Revolutionizing Axial Chiral Anilide Production: A New Era for Pharmaceutical Intermediate Manufacturing
The landscape of asymmetric synthesis is undergoing a significant transformation with the disclosure of patent CN112479920B, which introduces a groundbreaking methodology for constructing axial chiral anilide compounds. This intellectual property represents a pivotal advancement in the field of atroposelective chemistry, specifically addressing the long-standing challenges associated with the C-N chiral axis. Traditionally, the synthesis of such motifs has been hindered by low rotational barriers and the difficulty in controlling stereochemistry during bond formation. However, this new technical solution leverages a desymmetrized Sonogashira coupling strategy that fundamentally shifts the paradigm from N-terminal to C-terminal functionalization. For R&D directors and process chemists seeking reliable axial chiral anilide supplier capabilities, this patent offers a robust pathway to access high-value scaffolds that are critical for next-generation bioactive molecules and chiral ligands. The ability to generate these structures with exceptional enantioselectivity opens new doors for drug discovery programs targeting complex biological pathways where chirality dictates efficacy.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of axially chiral anilides has relied heavily on strategies that are inherently limited in scope and efficiency. Early approaches often utilized chiral pool strategies, which necessitated the use of stoichiometric amounts of expensive chiral reagents, thereby driving up costs and generating significant waste. Furthermore, many catalytic methods reported in the prior art, such as Pd-catalyzed N-allylation or N-arylation, focused on modifying the nitrogen atom or its existing substituents. These N-terminal functionalization routes frequently struggle with the low rotational barrier of the C-N axis compared to biaryl analogs, making it difficult to lock the conformation and maintain high optical purity. Additionally, some existing C-terminal functionalization methods have proven difficult to reproduce across different substrate scopes, limiting their utility in diverse medicinal chemistry campaigns. These constraints have created a bottleneck in the commercial scale-up of complex chiral ligands and pharmaceutical intermediates, forcing manufacturers to rely on inefficient resolution processes or multi-step sequences that erode overall yield.
The Novel Approach
In stark contrast to these legacy techniques, the methodology outlined in CN112479920B establishes a new direction by targeting the C-terminal functional group through a catalytic desymmetrization process. By employing a di-iodo aniline derivative as the substrate and reacting it with a terminal alkyne in the presence of a palladium catalyst, copper co-catalyst, and a specialized chiral phosphine ligand, the reaction achieves precise kinetic resolution. This approach effectively bypasses the issues associated with N-terminal reactivity and leverages the steric bulk of the incoming alkyne to induce axial chirality with remarkable fidelity. The result is a streamlined, one-step catalytic process that delivers atropisomeric anilide compounds with very high enantiomeric excess, often exceeding 95% ee. This innovation not only simplifies the synthetic route but also enhances the structural diversity accessible to chemists, allowing for the rapid exploration of chemical space in the search for new therapeutic agents and advanced materials.
