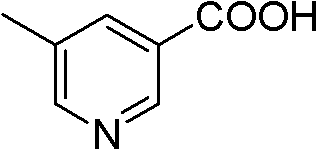
Advanced Oxidation Technology for High-Purity 5-Methylnicotinic Acid Production
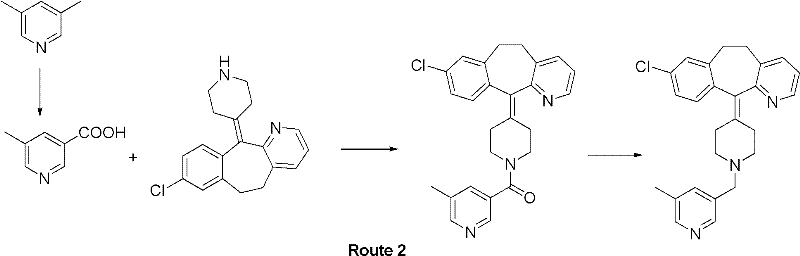
The pharmaceutical industry continuously seeks robust synthetic routes for critical intermediates, and patent CN102558043A presents a significant advancement in the production of 5-methylnicotinic acid. This compound serves as a pivotal building block in the synthesis of Rupatadine, a potent antihistamine used globally for treating allergic rhinitis. The disclosed technology leverages a phase transfer catalysis (PTC) strategy to optimize the oxidation of 3,5-lutidine using potassium permanganate. By shifting away from traditional, resource-intensive methods, this innovation addresses key pain points in fine chemical manufacturing, specifically focusing on yield enhancement and solvent reduction. The implementation of quaternary ammonium salts as catalysts allows the reaction to proceed smoothly under mild thermal conditions, offering a compelling value proposition for manufacturers aiming to streamline their supply chains for antiallergic medications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 5-methylnicotinic acid via potassium permanganate oxidation has been plagued by significant inefficiencies that hinder commercial scalability. Traditional protocols, such as those cited in earlier literature like J. Org. Chem. 1988, often suffer from extremely low conversion rates, with yields frequently stagnating around 20% to 50% even after optimization attempts. A major bottleneck in these legacy processes is the inhomogeneous nature of the reaction system, where the organic substrate and the aqueous oxidant have poor interfacial contact. To compensate for the limited solubility of potassium permanganate and to manage heat dissipation, conventional methods require vast quantities of water as a solvent. This excessive dilution not only lowers the volumetric productivity of the reactor but also imposes a heavy burden on downstream processing, requiring substantial energy inputs for heating, stirring, and eventual wastewater treatment.
The Novel Approach
In stark contrast, the methodology outlined in CN102558043A introduces a transformative improvement by incorporating specific phase transfer catalysts into the reaction matrix. By utilizing agents such as tetrabutylammonium bromide or benzyltriethylammonium chloride, the process effectively bridges the phase gap between the lipophilic 3,5-lutidine and the hydrophilic oxidant. This catalytic enhancement enables the reaction to achieve yields exceeding 65%, with specific embodiments demonstrating efficiencies as high as 88%. Furthermore, the new approach allows for a dramatic reduction in water usage, effectively doubling the single-batch output capacity compared to traditional techniques. The ability to operate at moderate temperatures ranging from 20°C to 75°C further simplifies the thermal management requirements, making the process safer and more energy-efficient for large-scale operations.
Mechanistic Insights into Phase Transfer Catalyzed Oxidation
The core mechanism driving this enhanced efficiency lies in the interfacial activity of the quaternary ammonium salts employed as phase transfer catalysts. In the absence of these catalysts, the oxidation of the methyl group on the pyridine ring is kinetically limited by the mass transfer resistance between the organic and aqueous phases. The catalyst molecules possess both lipophilic alkyl chains and hydrophilic cationic heads, allowing them to shuttle permanganate ions into the organic phase or bring the organic substrate to the interface where oxidation can occur rapidly. This facilitation ensures that the oxidant is utilized much more effectively, minimizing the formation of incomplete oxidation byproducts and preventing the degradation of the pyridine ring which can occur under harsher, uncontrolled conditions. The result is a cleaner reaction profile that simplifies purification and maximizes the recovery of the desired carboxylic acid functionality.
Impurity control is another critical aspect where this mechanistic understanding translates to practical benefits. The patent specifies that adding potassium permanganate in batches is crucial to prevent local excesses of the oxidant, which could lead to over-oxidation or the formation of dicarboxylic acid impurities. The presence of the phase transfer catalyst ensures that each batch of oxidant is consumed efficiently before the next addition, maintaining a steady state of reaction progress. Additionally, the mild temperature window of 20°C to 40°C identified as optimal helps suppress thermal degradation pathways. By carefully managing the stoichiometry and addition rate, manufacturers can achieve a highly consistent impurity profile, which is essential for meeting the stringent quality standards required for pharmaceutical intermediates destined for human consumption.
