Advanced Synthesis of 1-Substituted Benzylidene-2-Naphthalenone Derivatives for Oncology Applications
Introduction to Novel Antitumor Intermediates
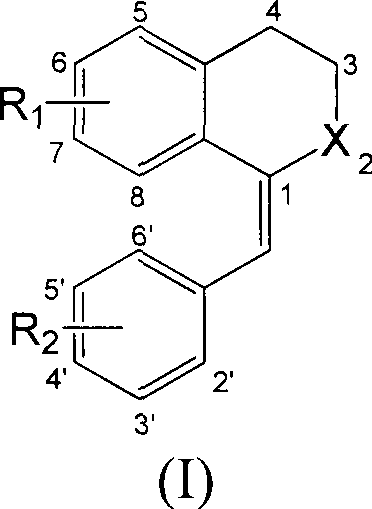
The pharmaceutical landscape is continuously evolving with the discovery of potent small molecule inhibitors targeting critical cellular machinery. Patent CN103130632A introduces a significant advancement in this domain by disclosing a class of 1-substituted benzylidene-2-naphthalenone derivatives with remarkable antitumor properties. These compounds are specifically engineered to inhibit tubulin polymerization, a mechanism crucial for arresting cell division in malignant tissues. For research and development directors seeking robust candidates for oncology pipelines, this technology offers a validated scaffold with demonstrated efficacy against leukemia and solid tumors. The strategic value of this patent lies not only in its biological potency but also in the feasibility of its synthetic route, which utilizes readily available starting materials and standard organic transformations. This report analyzes the technical merits and commercial viability of these derivatives, providing a comprehensive overview for stakeholders involved in the procurement and scale-up of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing tubulin inhibitors often rely on complex natural product extraction or multi-step total syntheses that suffer from low overall yields and poor scalability. Many existing colchicine site binders are derived from natural sources, which introduces variability in supply and challenges in maintaining consistent purity levels required for clinical applications. Furthermore, conventional synthetic routes frequently employ harsh reaction conditions, expensive transition metal catalysts, or protecting group strategies that increase both the environmental footprint and the cost of goods sold. These limitations create significant bottlenecks for procurement managers aiming to secure reliable supply chains for late-stage clinical candidates. The reliance on scarce natural precursors also poses a risk to supply continuity, making it difficult to guarantee the long-term availability needed for commercial drug production. Consequently, there is a pressing industry need for fully synthetic alternatives that offer greater control over impurity profiles and manufacturing costs.
The Novel Approach
The methodology outlined in patent CN103130632A presents a streamlined synthetic strategy that overcomes many of the hurdles associated with traditional tubulin inhibitor production. By utilizing a modular approach based on the naphthalenone scaffold, manufacturers can easily introduce diverse substituents to optimize biological activity without redesigning the entire synthetic pathway. This flexibility allows for rapid structure-activity relationship (SAR) studies, enabling R&D teams to identify lead candidates more efficiently. The process avoids the use of precious metal catalysts, relying instead on common reagents like sodium borohydride and piperidine acetate, which significantly reduces raw material costs and simplifies waste management. For supply chain heads, this translates to a more resilient manufacturing process that is less susceptible to fluctuations in the market prices of exotic reagents. The ability to produce these derivatives through a concise three-step sequence ensures that commercial scale-up can be achieved with minimal technical risk, supporting the timely delivery of critical intermediates for drug development programs.
Mechanistic Insights into Tubulin Polymerization Inhibition
The core innovation of this technology lies in the specific interaction between the 1-substituted benzylidene-2-naphthalenone scaffold and the colchicine binding site on tubulin. Molecular docking simulations referenced in the patent suggest that the planar structure of the naphthalenone ring system facilitates strong hydrophobic interactions within the binding pocket, while the substituents at the R1 and R2 positions provide additional hydrogen bonding opportunities. This precise molecular fit disrupts the dynamic equilibrium of microtubule assembly, preventing the formation of the mitotic spindle necessary for cell division. As a result, tumor cells are arrested in the G2/M phase of the cell cycle, eventually triggering apoptosis. Understanding this mechanism is vital for R&D directors evaluating the potential of these compounds to overcome drug resistance mechanisms often seen with other classes of antimitotic agents. The structural versatility of the scaffold allows for fine-tuning of physicochemical properties, such as solubility and metabolic stability, which are critical parameters for in vivo efficacy.
Impurity control is another critical aspect of the mechanistic profile, particularly given the potent biological activity of these compounds. The synthetic route is designed to minimize the formation of regioisomers and side products that could complicate downstream purification. The use of mild reaction conditions during the aldol condensation step helps to preserve the integrity of sensitive functional groups, reducing the generation of degradation products. For quality assurance teams, this means that achieving the stringent purity specifications required for pharmaceutical intermediates is more attainable with this process. The ability to consistently produce material with a well-defined impurity profile reduces the regulatory burden during the filing of Investigational New Drug (IND) applications. Furthermore, the predictable crystallization behavior of the final naphthol derivatives facilitates efficient isolation and drying, ensuring that the physical form of the material is suitable for subsequent formulation steps.
How to Synthesize 1-Substituted Benzylidene-2-Naphthalenone Efficiently
The synthesis of these high-value intermediates follows a logical progression that balances yield with operational simplicity. The process begins with the preparation of the key naphthalenone core, followed by functionalization to introduce the necessary pharmacophores. This standardized approach allows for seamless technology transfer between laboratory and production scales. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and compliance with Good Manufacturing Practices (GMP).
