Revolutionizing Mercaptoalkenoic Ester Production: A Safe, Scalable Route for Global API Manufacturing
Revolutionizing Mercaptoalkenoic Ester Production: A Safe, Scalable Route for Global API Manufacturing
The landscape of pharmaceutical intermediate synthesis is undergoing a critical transformation driven by the urgent need for safer, more sustainable manufacturing processes. A pivotal advancement in this domain is detailed in patent CN113135848A, which discloses a novel preparation method for mercaptoalkenoic ester compounds. These compounds serve as indispensable building blocks for the synthesis of next-generation therapeutics, particularly those targeting severe conditions such as septicemia. The traditional reliance on hazardous reagents has long been a bottleneck for reliable pharmaceutical intermediate suppliers, but this new technology offers a paradigm shift. By replacing volatile, toxic gases with stable liquid reagents and leveraging modern protecting group chemistry, the invention resolves the industrial problem of high-risk scale-up production. This report provides a deep technical and commercial analysis of this breakthrough, demonstrating how it aligns with the strategic goals of R&D directors seeking purity, procurement managers demanding cost efficiency, and supply chain heads requiring continuity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of mercaptoenol ester compounds has been plagued by significant safety and environmental challenges that hinder industrial adoption. The prevailing synthetic route, as illustrated in the background art, necessitates the preparation of high-concentration hydrogen sulfide methanol solutions followed by the introduction of hydrogen chloride gas into the reaction mixture. 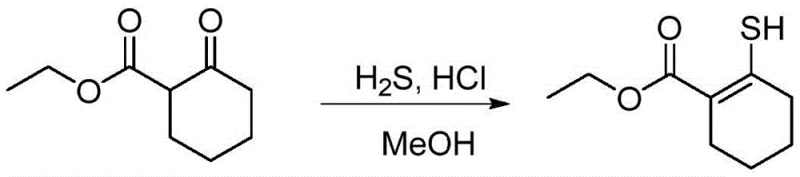 While chemically straightforward on a small laboratory scale, this approach presents catastrophic risks when translated to commercial manufacturing. Hydrogen sulfide is a potent neurotoxin with a notoriously foul odor, creating immediate health hazards for plant personnel and requiring expensive, specialized containment infrastructure. Furthermore, the use of hydrogen chloride gas introduces strong irritation and corrosion issues, complicating equipment maintenance and waste treatment. These factors collectively make safety control extremely difficult, rendering the conventional route unsuitable for the rigorous standards of modern cost reduction in API manufacturing where operator safety and environmental compliance are non-negotiable priorities.
While chemically straightforward on a small laboratory scale, this approach presents catastrophic risks when translated to commercial manufacturing. Hydrogen sulfide is a potent neurotoxin with a notoriously foul odor, creating immediate health hazards for plant personnel and requiring expensive, specialized containment infrastructure. Furthermore, the use of hydrogen chloride gas introduces strong irritation and corrosion issues, complicating equipment maintenance and waste treatment. These factors collectively make safety control extremely difficult, rendering the conventional route unsuitable for the rigorous standards of modern cost reduction in API manufacturing where operator safety and environmental compliance are non-negotiable priorities.
The Novel Approach
In stark contrast to the hazardous legacy methods, the invention disclosed in patent CN113135848A introduces a sophisticated three-step synthetic strategy that completely eliminates the need for toxic gaseous reagents. 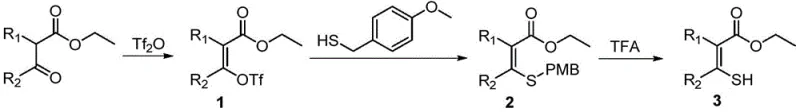 The new pathway begins with the activation of 2-ketoformate starting materials using trifluoromethanesulfonic anhydride (Tf2O) to generate a stable enol triflate intermediate. This is followed by a nucleophilic substitution with a thionation reagent, specifically 4-methoxybenzyl mercaptan, to install a protected sulfur group. Finally, a mild acidic deprotection step yields the target mercaptoalkenoic ester. This modular approach not only drastically simplifies the operational complexity but also enhances the overall safety and environmental profile of the process. By utilizing liquid reagents and standard organic solvents like dichloromethane, the method is inherently more suitable for large-scale production, offering a viable solution for commercial scale-up of complex pharmaceutical intermediates without the burden of gas handling systems.
