°C, preventing thermal runaway that could lead to degradation. Detailed standardized operating procedures for mixing, gas introduction, and endpoint monitoring via gas chromatography are essential to replicate the high yields (>90%) reported in the experimental examples consistently.
- Prepare the reactor by adding the alpha,gamma-dicarbonyl raw material and anhydrous aluminum chloride catalyst (molar ratio 0.03-0.2: 1) under solvent-free conditions.
- Maintain the reaction temperature between 20-50°C while introducing chlorine gas at a controlled rate (1-3 g/h per gram of catalyst) with continuous stirring.
- Monitor the reaction via gas chromatography until raw material content is below 1%, then perform a simple water wash and drying process to isolate the product with >90% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this solvent-free chlorination technology translates directly into tangible operational efficiencies and risk mitigation strategies. The elimination of organic solvents removes a major variable from the supply chain, reducing dependency on volatile solvent markets and eliminating the capital expenditure associated with solvent recovery infrastructure. Moreover, the switch from expensive, specialized chlorinating agents to commodity chlorine gas drastically reduces the direct material cost per kilogram of product, while the simplified aqueous workup minimizes waste treatment volumes and associated disposal fees. These factors combine to create a manufacturing process that is not only more cost-effective but also more resilient to supply chain disruptions, as the key reagents are globally available and the process equipment requirements are less specialized.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the substitution of high-cost reagents with low-cost commodity chemicals and the removal of solvent-related expenses. By utilizing chlorine gas instead of sulfuryl chloride or phosgene, manufacturers avoid the premium pricing of these specialized reagents and the regulatory costs associated with their transport and storage. Additionally, the solvent-free nature of the reaction means that there is no need to purchase, recover, or dispose of large volumes of organic solvents, which traditionally account for a significant portion of the variable costs in fine chemical synthesis. The simplified post-treatment, which requires only water washing and drying, further reduces utility consumption and labor hours, leading to a leaner, more profitable production model.
- Enhanced Supply Chain Reliability: From a logistics perspective, relying on chlorine gas and aluminum chloride offers a more stable supply base compared to reagents that may be subject to stricter transportation regulations or limited manufacturer capacity. The robustness of the reaction conditions, which tolerate a range of temperatures and do not require cryogenic cooling or high-pressure vessels, allows for greater flexibility in production scheduling and facility utilization. This reliability ensures that downstream customers in the agrochemical sector can maintain consistent inventory levels of critical intermediates, reducing the risk of production stoppages due to raw material shortages or quality inconsistencies often seen with more complex synthetic routes.
- Scalability and Environmental Compliance: The environmental profile of this synthesis is a key driver for its scalability, as it inherently avoids the generation of sulfur dioxide, a regulated pollutant that requires expensive abatement technology. The absence of solvent waste streams simplifies the environmental permitting process for new production lines and reduces the long-term liability associated with hazardous waste management. As global regulations on industrial emissions tighten, having a process that is intrinsically green and compliant provides a competitive advantage, allowing manufacturers to scale production to meet growing demand for organochlorine pesticides without facing regulatory bottlenecks or needing costly retrofits to existing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chlorination technology, derived directly from the patent's experimental data and comparative analysis. Understanding these nuances is critical for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers provided reflect the specific advantages in selectivity, yield, and environmental impact documented in the intellectual property.
Q: What are the primary advantages of using chlorine gas over sulfuryl chloride in this synthesis?
A: Using chlorine gas eliminates the generation of sulfur dioxide (SO2), a significant environmental pollutant associated with sulfuryl chloride. Additionally, chlorine gas is a commodity chemical with lower procurement costs, and the solvent-free nature of the new process removes the need for expensive solvent recovery systems.
Q: How does the anhydrous aluminum chloride catalyst improve selectivity?
A: Anhydrous aluminum chloride acts as a Lewis acid that complexes with chlorine gas, moderating its reactivity. This controlled activation prevents multi-substitution side reactions at other positions, ensuring high selectivity for the beta-position and resulting in product purity exceeding 90% without complex purification steps.
Q: Is this process scalable for industrial production of organochlorine pesticide intermediates?
A: Yes, the process is highly scalable due to its solvent-free design, which simplifies heat transfer and reactor volume requirements. The mild reaction conditions (20-50°C) and simple aqueous workup make it ideal for large-scale manufacturing, reducing both operational complexity and waste treatment costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Chloro Alpha,Gamma-Dicarbonyl Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this solvent-free chlorination technology for the agrochemical industry and are fully equipped to support its commercial deployment. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to full-scale manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of beta-chloro alpha,gamma-dicarbonyl compound meets the exacting standards required for pesticide synthesis, providing our partners with the confidence needed to accelerate their own product development timelines.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific production needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential economic benefits specific to your operation. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise in green chemical synthesis can drive value and sustainability in your supply chain.
