Advanced C-H Functionalization for Scalable Quinoline Derivative Production
The pharmaceutical industry continuously seeks robust methodologies for constructing complex heterocyclic scaffolds, particularly quinoline derivatives which serve as critical backbones in numerous bioactive molecules. Patent CN109776507B introduces a groundbreaking preparation method for 2-methyl-4-(tetrahydrofuran-2-yl) quinoline derivatives, leveraging a sophisticated Selectfluor and silver nitrate catalytic system. This innovation represents a significant leap forward in C-H bond functionalization, moving away from harsh traditional conditions toward a more sustainable and efficient aqueous-phase protocol. By utilizing tetrahydrofuran not merely as a solvent but as a direct coupling partner, the process achieves remarkable regioselectivity at the C4 position of the quinoline ring. For R&D directors and process chemists, this patent offers a compelling solution to the longstanding challenge of introducing tetrahydrofuran moieties onto electron-deficient nitrogen heterocycles without compromising yield or purity. The methodology underscores a shift towards greener chemistry, utilizing water as a co-solvent to enhance solubility and reaction control while minimizing environmental impact.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the functionalization of N-heteroaromatics like quinolines has relied heavily on methods involving alkyl carboxylic acids combined with one-electron oxidants to generate carbon free radicals. While effective in specific contexts, these prior art techniques suffer from significant limitations regarding substrate universality and operational complexity. The requirement for specific carboxylic acid precursors often restricts the scope of accessible derivatives, forcing synthetic chemists to design lengthy multi-step routes to install simple alkyl or cyclic ether groups. Furthermore, the reaction conditions associated with these older methods can be quite harsh, often necessitating high temperatures or strong acidic environments that degrade sensitive functional groups on the quinoline core. The reliance on organic solvents exclusively also raises concerns regarding waste generation and process safety, particularly when scaling up for commercial manufacturing. Consequently, the industry has faced a persistent bottleneck in efficiently accessing diverse libraries of 4-substituted quinolines, driving up costs and extending development timelines for new drug candidates.
The Novel Approach
In stark contrast, the novel approach detailed in CN109776507B utilizes a direct dehydrogenative coupling between 2-methylquinoline derivatives and tetrahydrofuran under mild catalytic conditions. This method employs a dual system of Selectfluor as a potent fluorinating agent and oxidant alongside a catalytic amount of silver nitrate to facilitate the radical generation and subsequent coupling. 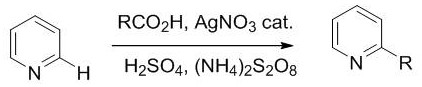 The reaction proceeds smoothly in a mixed solvent system of water and tetrahydrofuran, typically at a ratio of 1:3, which significantly improves the solubility of the polar intermediates and stabilizes the radical species. Operating at a moderate temperature of 50°C, this protocol avoids the thermal degradation issues common in high-temperature syntheses, ensuring that delicate substituents such as halogens or alkoxy groups remain intact. The result is a highly controllable process that delivers excellent yields, as demonstrated by the 90% yield achieved with 2-methyl-6-fluoroquinoline, showcasing the method's robustness across various electronic environments.
The reaction proceeds smoothly in a mixed solvent system of water and tetrahydrofuran, typically at a ratio of 1:3, which significantly improves the solubility of the polar intermediates and stabilizes the radical species. Operating at a moderate temperature of 50°C, this protocol avoids the thermal degradation issues common in high-temperature syntheses, ensuring that delicate substituents such as halogens or alkoxy groups remain intact. The result is a highly controllable process that delivers excellent yields, as demonstrated by the 90% yield achieved with 2-methyl-6-fluoroquinoline, showcasing the method's robustness across various electronic environments.
