Scalable Synthesis of Fungicidal Pyrido[4,3-d]pyrimidine Derivatives for Global Agrochemical Supply Chains
The agricultural chemical sector is constantly evolving, driven by the urgent need for more effective and environmentally sustainable crop protection agents. Patent CN101323617B introduces a significant advancement in this domain by disclosing a novel class of polysubstituted pyrido[4,3-d]pyrimidine derivatives with potent sterilization activity. These compounds, represented by the general structural Formula I, encompass three distinct sub-families (Formula I-1, I-2, and I-3) that have demonstrated remarkable efficacy against a wide spectrum of phytopathogens. The innovation lies not only in the biological potency but also in the versatility of the chemical scaffold, which allows for extensive structural modification to optimize physicochemical properties and bioavailability. As a leading entity in fine chemical manufacturing, we recognize the strategic importance of such intermediates in developing next-generation fungicides that can combat resistant strains of cotton fusarium wilt, rice sheath blight, and cucumber gray mold.
![General Structural Formula I showing the core pyrido[4,3-d]pyrimidine scaffold with variable substituents R, X, Y, Z, and R1](/insights/img/pyrido-pyrimidine-synthesis-agrochemical-supplier-20260313200321-01.png)
The structural diversity offered by this patent is substantial, covering variations where R can be alkyl, phenyl, or substituted benzyl groups, while X represents oxygen, sulfur, or nitrogen linkers. This flexibility is crucial for medicinal chemists and agrochemical formulators aiming to fine-tune the lipophilicity and metabolic stability of the final active ingredient. The core pyrido[4,3-d]pyrimidine skeleton is known for its stability and potential for interaction with biological targets, yet traditional synthesis methods often suffer from low yields or require hazardous conditions. The methodologies described in this patent provide a robust framework for accessing these valuable heterocycles, positioning them as high-priority targets for reliable agrochemical intermediate supplier networks seeking to diversify their portfolio with high-efficacy fungicidal scaffolds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of fused pyrimidine systems has presented significant challenges in process chemistry. Conventional routes often rely on high-temperature cyclizations that can lead to decomposition of sensitive functional groups or the formation of complex impurity profiles that are difficult to separate. Many traditional methods necessitate the use of strong bases or corrosive acids, which pose safety risks and increase the cost of waste disposal and equipment maintenance. Furthermore, older synthetic pathways frequently lack regioselectivity, resulting in mixtures of isomers that reduce the overall yield of the desired pharmacophore. For procurement managers, these inefficiencies translate into higher raw material costs and unpredictable lead times, as batch-to-batch consistency becomes harder to maintain. The reliance on scarce or expensive catalysts in some legacy processes further exacerbates the economic burden, making cost reduction in fungicide manufacturing a critical objective for the industry.
The Novel Approach
The synthetic strategy outlined in Patent CN101323617B offers a transformative solution to these longstanding issues by utilizing mild and efficient cyclization protocols. For the synthesis of Formula I-1 and I-2 compounds, the process begins with the reaction of a substituted aminopyridine with triethyl orthoformate to generate a pyridine ether amidine intermediate. This key intermediate then undergoes ring closure with primary amines or ammoniacal liquor under relatively gentle conditions, often at room temperature or with moderate heating. This approach significantly reduces energy consumption and minimizes the risk of thermal degradation. For the Formula I-3 series, the patent describes an innovative route involving the formation of a phosphinimine intermediate using triphenylphosphine and hexachloroethane, followed by reaction with aromatic isocyanates. This method allows for the introduction of diverse amine substituents with high precision.
The versatility of this novel approach is evident in its ability to accommodate a wide range of substituents without compromising yield or purity. By avoiding harsh reaction conditions, the process preserves the integrity of sensitive functional groups, thereby expanding the chemical space available for optimization. This is particularly advantageous for R&D directors who require rapid access to diverse analog libraries for structure-activity relationship (SAR) studies. The simplified workup procedures, often involving straightforward filtration or recrystallization, further enhance the operational efficiency of the manufacturing process. Consequently, this methodology supports the commercial scale-up of complex agrochemical intermediates, ensuring a stable and continuous supply of high-quality materials for downstream formulation development.
