Scalable Manufacturing of Remdesivir Key Intermediate INT via Novel Purification Technology
The global demand for effective antiviral therapeutics has placed immense pressure on the supply chains of critical pharmaceutical intermediates, specifically those required for the synthesis of nucleotide analog prodrugs. Patent CN112645982A addresses a significant bottleneck in the manufacturing of Remdesivir by disclosing a highly efficient preparation and purification method for its key intermediate, designated as INT. This technical breakthrough moves away from the limitations of earlier synthetic routes which relied heavily on complex resolution techniques or hazardous leaving groups. By integrating a specialized Lewis acid-catalyzed coupling strategy with a novel perfluorinated solvent extraction workup, the disclosed process achieves exceptional control over isomeric impurities and byproduct removal. For industrial partners, this represents a pivotal shift towards a more robust, scalable, and economically viable production model for high-value antiviral ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this critical nucleoside phosphoramidate has been plagued by significant scalability challenges and safety concerns inherent to first and second-generation methodologies. The initial synthetic pathways, as illustrated in the prior art, necessitated a glycosylation reaction followed by cyanation and debenzylation to yield a racemic mixture that required Supercritical Fluid Chromatography (SFC) for chiral resolution. 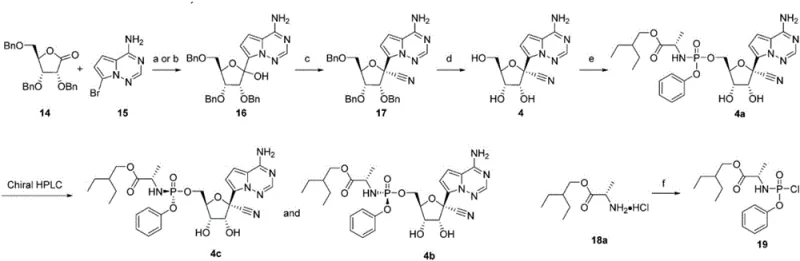 This reliance on SFC is a major impediment for commercial scale-up, as chromatographic separations are inherently low-throughput, solvent-intensive, and difficult to operate continuously in large-scale reactor trains. Furthermore, alternative second-generation approaches attempted to bypass resolution by utilizing p-nitrophenol as a leaving group; however, this introduced severe safety liabilities due to the potential genotoxicity of nitro-aromatic compounds, creating regulatory hurdles for drug substance approval and requiring extensive purge studies.
This reliance on SFC is a major impediment for commercial scale-up, as chromatographic separations are inherently low-throughput, solvent-intensive, and difficult to operate continuously in large-scale reactor trains. Furthermore, alternative second-generation approaches attempted to bypass resolution by utilizing p-nitrophenol as a leaving group; however, this introduced severe safety liabilities due to the potential genotoxicity of nitro-aromatic compounds, creating regulatory hurdles for drug substance approval and requiring extensive purge studies.
The Novel Approach
The methodology presented in CN112645982A offers a transformative solution by optimizing the coupling reaction between the protected nucleoside (Compound 1) and the alanine phosphoramidate derivative (Compound 2). Unlike previous iterations that struggled with the removal of the pentafluorophenol leaving group, this invention leverages the unique physicochemical properties of perfluorinated solvents. 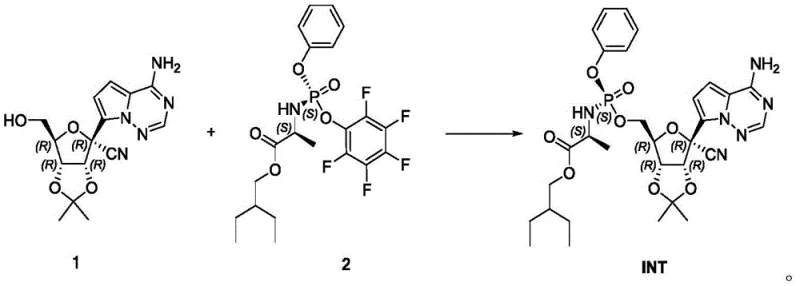 The process facilitates a clean nucleophilic substitution where the pentafluorophenol byproduct is efficiently partitioned into a perfluorinated phase, such as perfluoroheptane, leaving the desired product in the organic layer. This eliminates the need for complex chromatographic purification or hazardous nitro-compounds, resulting in a streamlined workflow that delivers the target intermediate INT with yields exceeding 90% and purity levels approaching 99.6%, effectively resolving the historical trade-off between safety, scalability, and quality.
The process facilitates a clean nucleophilic substitution where the pentafluorophenol byproduct is efficiently partitioned into a perfluorinated phase, such as perfluoroheptane, leaving the desired product in the organic layer. This eliminates the need for complex chromatographic purification or hazardous nitro-compounds, resulting in a streamlined workflow that delivers the target intermediate INT with yields exceeding 90% and purity levels approaching 99.6%, effectively resolving the historical trade-off between safety, scalability, and quality.
Mechanistic Insights into Lewis Acid-Catalyzed Phosphorylation
The core of this synthetic advancement lies in the precise activation of the phosphoramidate coupling partner through Lewis acid catalysis. The reaction mechanism involves the coordination of the Lewis acid, preferably magnesium chloride or aluminum trichloride, to the phosphoryl oxygen of Compound 2. This coordination significantly enhances the electrophilicity of the phosphorus center, thereby facilitating the nucleophilic attack by the primary hydroxyl group of the nucleoside Compound 1. The choice of Lewis acid is critical; experimental data within the patent indicates that magnesium chloride provides an optimal balance, promoting rapid conversion while minimizing the formation of diastereomeric impurities at the phosphorus center. The presence of an organic base, such as triethylamine or N,N-diisopropylethylamine, serves to scavenge the proton released during the bond formation and neutralizes the acidic byproducts, ensuring the reaction proceeds to completion under mild thermal conditions ranging from 0°C to 20°C.