Mechanistic Insights into Selectfluor/AgNO3 Catalyzed C-H Activation
The core of this technological breakthrough lies in the intricate interplay between the silver catalyst and the Selectfluor oxidant to generate reactive radical species selectively. Mechanistically, the silver ion likely coordinates with the nitrogen atom of the quinoline or interacts with the oxidant to lower the activation energy for hydrogen abstraction. Selectfluor serves as a single-electron oxidant that facilitates the formation of an alpha-oxy radical from the tetrahydrofuran solvent, which is the key nucleophilic species in this transformation. This radical then attacks the electron-deficient C4 position of the protonated or coordinated 2-methylquinoline, driven by the inherent electronic bias of the heterocyclic ring. The presence of water in the solvent mixture plays a crucial role in stabilizing the transition states and potentially assisting in the proton transfer steps required to restore aromaticity after the coupling event. Understanding this mechanism allows process chemists to fine-tune reaction parameters, such as the molar ratio of Selectfluor to substrate (optimized at 1:4), to maximize efficiency and minimize the formation of over-oxidized byproducts.
From an impurity control perspective, the specificity of this radical coupling mechanism offers distinct advantages over non-catalytic thermal methods. The mild conditions prevent the polymerization of tetrahydrofuran or the decomposition of the quinoline scaffold, which are common side reactions in more aggressive protocols. 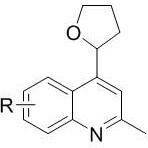 The patent data indicates that varying the R group on the quinoline ring (such as fluoro, chloro, bromo, methyl, or methoxy) has a minimal detrimental effect on the reaction outcome, suggesting a broad tolerance for electronic variations. This consistency is vital for maintaining a clean impurity profile, as it reduces the likelihood of generating hard-to-remove structural analogs that could complicate downstream purification. For quality control teams, this means that the crude product typically requires less rigorous purification steps, often achievable through standard silica gel column chromatography using ethyl acetate and n-hexane. The ability to predictably manage the impurity profile translates directly into higher overall process reliability and reduced batch-to-batch variability in a commercial setting.
The patent data indicates that varying the R group on the quinoline ring (such as fluoro, chloro, bromo, methyl, or methoxy) has a minimal detrimental effect on the reaction outcome, suggesting a broad tolerance for electronic variations. This consistency is vital for maintaining a clean impurity profile, as it reduces the likelihood of generating hard-to-remove structural analogs that could complicate downstream purification. For quality control teams, this means that the crude product typically requires less rigorous purification steps, often achievable through standard silica gel column chromatography using ethyl acetate and n-hexane. The ability to predictably manage the impurity profile translates directly into higher overall process reliability and reduced batch-to-batch variability in a commercial setting.
How to Synthesize 2-Methyl-4-(tetrahydrofuran-2-yl) Quinoline Efficiently
To implement this synthesis effectively, operators must adhere to precise stoichiometric ratios and temperature controls to replicate the high yields reported in the patent examples. The process begins by dissolving the 2-methylquinoline derivative in a pre-mixed solution of tetrahydrofuran and water, followed by the sequential addition of Selectfluor and the silver nitrate catalyst. Maintaining the reaction temperature strictly at 50°C is critical, as comparative data shows that deviations to 30°C or 70°C result in significantly diminished yields due to incomplete conversion or thermal instability. After the designated reaction time of approximately 3 hours, the mixture is carefully neutralized with saturated sodium bicarbonate to quench any remaining acidic species before extraction. Detailed standardized synthesis steps follow below to ensure reproducibility and safety during scale-up operations.
- Combine 2-methylquinoline derivative, Selectfluor, and AgNO3 catalyst in a THF/water mixture (3: 1 ratio).
- Heat the reaction mixture to 50°C and maintain stirring for 3 hours to ensure complete conversion.