Mechanistic Insights into Ether Amidine Cyclization and Phosphinimine Chemistry
The mechanistic pathway for generating the pyrido[4,3-d]pyrimidine core involves a fascinating sequence of condensation and nucleophilic attack steps. In the formation of Formula I-1 and I-2 derivatives, the initial reaction between the aminopyridine and triethyl orthoformate proceeds via the elimination of ethanol to form an imidate ether species. This electrophilic intermediate is then susceptible to nucleophilic attack by the exocyclic amine nitrogen of the incoming primary amine or ammonia. The subsequent intramolecular cyclization is driven by the formation of the aromatic pyrimidine ring, which provides a strong thermodynamic driving force for the reaction. The presence of electron-withdrawing groups, such as the cyano group at the 8-position, further activates the system towards nucleophilic attack, facilitating the ring closure even under mild conditions. This mechanistic understanding allows for precise control over reaction parameters, ensuring high conversion rates and minimizing side reactions.
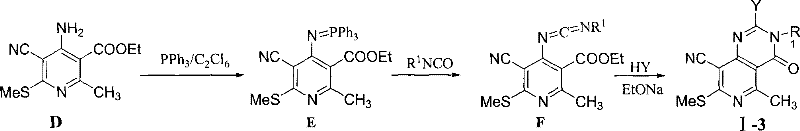
For the Formula I-3 series, the mechanism diverges through the formation of a phosphinimine intermediate, a classic example of aza-Wittig type chemistry. The reaction of the aminopyridine with triphenylphosphine and hexachloroethane generates an iminophosphorane species, which is highly reactive towards isocyanates. This step forms a carbodiimide intermediate, which subsequently undergoes nucleophilic attack by an amine or alcohol to close the ring and form the pyrimidin-4-one structure. The use of sodium alkoxide as a base in the final cyclization step ensures deprotonation and drives the equilibrium towards the product. This pathway is particularly valuable for introducing urea-like linkages and carbonyl functionalities that are often challenging to install via direct condensation. The ability to toggle between these two distinct mechanistic routes provides manufacturers with the flexibility to select the most cost-effective and scalable pathway based on the specific substitution pattern required for the target molecule.
How to Synthesize Polysubstituted Pyrido[4,3-d]pyrimidine Derivatives Efficiently
The practical execution of these synthetic routes requires careful attention to stoichiometry and reaction monitoring to maximize yield and purity. The preparation of the ether amidine intermediate typically involves heating the aminopyridine with an excess of triethyl orthoformate, optionally with a catalytic amount of acid such as acetic anhydride or p-toluenesulfonic acid. Once the intermediate is isolated, it can be reacted with the desired amine in a polar aprotic solvent like DMF or acetonitrile. For the phosphinimine route, strict exclusion of moisture is critical during the formation of the iminophosphorane to prevent hydrolysis. The subsequent reaction with isocyanates should be performed under an inert atmosphere to ensure high conversion. Detailed standardized synthesis steps see the guide below.
- Preparation of Ether Amidine Intermediate: React substituted aminopyridine with triethyl orthoformate under catalytic acidic conditions at elevated temperatures to form the key pyridine ether amidine precursor.
- Cyclization to Target Scaffold: Treat the ether amidine intermediate with primary amines or ammoniacal liquor in organic solvents to effect ring closure, yielding the dihydropyrido[4,3-d]pyrimidine core.