The new pathway begins with the activation of 2-ketoformate starting materials using trifluoromethanesulfonic anhydride (Tf2O) to generate a stable enol triflate intermediate. This is followed by a nucleophilic substitution with a thionation reagent, specifically 4-methoxybenzyl mercaptan, to install a protected sulfur group. Finally, a mild acidic deprotection step yields the target mercaptoalkenoic ester. This modular approach not only drastically simplifies the operational complexity but also enhances the overall safety and environmental profile of the process. By utilizing liquid reagents and standard organic solvents like dichloromethane, the method is inherently more suitable for large-scale production, offering a viable solution for commercial scale-up of complex pharmaceutical intermediates without the burden of gas handling systems.
Mechanistic Insights into Enol Triflate Activation and Thioether Deprotection
The core innovation of this synthesis lies in the precise control of enolate chemistry and the strategic use of the p-methoxybenzyl (PMB) protecting group. The first step involves the generation of a kinetic enolate using a strong base such as sodium hydride (NaH) at cryogenic temperatures ranging from -60°C to -70°C. Under these conditions, the subsequent addition of Tf2O (1.3 to 1.6 equivalents) ensures the exclusive formation of the vinyl triflate species (Intermediate 1) with high regioselectivity. This activation transforms a relatively unreactive ketone carbonyl into a highly electrophilic center, primed for nucleophilic attack. The use of triflate as a leaving group is superior to traditional halides in this context, facilitating smoother downstream transformations and minimizing side reactions that could compromise the purity of the final API intermediate.
Following the formation of the enol triflate, the process employs a substitution reaction with 4-methoxybenzyl mercaptan to introduce the sulfur functionality. This step effectively masks the reactive thiol group as a stable thioether (Intermediate 2), preventing unwanted oxidation or polymerization during the synthesis. The final deprotection is achieved using trifluoroacetic acid (TFA) at moderate temperatures (40-50°C), which cleanly cleaves the PMB group to reveal the free mercapto group. This mechanistic sequence ensures excellent impurity control; by avoiding the harsh conditions of H2S/HCl, the formation of polysulfides or chlorinated byproducts is suppressed. The result is a high-purity product stream that requires less intensive purification, directly supporting the stringent quality specifications demanded by global regulatory bodies for septicemia drug precursors.
How to Synthesize Ethyl 2-mercaptocyclohexenecarboxylate Efficiently
The practical implementation of this chemistry is demonstrated through specific embodiments that highlight its operational simplicity and robustness. The patent details a standardized protocol starting with the conversion of ethyl 2-oxocyclohexanecarboxylate into its corresponding triflate derivative, followed by thiolation and deprotection. This sequence has been validated across multiple ring sizes, proving its versatility for generating diverse analogues. For R&D teams looking to adopt this technology, the process offers a clear, reproducible workflow that minimizes the need for specialized equipment. The detailed standardized synthesis steps see the guide below, which outlines the precise stoichiometry, temperature controls, and workup procedures required to achieve the reported high yields.
- Generate the enol triflate intermediate by reacting 2-ketoformate with Tf2O and base (NaH) at cryogenic temperatures (-60 to -70°C).
- Perform nucleophilic substitution using 4-methoxybenzyl mercaptan to install the protected thiol group, yielding the PMB-thioether intermediate.
