Advanced Synthetic Route for Bazedoxifene Intermediates Enhancing Commercial Scalability
Advanced Synthetic Route for Bazedoxifene Intermediates Enhancing Commercial Scalability
The pharmaceutical industry constantly seeks robust manufacturing pathways for Selective Estrogen Receptor Modulators (SERMs) to treat postmenopausal osteoporosis effectively. Patent CN102690225A discloses a groundbreaking synthetic methodology for Bazedoxifene, also known as WAY 140424, which addresses critical bottlenecks found in prior art. This innovative approach utilizes azepane as a primary starting material, undergoing a sequence of bromination, substitution, reductive amination, and cyclization to construct the complex molecular architecture. By shifting away from hazardous reagents and low-yielding steps, this technology offers a compelling value proposition for reliable pharmaceutical intermediate suppliers aiming to optimize their production portfolios. The structural complexity of the target molecule requires precise control over regioselectivity and functional group tolerance, which this new route manages with exceptional efficiency.
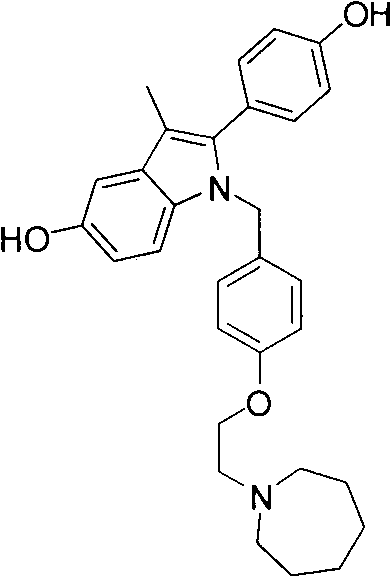
Bazedoxifene represents a third-generation SERM with a distinct chemical profile characterized by a 1-(4-(2-(azepan-1-yl)ethoxy)phenyl)methyl group attached to a 2-(4-hydroxyphenyl)-3-methyl-1H-indole-5-ol core. The strategic placement of the azepane ring via an ethoxy linker is crucial for its biological activity and receptor binding affinity. Traditional syntheses often struggle with the introduction of this bulky amine moiety late in the sequence, leading to purification nightmares and yield losses. The disclosed method front-loads the construction of the side chain, creating a stable aldehyde intermediate that serves as a versatile pivot point for subsequent coupling reactions. This strategic disconnection not only simplifies the retrosynthetic analysis but also aligns perfectly with the principles of green chemistry by minimizing waste generation and energy consumption throughout the manufacturing lifecycle.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
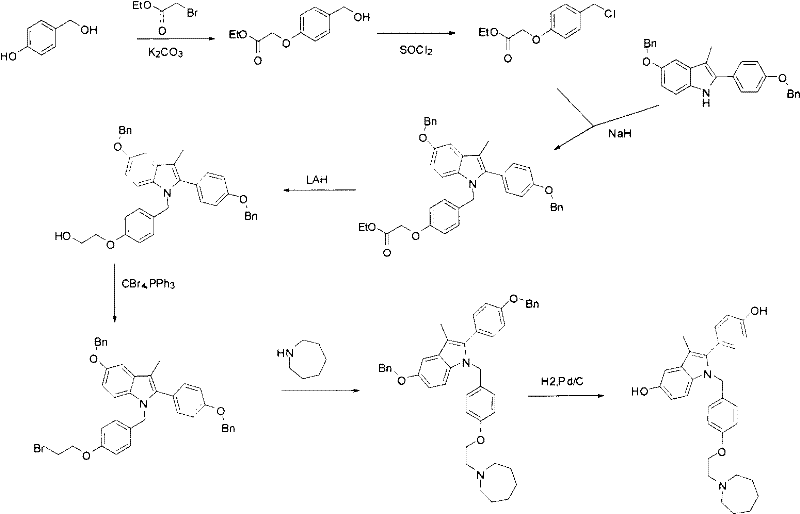
Historical precedents for synthesizing Bazedoxifene intermediates, such as those described in EP 0802183 and US 5998402, rely heavily on aggressive alkylation strategies that pose significant operational risks. These conventional routes typically employ sodium hydride (NaH) for N-alkylation of the indole nitrogen, a reagent known for its pyrophoric nature and difficulty in safe handling on a large industrial scale. Furthermore, the reduction steps often utilize lithium aluminum hydride (LAH), which presents severe safety hazards due to its violent reaction with moisture and the generation of flammable hydrogen gas. Beyond safety concerns, the chemical efficiency of these legacy processes is suboptimal, with the critical indole cyclization step frequently delivering yields in the range of only 50% to 60%. Such low conversion rates necessitate larger reactor volumes and increased solvent usage to achieve the same output, directly inflating the cost of goods sold (COGS) and complicating waste management protocols.