- Alternative Phosphinimine Route: For carbonyl-substituted variants, generate a phosphinimine from the aminopyridine using triphenylphosphine and hexachloroethane, followed by reaction with isocyanates and nucleophilic ring closure.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of these synthetic methodologies offers profound benefits for supply chain stability and cost management. The reliance on commodity chemicals such as triethyl orthoformate, triphenylphosphine, and common amines means that raw material sourcing is robust and less susceptible to market volatility. This availability ensures enhanced supply chain reliability, allowing manufacturers to maintain consistent production schedules without the risk of bottlenecks associated with specialty reagents. Furthermore, the mild reaction conditions reduce the energy footprint of the manufacturing process, aligning with global sustainability goals and potentially lowering utility costs. The simplified purification steps, often requiring only recrystallization or filtration, minimize solvent usage and waste generation, contributing to a greener manufacturing profile that is increasingly demanded by regulatory bodies and end-users.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of ambient or moderate temperature conditions significantly lower the operational expenditure associated with production. By streamlining the synthesis to fewer steps with higher atom economy, the overall cost of goods sold is reduced, providing a competitive edge in the marketplace. The avoidance of complex chromatographic purifications in favor of crystallization further drives down processing costs, making these intermediates economically viable for large-volume agrochemical applications.
- Enhanced Supply Chain Reliability: The use of widely available starting materials mitigates the risk of supply disruptions that can plague specialized chemical supply chains. This resilience is critical for maintaining uninterrupted production of active ingredients, ensuring that farmers have timely access to crop protection solutions. The robustness of the chemistry also means that technology transfer to different manufacturing sites is straightforward, facilitating geographic diversification of supply sources to further de-risk the supply chain.
- Scalability and Environmental Compliance: The processes described are inherently scalable, having been demonstrated to work effectively from gram to multi-kilogram scales in the patent examples. The reduced generation of hazardous waste and the lower energy requirements simplify environmental compliance, reducing the regulatory burden on manufacturing facilities. This ease of scale-up supports the commercialization of new fungicidal active ingredients, accelerating their time to market and maximizing their potential impact on global food security.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical specifications and application potential of these pyrido[4,3-d]pyrimidine derivatives. Understanding these aspects is vital for stakeholders evaluating the integration of these intermediates into their development pipelines. The answers provided are derived directly from the technical disclosures and experimental data within the patent documentation, ensuring accuracy and relevance for decision-making processes.
Q: What is the spectrum of fungicidal activity for these pyrido[4,3-d]pyrimidine derivatives?
A: The compounds exhibit significant inhibitory effects against a broad range of agricultural pathogens, including cotton fusarium wilt, rice sheath blight, cucumber gray mold, wheat gibberella, apple ring spot, and capsicum blight, making them versatile candidates for crop protection formulations.
Q: How does the novel synthetic route improve upon conventional heterocycle manufacturing?
A: The patented methodology utilizes mild cyclization conditions, often at room temperature or moderate heating, avoiding the need for harsh reagents or expensive transition metal catalysts. This simplifies purification and enhances overall process safety and yield consistency.
Q: Are these intermediates suitable for large-scale commercial production?
A: Yes, the synthesis relies on readily available starting materials like substituted aminopyridines and triethyl orthoformate. The reaction steps are robust and amenable to scale-up, supporting reliable supply chains for agrochemical active ingredient manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrido[4,3-d]pyrimidine Derivatives Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from laboratory discovery to industrial reality. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch against the highest industry standards. We understand the critical nature of agrochemical supply chains and are dedicated to providing a reliable source of high-purity intermediates that meet the demanding requirements of global crop protection companies. Our technical team is well-versed in the nuances of heterocyclic chemistry and can offer valuable insights into process optimization and impurity control.
We invite you to engage with our technical procurement team to discuss your specific requirements and explore how our capabilities can support your strategic goals. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of partnering with us for your pyrido[4,3-d]pyrimidine needs. We encourage you to reach out for specific COA data and route feasibility assessments to ensure that our solutions align perfectly with your development timelines and quality expectations. Let us be your partner in advancing the next generation of agricultural solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