How to Synthesize 5-Methylnicotinic Acid Efficiently
The synthesis protocol described in the patent offers a straightforward pathway that balances high yield with operational simplicity, making it ideal for adoption in standard chemical processing facilities. The process begins with the preparation of an aqueous reaction mixture containing the substrate and the catalyst, followed by the controlled addition of the oxidant. Detailed standard operating procedures regarding specific molar ratios, temperature ramping, and workup pH adjustments are critical for replicating the high yields reported in the patent examples. For a comprehensive guide on executing this synthesis with precision, please refer to the standardized steps provided below.
- Prepare the reaction mixture by dissolving 3,5-lutidine and a quaternary ammonium phase transfer catalyst in water, then heat to the specified range.
- Gradually add potassium permanganate in batches to control exothermicity and minimize over-oxidation byproducts.
- Filter the cooled mixture, adjust the pH of the filtrate to precipitate the product, and collect the purified 5-methylnicotinic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this PTC-enhanced oxidation method presents a multitude of strategic benefits that extend beyond mere chemical yield. The primary advantage lies in the substantial reduction of raw material waste and solvent consumption, which directly correlates to lower variable costs per kilogram of produced intermediate. By eliminating the need for massive volumes of water, facilities can significantly reduce their energy footprint associated with heating and cooling large reactor masses, as well as lower the costs related to wastewater disposal and environmental compliance. This efficiency gain allows for a more competitive pricing structure for the final API intermediate, providing a buffer against market volatility in raw material costs.
- Cost Reduction in Manufacturing: The elimination of excessive solvent usage and the improvement in reaction yield fundamentally alter the cost structure of production. By achieving higher conversion rates with less input material, the effective cost of goods sold is driven down without compromising on quality. Furthermore, the simplified workup procedure, which relies on basic pH adjustment and filtration rather than complex extraction or chromatography, reduces labor hours and equipment occupancy time. These operational savings accumulate to provide a significant margin improvement, making the sourcing of this intermediate more economically viable for long-term contracts.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures a more stable and predictable supply of 5-methylnicotinic acid. Because the reaction is less sensitive to minor fluctuations in temperature and mixing efficiency compared to the uncatalyzed version, the risk of batch failures is minimized. This reliability is crucial for maintaining continuous production schedules for downstream API manufacturing, preventing costly delays in the formulation of antiallergic medications. Suppliers utilizing this technology can offer more consistent lead times, fostering stronger partnerships with pharmaceutical clients who prioritize supply security.
- Scalability and Environmental Compliance: From a scale-up perspective, the reduced solvent load means that existing reactor infrastructure can produce significantly more product per batch, effectively increasing capacity without capital expenditure on new vessels. This intensification of the process aligns perfectly with modern green chemistry initiatives, as it generates less aqueous waste and consumes less energy. For supply chain heads focused on sustainability metrics, adopting this method demonstrates a commitment to environmentally responsible manufacturing, which is increasingly becoming a prerequisite for vendor qualification in the global pharmaceutical market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 5-methylnicotinic acid using this advanced oxidation technology. These insights are derived directly from the experimental data and process descriptions found in the underlying patent documentation, ensuring that the information provided is grounded in verified chemical science. Understanding these details helps stakeholders make informed decisions regarding process integration and quality assurance protocols.
Q: What is the primary advantage of using a phase transfer catalyst in this oxidation?
A: The addition of a phase transfer catalyst significantly accelerates the reaction rate and improves yield by facilitating the interaction between the organic substrate and the aqueous oxidant, overcoming the limitations of inhomogeneous reaction systems.
Q: How does this method impact water consumption compared to traditional protocols?
A: This novel approach drastically reduces the volume of water required as a solvent compared to conventional methods, which typically necessitate large excesses of water to dissolve the oxidant, thereby improving single-pot production capacity.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the method operates under mild temperatures and utilizes simple workup procedures involving pH adjustment and filtration, making it highly scalable and cost-effective for industrial API intermediate production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Methylnicotinic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of life-saving medications like Rupatadine. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We adhere to stringent purity specifications and utilize rigorous QC labs to verify that every batch of 5-methylnicotinic acid meets the exacting standards necessary for pharmaceutical synthesis. Our commitment to process excellence means that we can deliver materials that facilitate smooth downstream processing for our clients.
We invite you to collaborate with us to optimize your supply chain for antiallergic drug production. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs, demonstrating how our efficient manufacturing processes can reduce your overall procurement costs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced capabilities can support your project timelines and quality goals.