The Novel Approach
In stark contrast, the methodology outlined in patent CN102690225A introduces a paradigm shift by leveraging reductive amination and mild etherification techniques. Instead of forcing the azepane ring onto a pre-formed indole under harsh basic conditions, the new route constructs the side chain first. It begins with the reaction of azepane with 1,2-dibromoethane to generate a reactive bromo-ethyl intermediate, which is then coupled with p-hydroxybenzaldehyde. This sequence avoids the use of dangerous hydride reagents entirely, substituting them with sodium triacetoxyborohydride for the reductive amination step, which operates safely at near-ambient temperatures. The result is a streamlined process that not only enhances operator safety but also significantly improves the overall mass balance of the synthesis, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Reductive Amination and Cyclization
The core innovation of this synthetic strategy lies in the mechanistic elegance of the reductive amination step, which couples the aldehyde intermediate with 4-(benzyloxy)aniline. Mechanistically, this reaction proceeds through the formation of an imine or iminium ion intermediate, which is subsequently reduced in situ by sodium triacetoxyborohydride. This reagent is highly chemoselective, reducing the imine functionality without affecting other sensitive groups such as the benzyl ether protections or the aromatic rings. The reaction conditions are remarkably mild, typically initiated at 0-5°C and warmed to 35°C, which minimizes the formation of thermal degradation byproducts and oligomers. This level of control is paramount for maintaining a clean impurity profile, ensuring that the downstream purification burden is drastically reduced compared to the messy mixtures generated by non-selective reducing agents like LAH.
Following the amine coupling, the construction of the indole core is achieved through a cyclization reaction with 1-(4-(benzyloxy)phenyl)-2-bromoethanone. This step effectively closes the five-membered pyrrole ring onto the aniline nitrogen, driven by the nucleophilic attack of the amine on the alpha-bromo ketone. The use of triethylamine as a base in dimethylformamide (DMF) at elevated temperatures (120°C) facilitates this intramolecular condensation efficiently. Crucially, because the side chain is already fully assembled and stable, the cyclization does not risk disrupting the azepane moiety. The final deprotection via catalytic hydrogenation (H2, Pd/C) cleanly removes the benzyl groups to reveal the phenolic hydroxyls, completing the synthesis of the target molecule with high fidelity and minimal structural rearrangement.
How to Synthesize Bazedoxifene Efficiently
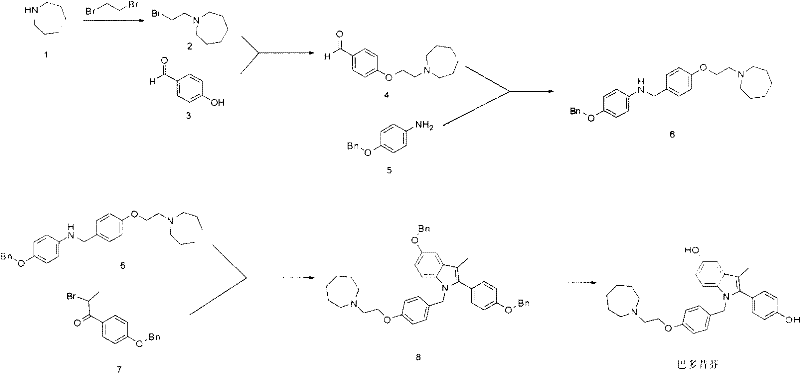
The execution of this synthetic pathway requires careful attention to stoichiometry and temperature control, particularly during the initial bromination and the final cyclization steps. The process is designed to be telescoped where possible, reducing the number of isolation events and solvent swaps. For R&D teams looking to implement this technology, the protocol emphasizes the use of common organic solvents like acetonitrile, dichloromethane, and ethanol, which are easily recovered and recycled in a GMP environment. The detailed standard operating procedures for each transformation, including specific workup instructions and purification parameters, are essential for replicating the high yields reported in the patent literature. Below is the structured guide for the key transformations involved in this efficient manufacturing process.