- Execute acidic deprotection using trifluoroacetic acid (TFA) at 40-50°C to remove the PMB group and isolate the final mercaptoalkenoic ester.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the transition to this novel synthetic route represents a significant opportunity to optimize operational expenditures and mitigate supply risks. The elimination of hydrogen sulfide gas removes the necessity for costly gas scrubbing towers, negative pressure reactors, and extensive personal protective equipment (PPE) protocols associated with toxic gas handling. This fundamental change in the process safety profile translates directly into reduced capital expenditure (CAPEX) for facility upgrades and lower operating expenses (OPEX) related to safety compliance and waste disposal. Furthermore, the use of commercially available, stable liquid reagents like Tf2O and 4-methoxybenzyl mercaptan ensures a more reliable supply chain compared to the logistical challenges of sourcing and storing high-pressure toxic gases.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the simplification of the reaction workflow and the improvement in overall yield efficiency. By avoiding the losses associated with gas absorption and the formation of intractable tars common in H2S reactions, the process achieves isolated yields consistently above 85% across different substrates. The removal of transition metal catalysts or exotic reagents further lowers the raw material costs. Additionally, the simplified workup procedures, which involve standard aqueous washes and distillation rather than complex gas neutralization steps, reduce labor hours and utility consumption, leading to substantial cost savings in the final production of mercaptoalkenoic esters.
- Enhanced Supply Chain Reliability: Supply chain continuity is significantly bolstered by the robustness of the new reagents. Unlike hydrogen sulfide, which requires specialized transport and storage cylinders, the liquid reagents used in this invention (Tf2O, PMB-SH, TFA) are standard commodities in the fine chemical industry with multiple global suppliers. This diversification of the supply base reduces the risk of production stoppages due to raw material shortages. Moreover, the stability of the intermediates allows for potential telescoping of steps or storage of semi-finished goods, providing greater flexibility in production scheduling and inventory management to meet fluctuating market demands for sepsis treatment intermediates.
- Scalability and Environmental Compliance: From an environmental perspective, this method aligns perfectly with green chemistry principles by eliminating the emission of foul-smelling and toxic sulfur gases. The waste streams generated are primarily organic solvents and aqueous salts, which are far easier to treat and dispose of than sulfide-contaminated waste. This ease of waste management facilitates faster regulatory approvals for new manufacturing sites and reduces the environmental liability of the production facility. The process has been proven scalable from gram to multi-kilogram levels in the patent examples, demonstrating that the reaction kinetics and heat transfer profiles remain manageable even as batch sizes increase, ensuring a smooth path to commercial tonnage production.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating this technology, we have compiled answers to common inquiries regarding the safety, efficiency, and applicability of this synthetic route. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, providing a factual basis for assessing the feasibility of integrating this method into your existing manufacturing portfolio. Understanding these nuances is critical for projecting accurate timelines and resource allocation for process development.
Q: Why is the traditional H2S method considered unsafe for large-scale production?
A: The conventional route relies on high-concentration hydrogen sulfide methanol solutions and hydrogen chloride gas. H2S is extremely toxic with a foul odor, posing severe safety hazards and requiring complex gas handling infrastructure, making scale-up risky and environmentally difficult.
Q: What are the key yield advantages of the new triflate-based pathway?
A: The novel three-step process demonstrates robust efficiency, with experimental data showing isolated yields ranging from 85% to 93.4% across different ring sizes (cyclopentane, cyclohexane, cycloheptane), ensuring high material throughput without the losses associated with gas scrubbing.
Q: Can this process be adapted for different cyclic ketone substrates?
A: Yes, the methodology is highly versatile. The patent explicitly validates the route on 5-membered (cyclopentane), 6-membered (cyclohexane), and 7-membered (cycloheptane) rings, indicating broad substrate tolerance for synthesizing various mercaptoalkenoic ester derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Mercaptoalkenoic Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of life-saving drugs depends on the reliability and quality of the underlying chemical supply chain. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent supply regardless of market volatility. Our state-of-the-art facilities are equipped with the necessary containment and safety systems to handle sensitive chemistries like the triflate activation described in CN113135848A, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the exacting standards required for pharmaceutical applications. We are committed to being a long-term strategic partner in your drug development journey.
We invite you to leverage our technical expertise to accelerate your project timelines. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how adopting this safer synthetic route can improve your bottom line. Please contact our technical procurement team today to request specific COA data for our mercaptoalkenoic ester portfolio and to discuss route feasibility assessments for your upcoming projects. Let us help you secure a safer, more efficient supply of these critical intermediates.