Beyond the coupling event, the purification mechanism represents a sophisticated application of fluorous phase chemistry. The pentafluorophenol leaving group, while sterically bulky enough to favor the desired stereoisomer during the reaction, poses a significant purification challenge in traditional organic solvents due to its tendency to form stable salts or co-crystals with the product. The innovation here utilizes the immiscibility of perfluorinated solvents with standard organic media like ethyl acetate. Upon extraction, the highly fluorinated phenol preferentially dissolves into the perfluorinated phase due to fluorous-fluorous affinity interactions, effectively stripping this impurity from the product stream. This orthogonal separation technique ensures that the final crystallization step starts with a materially cleaner feed, allowing for the growth of high-purity crystals without the entrapment of phenolic residues that typically degrade stability and bioavailability profiles in downstream processing.
How to Synthesize Remdesivir Intermediate INT Efficiently
The operational protocol for this synthesis is designed for reproducibility and safety in a GMP environment. The process begins with the dissolution of the nucleoside precursor in anhydrous tetrahydrofuran, followed by the sequential addition of the phosphoramidate reagent and the Lewis acid catalyst. Temperature control is paramount during the addition of the organic base to manage the exotherm and maintain stereochemical integrity. Following the reaction period, the workup procedure diverges from standard aqueous washes by incorporating the critical perfluorinated solvent extraction step.
- Dissolve the nucleoside precursor (Compound 1) in anhydrous tetrahydrofuran and add the phosphoramidate coupling partner (Compound 2).
- Introduce a Lewis acid catalyst such as magnesium chloride followed by an organic base like triethylamine at controlled low temperatures to initiate coupling.
- Perform a specialized workup involving ethyl acetate dilution and perfluorinated solvent extraction to remove pentafluorophenol byproducts before final crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement strategists and supply chain directors, the adoption of this patented methodology translates into tangible improvements in supply security and cost structure. By eliminating the dependency on Supercritical Fluid Chromatography (SFC), manufacturers can transition from batch-wise, low-volume purification to continuous or large-batch crystallization processes. This shift drastically reduces the capital expenditure required for specialized chromatographic equipment and lowers the operational costs associated with high-pressure CO2 and modifier solvents. Furthermore, the removal of genotoxic p-nitrophenol intermediates simplifies the regulatory filing process, reducing the time and cost associated with toxicological assessments and impurity qualification studies, thereby accelerating time-to-market for generic or novel antiviral formulations.
- Cost Reduction in Manufacturing: The implementation of perfluorinated solvent extraction replaces expensive and labor-intensive chromatographic purification steps. While perfluorinated solvents have an upfront cost, their ability to be recovered and recycled, combined with the significant increase in overall yield (demonstrated up to 90.3% in examples), results in a net reduction in cost per kilogram of the active intermediate. The elimination of SFC resolution also removes a major bottleneck, allowing existing reactor infrastructure to produce significantly higher volumes without additional capital investment in separation units.
- Enhanced Supply Chain Reliability: The robustness of the Lewis acid catalysis system ensures consistent batch-to-batch quality, minimizing the risk of production failures or off-spec material that can disrupt supply timelines. The reagents employed, such as magnesium chloride and triethylamine, are commodity chemicals with stable global supply chains, reducing the risk of raw material shortages. Additionally, the simplified purification train reduces the number of unit operations, decreasing the overall cycle time and enabling faster response to surges in market demand for antiviral therapies.
- Scalability and Environmental Compliance: This process is inherently designed for scale-up, moving away from analytical-scale resolution techniques to preparative crystallization. The use of perfluorinated solvents for extraction offers a closed-loop opportunity for solvent recovery, aligning with green chemistry principles by minimizing waste generation compared to traditional silica gel chromatography. The high purity achieved directly from crystallization reduces the need for re-processing or re-crystallization, further lowering the environmental footprint and energy consumption of the manufacturing facility.
Frequently Asked Questions (FAQ)
The following technical inquiries address common concerns regarding the implementation and validation of this synthesis route. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for technology transfer discussions.
Q: Why is perfluorinated solvent extraction critical in this synthesis?
A: Traditional methods struggle to remove pentafluorophenol byproducts which can form salts with the product. Perfluorinated solvents are immiscible with organic phases and selectively extract the fluorinated phenol, drastically improving purity without chromatography.
Q: What are the advantages over the first-generation SFC resolution method?
A: The first-generation route required Supercritical Fluid Chromatography (SFC) for chiral resolution, which is difficult to scale industrially. This novel method achieves high stereocontrol directly during synthesis, eliminating the need for expensive and low-throughput resolution steps.
Q: Which Lewis acids are compatible with this process?
A: The patent demonstrates successful coupling using magnesium chloride, aluminum trichloride, and tin tetrachloride. Magnesium chloride is particularly preferred for balancing reactivity and minimizing isomeric impurities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Remdesivir Intermediate INT Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep process engineering expertise. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the nuances of Lewis acid handling and perfluorinated solvent management are executed with precision. We maintain stringent purity specifications and operate rigorous QC labs equipped to detect trace isomeric impurities and residual solvents, guaranteeing that every batch of Remdesivir Intermediate INT meets the exacting standards required for global pharmaceutical registration.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our optimized version of this patented technology, we can help you secure a stable supply of high-purity intermediates while minimizing total landed costs. Please contact our technical procurement team to request specific COA data, route feasibility assessments, and samples for your validation protocols.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
