Advanced Synthesis of Nitrate-Group-Containing Dihydromyricetin Derivatives for Antiviral Applications
Advanced Synthesis of Nitrate-Group-Containing Dihydromyricetin Derivatives for Antiviral Applications
The pharmaceutical landscape for antiviral therapeutics is constantly evolving, driven by the urgent need for more effective treatments against persistent viral infections like Hepatitis B. Patent CN107311973B introduces a groundbreaking class of nitrate-group-containing dihydromyricetin derivatives that represent a significant leap forward in medicinal chemistry. These compounds are not merely structural analogs but are engineered prodrugs designed to release Nitric Oxide (NO) in vivo, leveraging the gasotransmitter's potent ability to induce apoptosis in viral-infected cells. For R&D directors and procurement specialists seeking high-purity pharmaceutical intermediates, this patent outlines a robust, semi-synthetic pathway that transforms a naturally abundant flavonoid into a sophisticated therapeutic agent. The strategic modification of the dihydromyricetin scaffold addresses critical limitations of current nucleoside analogs, such as drug resistance and rebound effects, offering a novel mechanism of action that targets the early stages of virus replication.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to utilizing dihydromyricetin, a major component of Ampelopsis grossedentata, have been hindered by its inherent physicochemical properties and limited bioactivity profile. While the natural extract possesses anti-inflammatory and hepatoprotective qualities, its direct application as a potent antiviral agent is often compromised by rapid metabolism and insufficient potency against established viral loads. Furthermore, relying solely on extraction from plant sources introduces variability in purity and supply chain consistency, which are unacceptable for GMP-grade API manufacturing. Conventional synthetic modifications often struggle to functionalize the crowded flavonoid skeleton without destroying the sensitive catechol moieties or requiring harsh conditions that degrade the core structure. This lack of precise chemical control results in complex impurity profiles that are difficult to characterize and remove, posing significant regulatory hurdles for clinical advancement.
The Novel Approach
The methodology disclosed in CN107311973B overcomes these challenges through a clever dual-modification strategy that preserves the antioxidant core while installing a pharmacologically active nitrate ester side chain. By first protecting the phenolic hydroxyl groups as acetates or methyl ethers, the synthesis creates a stable platform for selective acylation at the C-3 position. This regioselectivity is crucial for maintaining the structural integrity required for biological activity. The subsequent introduction of the nitrate group via an amino-alcohol linker effectively converts the molecule into a targeted NO donor. This approach not only enhances the antiviral efficacy through a synergistic mechanism but also improves the lipophilicity of the molecule, potentially aiding in membrane permeability. The versatility of this route is demonstrated by the ability to vary the R groups, allowing for the generation of a diverse library of derivatives tailored for specific pharmacokinetic profiles.
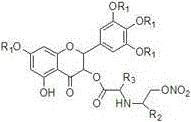
This structural flexibility is a key advantage for intellectual property positioning and optimization. As shown in the specific examples provided in the patent, variations in the amino-alcohol portion (R2 and R3) allow chemists to fine-tune the rate of NO release and the overall stability of the prodrug. This level of molecular engineering is far superior to simple extraction or random derivatization, providing a clear path for developing a next-generation antiviral candidate with a defined mechanism of action and manageable safety profile.
Mechanistic Insights into Regioselective Acylation and Nitration
The core chemical transformation in this patent relies on a sequence of protection, activation, and coupling reactions that demonstrate high chemoselectivity. The initial step involves the protection of the multiple hydroxyl groups on the dihydromyricetin backbone. Using acetic anhydride in pyridine or methyl iodide with potassium carbonate ensures that the reactive phenolic sites are masked, preventing unwanted side reactions during subsequent steps. This protection strategy is vital because the C-3 hydroxyl group, which is the target for functionalization, has different acidity and steric environments compared to the aromatic hydroxyls. Once protected, the intermediate is subjected to acylation using chloroacetyl chloride or 2-chloropropionyl chloride. This reaction installs a reactive chloro-acetyl handle at the C-3 position, creating an electrophilic site ready for nucleophilic attack.
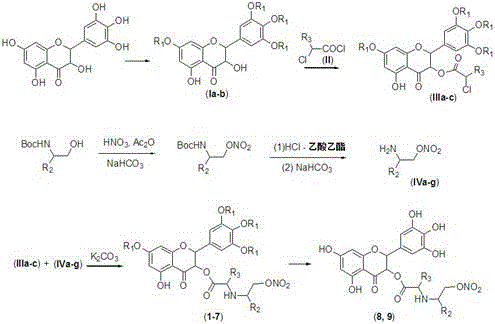
The second critical mechanistic component is the preparation of the nitrate-bearing nucleophile. The patent describes the nitration of Boc-protected amino alcohols using fuming nitric acid and acetic anhydride. This generates a nitrate ester on the alcohol functionality while keeping the amine protected. Subsequent deprotection of the amine yields a free amino-nitrate species. In the final coupling step, this amine acts as a nucleophile, displacing the chloride on the acylated dihydromyricetin intermediate. The use of mild bases like potassium carbonate in polar aprotic solvents like DMF facilitates this SN2-type substitution without compromising the sensitive nitrate ester bond. This careful orchestration of reaction conditions ensures that the high-energy nitrate group remains intact throughout the synthesis, resulting in the final stable derivative.
