Advanced Synthetic Route for High-Purity Polyhaloacridone Derivatives and Commercial Scale-Up
Advanced Synthetic Route for High-Purity Polyhaloacridone Derivatives and Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for synthesizing complex heterocyclic scaffolds that possess significant biological activity. Patent CN101391981A introduces a groundbreaking synthetic strategy for producing polyhaloacridone compounds, a class of molecules renowned for their potent anticancer and HIV-resistant properties. This technology leverages a concise three-step pathway starting from easily accessible polyhalogenated m-phthalonitriles, offering a reliable polyhaloacridone supplier solution for global markets. The core innovation lies in the efficient construction of the acridone backbone through a mild nucleophilic substitution followed by an acid-mediated cyclization, ensuring high structural integrity and minimal impurity formation. By optimizing reaction conditions such as temperature and solvent choice, this method achieves superior yields compared to traditional routes, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing.
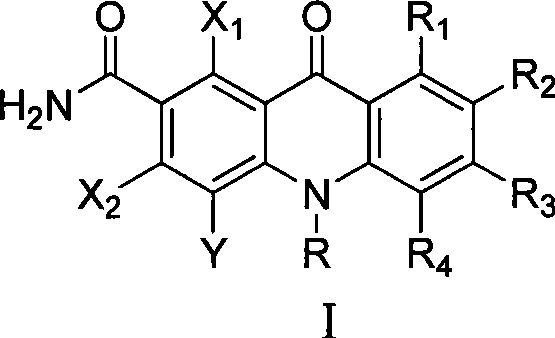
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of dihydroketoacridine derivatives has been plagued by significant challenges regarding reaction severity and substrate scope. Conventional pathways often require harsh reaction conditions, including extremely high temperatures and the use of corrosive reagents that can degrade sensitive functional groups on the aromatic rings. Furthermore, many traditional methods rely on expensive transition metal catalysts or complex multi-step sequences that result in low overall atom economy and substantial waste generation. The introduction of specific halogen atoms, particularly fluorine, which is crucial for enhancing metabolic stability and bioavailability in drug candidates, is often difficult to control in older synthetic protocols. These limitations lead to inconsistent batch-to-batch quality, higher production costs, and extended lead times, creating bottlenecks for supply chain heads who require consistent volumes of high-purity API intermediates for downstream drug development.
The Novel Approach
The methodology disclosed in the patent represents a paradigm shift by utilizing a direct condensation-cyclization strategy that bypasses the need for precious metal catalysis. By reacting polyhalogenated m-phthalonitrile with substituted anilines in aprotic solvents, the process forms a stable intermediate under mild thermal conditions ranging from 0 to 90°C. This approach not only preserves the integrity of delicate substituents like methoxy or amino groups but also allows for the precise installation of multiple halogen atoms on the phenyl ring. The subsequent cyclization step using sulfuric acid is highly efficient, converting the nitrile groups into the requisite amide functionality while closing the central ring system. This streamlined workflow drastically simplifies the purification process, as the intermediates often precipitate directly from the reaction mixture, thereby reducing solvent consumption and processing time significantly.
Mechanistic Insights into Nucleophilic Aromatic Substitution and Cyclization
The chemical foundation of this synthesis rests on a highly selective nucleophilic aromatic substitution (SnAr) mechanism, driven by the strong electron-withdrawing nature of the cyano and halogen groups on the phthalonitrile core. In the initial step, the lone pair of electrons on the nitrogen atom of the aniline derivative attacks the electron-deficient carbon atom of the phthalonitrile, displacing a halogen leaving group such as fluorine or chlorine. This reaction is facilitated by the use of polar aprotic solvents like N,N-dimethylformamide (DMF) or dimethyl sulfoxide (DMSO), which stabilize the transition state and enhance the nucleophilicity of the amine. The presence of base, such as potassium carbonate or triethylamine, further accelerates the reaction by scavenging the generated hydrogen halide, pushing the equilibrium towards the formation of the amino-benzonitrile intermediate. This mechanistic precision ensures that substitution occurs regioselectively, preventing the formation of unwanted isomers that could complicate downstream purification.
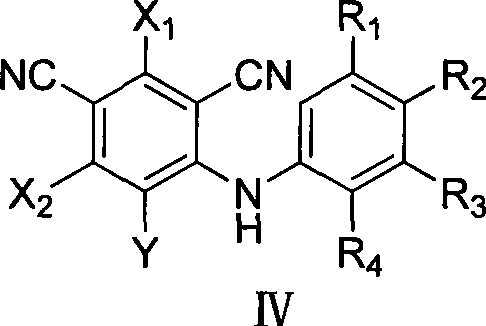
Following the formation of the intermediate, the mechanism shifts to an acid-catalyzed intramolecular cyclization that constructs the rigid acridone skeleton. Upon treatment with concentrated sulfuric acid, one of the nitrile groups undergoes hydrolysis to form an amide, which then attacks the adjacent aromatic ring to close the tricyclic system. This cyclization is thermodynamically driven by the formation of the stable conjugated pi-system characteristic of acridone derivatives. The reaction conditions are carefully tuned to temperatures between 60 and 120°C to ensure complete conversion without degrading the polyhalogenated framework. Finally, the nitrogen atom at the 10-position can be functionalized via alkylation or arylation using strong bases like sodium hydride, allowing for the fine-tuning of the molecule's physicochemical properties. This modular approach provides R&D directors with the flexibility to generate diverse libraries of compounds for structure-activity relationship studies.