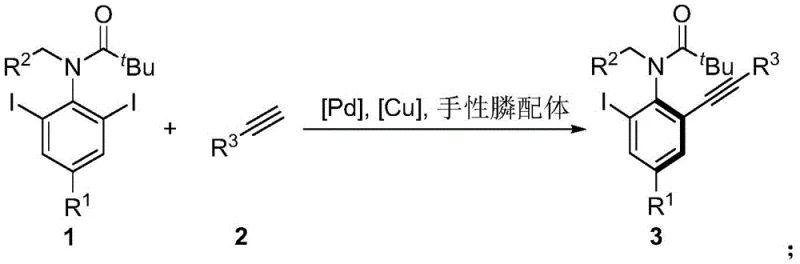
Mechanistic Insights into Desymmetrized Sonogashira Coupling
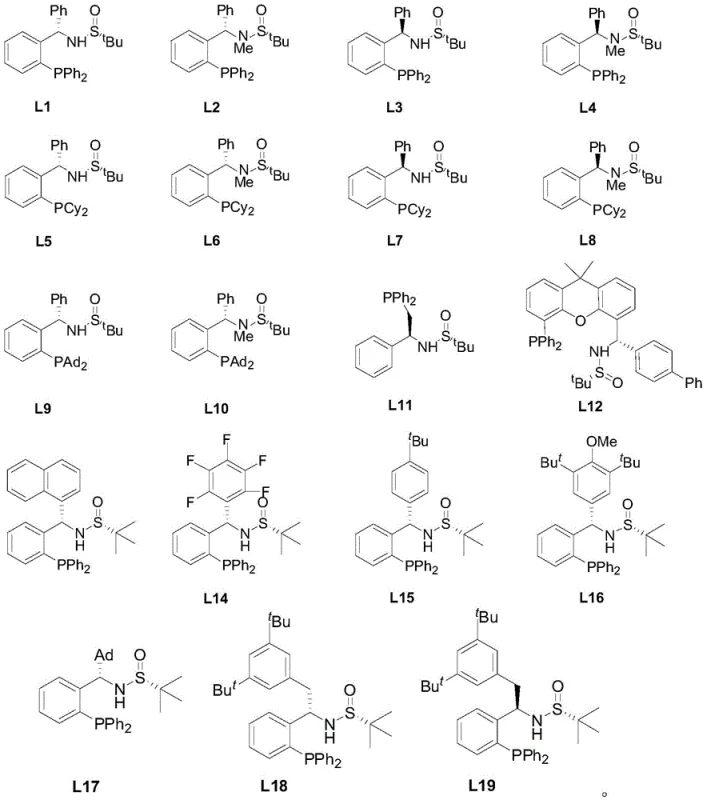
The core of this technological breakthrough lies in the intricate interplay between the palladium catalyst and the chiral phosphine ligand library (L1-L19). The mechanism initiates with the oxidative addition of the palladium species to one of the carbon-iodine bonds on the symmetric di-iodo substrate. The chiral environment created by the bulky phosphine ligand, such as L19, dictates the facial selectivity of the subsequent transmetallation with the copper-acetylide species. This step is critical, as it determines the trajectory of the alkyne approach and ultimately locks the rotation around the emerging C-N axis. The steric congestion introduced by the ortho-substituents on the aniline ring, combined with the bulky alkyne moiety, creates a high rotational barrier that stabilizes the atropisomer. Following reductive elimination, the chiral product is released, and the catalytic cycle regenerates. This precise control over the transition state geometry ensures that the formation of the desired enantiomer is heavily favored over its mirror image, resulting in the high optical purity observed in the experimental data.
From an impurity control perspective, this catalytic system offers distinct advantages over non-catalytic or resolution-based methods. Because the chirality is induced dynamically during the bond-forming event rather than separated post-synthesis, the formation of the undesired enantiomer is suppressed at the source. The use of mild reaction temperatures, typically ranging from 0°C to 25°C, further minimizes thermal racemization, which is a common risk for atropisomers with lower rotational barriers. Moreover, the specificity of the Sonogashira coupling reduces the formation of homocoupling byproducts (Glaser coupling) when optimized copper levels are used. This high level of chemoselectivity and stereoselectivity translates directly into a cleaner crude reaction profile, simplifying downstream purification and ensuring that the final high-purity pharmaceutical intermediates meet stringent regulatory specifications without the need for extensive recrystallization or chiral HPLC separation steps.
How to Synthesize Axial Chiral Anilide Efficiently
The practical implementation of this synthesis route is designed to be accessible for laboratory scale-up while maintaining the rigor required for industrial translation. The process begins with the activation of the catalyst system, where a palladium salt and the chosen chiral ligand are pre-stirred to ensure the formation of the active catalytic species. Subsequently, the substrates and base are introduced under inert atmosphere to prevent catalyst deactivation. The reaction proceeds under mild thermal conditions, allowing for excellent control over the exotherm and reaction kinetics. For detailed operational parameters, including specific molar ratios, solvent choices, and workup procedures that guarantee optimal yield and enantioselectivity, please refer to the standardized protocol below.
- Pre-stir a palladium salt catalyst (e.g., Pd(OAc)2) and a chiral phosphine ligand (e.g., L19) in anhydrous solvent under nitrogen protection for 30-60 minutes.
- Add copper salt catalyst (e.g., CuI), di-iodo substrate, terminal alkyne substrate, and a base (e.g., cesium acetate) to the mixture.