- React azepane with 1,2-dibromoethane to form 1-(2-bromoethyl)azepane, followed by etherification with p-hydroxybenzaldehyde.
- Perform reductive amination between the resulting aldehyde intermediate and 4-(benzyloxy)aniline using sodium triacetoxyborohydride.
- Execute cyclization with 1-(4-(benzyloxy)phenyl)-2-bromoethanone to form the protected indole core, followed by catalytic hydrogenation for deprotection.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route translates into tangible strategic benefits that extend beyond simple unit price negotiations. By eliminating the dependency on hazardous, specialty reagents like sodium hydride and lithium aluminum hydride, the supply chain becomes more resilient and less prone to disruptions caused by strict transportation regulations for dangerous goods. The use of commodity chemicals such as azepane, dibromoethane, and p-hydroxybenzaldehyde ensures a stable and competitive raw material market, shielding the project from volatile pricing spikes associated with exotic catalysts. Furthermore, the improved yield profile across the early stages of the synthesis means that less raw material is required to produce the same amount of finished intermediate, driving down the effective material cost per kilogram significantly.
- Cost Reduction in Manufacturing: The elimination of expensive and dangerous reagents directly lowers the variable cost of production. Traditional routes require specialized equipment and safety protocols to handle pyrophoric materials, which adds substantial overhead to the manufacturing budget. By switching to mild reductive amination conditions, facilities can utilize standard glass-lined or stainless steel reactors without the need for inert atmosphere gloveboxes or specialized quenching systems. Additionally, the higher yields in the initial steps (reported up to 93% for etherification) mean that the throughput of the plant is effectively increased without capital expenditure on new hardware, resulting in substantial cost savings over the product lifecycle.
- Enhanced Supply Chain Reliability: Sourcing raw materials for the conventional route can be challenging due to the regulatory scrutiny on hydride reagents. The new method relies on bulk chemicals that are widely produced globally, ensuring a diversified supplier base and reducing the risk of single-source bottlenecks. The robustness of the reaction conditions also implies fewer batch failures due to minor deviations in temperature or moisture content, leading to more predictable delivery schedules. This reliability is critical for maintaining continuous API production lines and meeting the rigorous just-in-time delivery expectations of major pharmaceutical clients.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is vastly superior. The avoidance of LAH eliminates the generation of aluminum waste sludge, which is difficult and costly to dispose of in compliance with environmental regulations. The milder reaction conditions reduce energy consumption for heating and cooling, contributing to a lower carbon footprint for the manufacturing process. The simplified purification steps, facilitated by cleaner reaction profiles, reduce solvent consumption and waste volume, aligning with modern green chemistry initiatives and making the process easier to scale from pilot plant to multi-ton commercial production.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthetic pathway is vital for stakeholders evaluating its implementation. The following questions address common concerns regarding safety, yield, and scalability, drawing directly from the experimental data and beneficial effects described in the patent documentation. These insights are intended to clarify the operational advantages and assist in the decision-making process for technology transfer and process validation.
Q: How does the new synthetic route improve safety compared to conventional methods?
A: The novel route eliminates the use of hazardous reagents such as sodium hydride (NaH) and lithium aluminum hydride (LAH), replacing them with milder conditions like sodium triacetoxyborohydride for reductive amination, significantly reducing explosion risks and handling difficulties.
Q: What are the yield improvements in the key intermediate steps?
A: The new method achieves high yields in early stages, such as 89.1% for the azepane bromination and 93.0% for the etherification step, contrasting sharply with the 50%-60% yields often seen in traditional indole synthesis routes.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process utilizes readily available raw materials like azepane and p-hydroxybenzaldehyde, operates under mild temperatures (0-120°C), and avoids cryogenic or pyrophoric conditions, making it highly amenable to multi-ton scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bazedoxifene Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this optimized synthetic route for the global supply of osteoporosis therapeutics. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific requirements of this chemistry, including the precise temperature control needed for the reductive amination and the robust filtration systems required for the final catalytic hydrogenation. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Bazedoxifene intermediate meets the highest international standards for identity, potency, and impurity control.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this advanced technology for their supply chains. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise in process chemistry can drive value and security for your critical drug development programs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