- Neutralize with saturated sodium bicarbonate, extract with ethyl acetate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented methodology offers substantial strategic benefits beyond mere technical feasibility. The shift towards an aqueous-based reaction system significantly reduces the dependency on large volumes of hazardous organic solvents, thereby lowering waste disposal costs and enhancing workplace safety standards. By eliminating the need for exotic or difficult-to-source alkyl carboxylic acid precursors, the supply chain becomes more resilient, relying instead on commodity chemicals like tetrahydrofuran and Selectfluor which are readily available in bulk quantities. This simplification of the raw material portfolio mitigates the risk of supply disruptions and allows for more accurate forecasting of production costs. Furthermore, the high atom economy and selectivity of the reaction mean that less starting material is wasted, directly contributing to a more sustainable and cost-effective manufacturing model that aligns with modern green chemistry initiatives.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts, other than the catalytic amount of silver which can potentially be recovered, combined with the use of water as a co-solvent, drastically reduces the raw material expenditure per kilogram of product. The simplified workup procedure, which avoids complex distillation or crystallization steps often required to remove high-boiling carboxylic acids, further lowers utility and labor costs associated with production. Additionally, the high yields observed across a range of substrates mean that the effective cost of goods sold is minimized, as less feedstock is required to produce the same amount of active pharmaceutical ingredient intermediate. These cumulative efficiencies create a compelling economic case for switching to this newer technology, offering significant margin improvements for high-volume contracts.
- Enhanced Supply Chain Reliability: Utilizing widely available reagents such as 2-methylquinoline derivatives and tetrahydrofuran ensures that the supply chain is not vulnerable to the bottlenecks often associated with specialized custom synthons. The robustness of the reaction conditions allows for flexible scheduling and rapid turnaround times, as the process is less sensitive to minor fluctuations in environmental conditions compared to moisture-sensitive organometallic reactions. This reliability is crucial for maintaining continuous production lines and meeting tight delivery deadlines for downstream API manufacturers. By securing a synthesis route that is both chemically stable and logistically straightforward, companies can build a more agile supply network capable of responding quickly to market demands without compromising on quality or compliance.
- Scalability and Environmental Compliance: The use of water as a major component of the reaction solvent system inherently reduces the fire hazard and volatile organic compound (VOC) emissions associated with traditional organic synthesis. This aligns perfectly with increasingly stringent environmental regulations, making it easier to obtain necessary permits for large-scale production facilities. The mild temperature profile (50°C) reduces the energy load required for heating and cooling, contributing to a lower carbon footprint for the manufacturing process. Moreover, the simplicity of the purification via column chromatography suggests that the process can be adapted for continuous flow chemistry or larger batch reactors with minimal engineering hurdles, facilitating a smooth transition from laboratory benchtop to multi-ton commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, derived directly from the experimental data and claims within the patent documentation. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this method into their existing manufacturing workflows. The answers provided reflect the specific advantages and operational parameters defined in CN109776507B, ensuring accuracy and relevance for decision-makers.
Q: What are the key advantages of the Selectfluor/AgNO3 system over traditional carboxylic acid methods?
A: The Selectfluor/AgNO3 system eliminates the need for alkyl carboxylic acids, offering broader substrate universality and operating in a greener aqueous phase with higher yields.
Q: How does this method impact the purity profile of the final quinoline derivative?
A: The mild reaction conditions (50°C) and specific radical mechanism minimize side reactions, resulting in a cleaner crude product that simplifies downstream purification.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the use of water as a co-solvent and commercially available reagents like Selectfluor makes the process highly scalable and economically viable for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Methyl-4-(tetrahydrofuran-2-yl) Quinoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced C-H functionalization technologies in accelerating drug discovery and development pipelines. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the Selectfluor/AgNO3 catalyzed route are seamlessly translated into industrial reality. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-methyl-4-(tetrahydrofuran-2-yl) quinoline derivative meets the highest international standards. We are committed to delivering high-purity pharmaceutical intermediates that empower our clients to bring life-saving therapies to market faster and more efficiently.
We invite you to collaborate with us to leverage this cutting-edge synthesis technology for your specific project needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our expertise can optimize your supply chain and reduce your overall manufacturing costs.