- Stir the reaction mixture at low temperature (0-25°C) for 18-108 hours, then concentrate and purify via column chromatography to isolate the chiral product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthetic route presents a compelling value proposition centered on efficiency and reliability. Traditional methods for producing axially chiral compounds often involve lengthy linear syntheses or expensive chiral resolution steps that introduce significant variability into the supply chain. By contrast, this desymmetrized coupling strategy consolidates multiple synthetic transformations into a single catalytic step, drastically reducing the number of unit operations required. This simplification leads to substantial cost savings in manufacturing by minimizing solvent consumption, reducing labor hours, and lowering the overall equipment footprint. Furthermore, the reliance on readily available starting materials, such as substituted di-iodo anilines and terminal alkynes, mitigates the risk of raw material shortages and price volatility, ensuring a more stable and predictable supply of critical intermediates for downstream drug production.
- Cost Reduction in Manufacturing: The elimination of stoichiometric chiral auxiliaries and the shift to a catalytic asymmetric process significantly lowers the cost of goods sold (COGS). By avoiding the need for expensive chiral pool reagents and reducing the waste associated with resolution processes, manufacturers can achieve a leaner production model. The high atom economy of the Sonogashira coupling, combined with the ability to recover and recycle precious metal catalysts, further contributes to economic efficiency. This process optimization allows for competitive pricing strategies without compromising on the quality or purity of the final product, making it an attractive option for cost-sensitive pharmaceutical projects.
- Enhanced Supply Chain Reliability: The robustness of this catalytic system enhances supply chain resilience by reducing dependency on niche reagents that may have long lead times. The substrates employed in this reaction are commodity chemicals or easily synthesized derivatives, which ensures consistent availability from multiple global suppliers. Additionally, the mild reaction conditions reduce the risk of batch failures due to thermal runaway or sensitivity issues, leading to higher first-pass yields and more reliable delivery schedules. This stability is crucial for maintaining continuous manufacturing flows and meeting the tight deadlines often imposed by clinical trial timelines and regulatory filings.
- Scalability and Environmental Compliance: From an environmental and safety standpoint, this methodology aligns well with green chemistry principles. The use of mild temperatures and potentially greener solvents like ethyl acetate reduces the energy intensity of the process and minimizes the generation of hazardous waste. The high selectivity of the reaction reduces the burden on waste treatment facilities by lowering the load of organic byproducts. As regulatory pressures regarding environmental compliance increase, adopting such efficient and clean technologies positions companies favorably for future audits and sustainability certifications, facilitating smoother market access and long-term operational viability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this axial chiral anilide synthesis technology. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on the practical aspects of the process. Understanding these details is essential for evaluating the feasibility of integrating this methodology into existing production pipelines or R&D workflows.
Q: What is the primary advantage of this new synthesis method over traditional approaches?
A: Unlike traditional methods that often rely on N-terminal functionalization or chiral pool strategies requiring equivalent chiral reagents, this patent describes a catalytic desymmetrized Sonogashira coupling at the C-terminal. This approach establishes a new direction for preparing atropisomeric anilides, offering significantly higher enantioselectivity (up to 98% ee) and avoiding the low rotational barrier issues associated with C-N axis formation in prior art.
Q: Which chiral ligands are most effective for this transformation?
A: The patent details a library of 19 specialized chiral phosphine ligands (L1-L19). Among these, ligand L19 has been demonstrated to provide exceptional stereocontrol across a broad range of substrates, consistently delivering products with high optical purity. The specific choice of ligand allows for fine-tuning the steric environment around the palladium center to maximize asymmetric induction.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the methodology is designed with industrialization in mind. It utilizes mild reaction conditions (0-25°C), commercially available starting materials, and standard palladium/copper catalytic systems. The avoidance of harsh conditions and the use of robust catalytic cycles suggest that the process can be scaled up effectively for the commercial production of complex chiral intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axial Chiral Anilide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the technologies described in CN112479920B for the advancement of chiral drug development. As a premier CDMO partner, we possess the technical expertise and infrastructure necessary to translate these innovative laboratory protocols into robust, GMP-compliant manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from gram-scale optimization to full-scale industrial output. We are committed to delivering products that meet stringent purity specifications, utilizing our rigorous QC labs to verify enantiomeric excess and chemical purity at every stage of production.
We invite you to collaborate with us to leverage this cutting-edge synthesis route for your specific application needs. Whether you require custom synthesis of novel axial chiral ligands or the large-scale production of key pharmaceutical intermediates, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your project volume. We encourage you to reach out for specific COA data and route feasibility assessments to determine how we can optimize your supply chain and accelerate your time to market with high-quality chiral building blocks.