- Preparation of 3,4-dihydro-2-naphthalenone via refluxing dimethoxynaphthalene with metallic sodium in ethanol followed by acidification.
- Condensation of 3,4-dihydro-2-naphthalenone with substituted benzaldehydes in dichloromethane using piperidine acetate catalysis at room temperature.
- Selective reduction of the ketone intermediate using sodium borohydride in a dichloromethane-methanol solution to yield the final naphthol derivatives.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers substantial benefits for organizations focused on cost optimization and supply chain reliability. The elimination of expensive transition metal catalysts from the process directly contributes to a reduction in raw material expenditures, which is a key metric for procurement managers evaluating vendor proposals. Additionally, the use of common solvents like dichloromethane and ethanol simplifies solvent recovery and recycling operations, further enhancing the economic efficiency of the manufacturing process. For supply chain heads, the robustness of the chemistry means that production schedules can be maintained with greater certainty, reducing the risk of delays that could impact clinical trial timelines. The scalability of the reaction conditions ensures that demand surges can be met without the need for significant capital investment in new equipment. This operational flexibility is a critical advantage in the fast-paced pharmaceutical industry where time-to-market is a decisive factor for commercial success.
- Cost Reduction in Manufacturing: The synthetic pathway leverages inexpensive and widely available starting materials such as dimethoxynaphthalene and substituted benzaldehydes, which are commodity chemicals with stable pricing structures. By avoiding the need for cryogenic conditions or high-pressure reactors, the process reduces energy consumption and utility costs associated with manufacturing. The high atom economy of the condensation and reduction steps minimizes waste generation, leading to lower disposal fees and a reduced environmental compliance burden. These factors collectively drive down the cost of goods sold, allowing for more competitive pricing strategies in the global market. For procurement teams, this translates into significant budget savings that can be reallocated to other critical areas of drug development.
- Enhanced Supply Chain Reliability: The reliance on a fully synthetic route eliminates the geopolitical and agricultural risks associated with natural product sourcing. All reagents used in the process are produced by multiple suppliers globally, ensuring that there are no single points of failure in the supply chain. This diversification of supply sources provides a buffer against market disruptions and ensures continuous availability of critical intermediates. For supply chain planners, this reliability is essential for maintaining inventory levels and meeting production targets. The stability of the intermediates also allows for longer storage times without significant degradation, providing greater flexibility in logistics and distribution planning. This resilience is a key differentiator for partners seeking long-term collaborations in the pharmaceutical sector.
- Scalability and Environmental Compliance: The process is designed with scale-up in mind, utilizing reaction conditions that are easily manageable in large-scale reactors. The absence of hazardous reagents simplifies safety protocols and reduces the risk of workplace accidents, aligning with modern corporate social responsibility goals. Waste streams generated during the synthesis are amenable to standard treatment methods, facilitating compliance with increasingly stringent environmental regulations. This proactive approach to environmental stewardship enhances the reputation of the manufacturing partner and reduces the risk of regulatory penalties. For organizations committed to sustainable practices, this technology offers a pathway to reduce the carbon footprint of their drug manufacturing operations. The combination of scalability and compliance makes this an attractive option for companies aiming to expand their production capacity responsibly.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical specifications and application potential of these derivatives. They are based on the detailed experimental data and claims presented in the patent documentation. Understanding these aspects is crucial for making informed decisions about integrating this technology into your development pipeline. The answers provided reflect the current state of knowledge regarding the efficacy and manufacturability of these compounds.
Q: What is the primary biological mechanism of these naphthalenone derivatives?
A: According to patent CN103130632A, these derivatives function as tubulin inhibitors by binding to the colchicine site, effectively blocking tumor cells in the mitotic metaphase and inhibiting proliferation.
Q: Which cancer cell lines have shown sensitivity to these compounds?
A: Experimental data indicates significant inhibitory activity against human leukemia CEM cells, as well as colon cancer HCT116 and breast cancer MDA-MB-435 cell lines, demonstrating broad-spectrum potential.
Q: How does the substitution pattern affect biological activity?
A: The introduction of methoxy groups at different positions on the naphthalene ring significantly enhances the inhibitory effect on cell proliferation in vitro compared to unsubstituted analogs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Substituted Benzylidene-2-Naphthalenone Derivatives Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis and manufacturing for the global pharmaceutical industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and efficiency. We understand the critical importance of stringent purity specifications and rigorous QC labs in delivering materials that meet the highest regulatory standards. Our commitment to quality is unwavering, and we employ state-of-the-art analytical techniques to verify the identity and purity of every batch produced. By partnering with us, you gain access to a wealth of technical expertise and a robust infrastructure capable of supporting your most challenging projects. We are dedicated to being a strategic ally in your journey from discovery to commercialization.
We invite you to contact our technical procurement team to discuss your specific requirements and explore how we can support your goals. Request a Customized Cost-Saving Analysis to understand the economic benefits of our manufacturing solutions. We are ready to provide specific COA data and route feasibility assessments to demonstrate our capability to deliver high-quality intermediates. Let us help you accelerate your development timeline with our reliable supply and technical excellence. Reach out today to initiate a conversation about your next project.