How to Synthesize Nitrate Dihydromyricetin Derivatives Efficiently
The synthesis protocol detailed in the patent offers a reproducible method for producing these complex molecules with high purity. The process begins with the isolation or purchase of high-quality dihydromyricetin, followed by the protection steps described earlier. The key to success lies in the rigorous control of reaction temperatures and stoichiometry, particularly during the nitration step which involves strong acids. The final coupling reaction requires careful monitoring to ensure complete conversion while minimizing the formation of quaternary ammonium salts or other byproducts. Purification is typically achieved through silica gel column chromatography, utilizing gradient elution systems to separate the product from unreacted starting materials and side products. For those looking to implement this in a pilot plant setting, the standardized nature of these organic transformations suggests a straightforward translation from bench to kilo-lab scale.
- Protect the hydroxyl groups of dihydromyricetin using acetic anhydride or methyl iodide to form intermediates Ia or Ib.
- Perform acylation at the C-3 position using chloroacetyl chloride or 2-chloropropionyl chloride to generate intermediate III.
- Synthesize amino-alcohol nitrate intermediates (IV) via nitration of Boc-protected amino alcohols, followed by deprotection.
- Couple intermediate III with intermediate IV using potassium carbonate in DMF to form the final nitrate ester derivatives.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain and cost perspective, the synthetic route described in CN107311973B presents several compelling advantages over alternative biotechnological or complex total synthesis methods. The reliance on commodity chemicals such as acetic anhydride, potassium carbonate, and common solvents like DMF and ethyl acetate significantly reduces the raw material cost burden. Unlike processes that require precious metal catalysts or specialized enzymes, this purely chemical approach utilizes widely available reagents that can be sourced from multiple global suppliers, mitigating the risk of single-source dependency. Furthermore, the reaction conditions are relatively mild, typically proceeding at room temperature or with moderate heating, which translates to lower energy consumption and reduced operational expenditures in a manufacturing environment. The absence of heavy metals in the catalytic system also simplifies the downstream purification process, eliminating the need for expensive scavenging resins or complex metal removal protocols.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts is a major driver for cost efficiency. In traditional cross-coupling reactions, the cost of palladium or platinum catalysts, combined with the stringent requirements for residual metal limits in APIs, adds significant expense. By utilizing a nucleophilic substitution strategy with inexpensive inorganic bases, this process drastically lowers the cost of goods sold (COGS). Additionally, the high yields reported in the examples, often exceeding 70-80% for key steps, minimize waste and maximize the throughput of the final active pharmaceutical ingredient. This efficiency directly impacts the bottom line, making the commercial production of these derivatives economically viable even at large scales.
- Enhanced Supply Chain Reliability: The starting material, dihydromyricetin, is a natural product extracted from Ampelopsis grossedentata, a plant that is widely cultivated. However, the semi-synthetic nature of this process means that manufacturers are not solely dependent on seasonal harvests for the final drug substance. By converting the natural extract into stable protected intermediates, production can be decoupled from agricultural cycles. The synthetic reagents required for the modification steps are bulk petrochemical derivatives with stable global supply chains. This hybrid approach ensures a consistent and reliable supply of the critical intermediate, reducing the risk of shortages that often plague purely botanical supply chains.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability potential due to the use of standard unit operations such as stirring, filtration, and distillation. The solvents used, while requiring proper management, are common in the industry and have well-established recovery and recycling protocols. The avoidance of hazardous reagents like thionyl chloride or phosphorus halides in the main coupling steps further enhances the environmental profile of the synthesis. This aligns with modern green chemistry principles, facilitating easier regulatory approval and reducing the environmental footprint of the manufacturing facility. The robustness of the reaction conditions suggests that the process can be safely scaled from grams to tons without encountering significant exothermic hazards or stability issues.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these nitrate derivatives. Understanding the nuances of the synthesis and the biological rationale is essential for stakeholders evaluating this technology for licensing or procurement. The answers provided are derived directly from the technical disclosures within the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: What is the primary biological advantage of adding a nitrate group to dihydromyricetin?
A: The addition of a nitrate group transforms the molecule into a Nitric Oxide (NO) donor. NO acts on the early stage of virus replication, preventing diffusion and promoting elimination, thereby enhancing the antiviral efficacy against Hepatitis B Virus compared to the parent compound.
Q: How does this synthesis route address the solubility issues of natural flavonoids?
A: Natural dihydromyricetin often suffers from poor bioavailability. By introducing lipophilic acyl groups and specific nitrate esters at the C-3 position, the derivatives exhibit improved physicochemical properties, facilitating better cellular uptake and metabolic stability.
Q: Are the reagents used in this patent scalable for industrial production?
A: Yes, the process utilizes common industrial reagents such as acetic anhydride, potassium carbonate, and DMF. The avoidance of expensive transition metal catalysts and the use of standard purification methods like silica gel chromatography make the route highly amenable to commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nitrate Dihydromyricetin Derivative Supplier
As the demand for novel antiviral agents continues to grow, the ability to reliably manufacture complex flavonoid derivatives becomes a critical competitive advantage. NINGBO INNO PHARMCHEM stands at the forefront of this capability, offering comprehensive CDMO services tailored to the specific needs of the pharmaceutical industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from clinical trials to market launch is seamless. We understand the stringent purity specifications required for antiviral intermediates and employ rigorous QC labs to verify every batch against the highest international standards. Our commitment to quality assurance means that every gram of nitrate dihydromyricetin derivative we produce meets the exacting criteria necessary for downstream drug formulation.
We invite potential partners to engage with our technical procurement team to discuss how we can support your specific project requirements. Whether you need a Customized Cost-Saving Analysis for your supply chain or require specific COA data to validate our capabilities, we are ready to provide the transparency and expertise you need. By leveraging our optimized synthetic routes and robust manufacturing infrastructure, we can help you reduce lead time for high-purity pharmaceutical intermediates and accelerate your time to market. Contact us today to request route feasibility assessments and discover how NINGBO INNO PHARMCHEM can be your strategic partner in developing the next generation of antiviral therapeutics.