How to Synthesize Polyhaloacridones Efficiently
The practical execution of this synthesis involves three distinct operational stages that are designed for scalability and safety in a commercial plant environment. The process begins with the condensation of the phthalonitrile and aniline components, followed by the critical cyclization step, and concludes with the N-substitution to finalize the molecular architecture. Each stage has been optimized to maximize yield and minimize waste, utilizing standard chemical engineering unit operations. For detailed procedural specifics regarding stoichiometry, workup procedures, and quality control checkpoints, please refer to the standardized synthesis guide below.
- React polyhalogenated m-phthalonitrile with an aniline derivative in an aprotic solvent at 0-90°C to form the amino-benzonitrile intermediate.
- Treat the intermediate with concentrated sulfuric acid at 60-120°C to induce intramolecular cyclization, forming the dihydroacridone core.
- Perform N-alkylation or arylation on the dihydroacridone nitrogen using alkali and halohydrocarbons to yield the final polyhaloacridone product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthetic route offers substantial economic benefits by eliminating the reliance on scarce and expensive noble metal catalysts often found in cross-coupling reactions. The primary raw materials, polyhalogenated phthalonitriles and substituted anilines, are commodity chemicals available from multiple global suppliers, which mitigates supply risk and stabilizes pricing volatility. The use of common industrial solvents like DMF and ethyl acetate further reduces operational expenditures, as these can be easily recovered and recycled within a closed-loop system. Additionally, the high yields reported in the patent embodiments, often exceeding 80% for the initial steps, translate directly into lower cost of goods sold (COGS) and improved margin potential for the final active pharmaceutical ingredient. This efficiency makes the technology highly attractive for cost reduction in pharmaceutical intermediate manufacturing, allowing companies to allocate resources to other critical areas of drug development.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes the need for costly and time-consuming metal scavenging steps, which are typically required to meet stringent regulatory limits for residual metals in drug substances. Furthermore, the high atom economy of the condensation reaction ensures that the majority of the starting material mass is incorporated into the final product, minimizing raw material waste. The ability to isolate intermediates via simple filtration rather than complex chromatography significantly lowers labor and solvent costs associated with purification. These factors combine to create a lean manufacturing process that delivers significant cost savings without compromising on the quality or purity of the output.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which tolerate a wide range of temperatures and do not require inert atmospheres for all steps, enhances the reliability of production schedules. Since the raw materials are chemically stable and widely produced, the risk of supply disruption due to raw material shortages is significantly minimized compared to routes relying on bespoke reagents. The scalability of the process from gram-scale laboratory experiments to multi-ton commercial production has been demonstrated through the use of standard reactor types, ensuring that supply chain heads can confidently plan for long-term inventory needs. This stability is crucial for maintaining continuous manufacturing lines and meeting the just-in-time delivery requirements of major pharmaceutical clients.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste, as the primary byproduct is inorganic salt which can be treated using standard wastewater management protocols. The avoidance of chlorinated solvents in favor of greener alternatives like ethyl acetate for recrystallization aligns with modern environmental, health, and safety (EHS) standards. The high conversion rates reduce the volume of unreacted starting materials that need to be disposed of, lowering the overall environmental footprint of the manufacturing site. This compliance with green chemistry principles not only reduces disposal costs but also enhances the corporate sustainability profile, which is increasingly important for securing contracts with environmentally conscious multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these specialized chemical intermediates. The answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing production pipelines.
Q: What are the key advantages of this polyhaloacridone synthesis route?
A: The process utilizes readily available raw materials like polyhalogenated phthalonitriles and operates under relatively mild conditions (0-90°C for the first step), avoiding expensive transition metal catalysts and ensuring high yields up to 92%.
Q: How is purity controlled in the production of these intermediates?
A: Purity is managed through precise temperature control during the sulfuric acid cyclization step and rigorous recrystallization processes using solvents like acetone or ethyl acetate, which effectively remove unreacted starting materials and byproducts.
Q: Is this method scalable for industrial manufacturing?
A: Yes, the synthesis relies on common industrial solvents such as DMF and DMSO and standard unit operations like filtration and neutralization, making it highly suitable for commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polyhaloacridones Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the laboratory bench to full-scale manufacturing. We adhere to stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch meets the highest international standards. Our commitment to technical excellence allows us to handle complex synthetic challenges, such as the polyhalogenation and cyclization steps described in this patent, with precision and consistency.
We invite you to collaborate with us to leverage this advanced synthetic technology for your specific drug development programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and timeline. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can accelerate your path to market while optimizing your supply chain efficiency.
